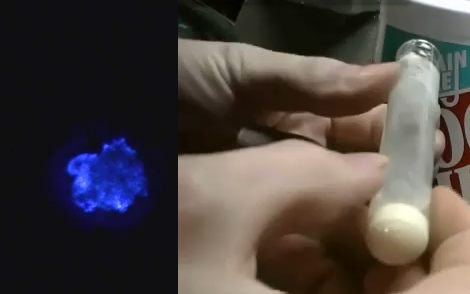
[Jeri Ellsworth] continues her experiments with electroluminescence, this time she’s making EL ink. The ink she’s looking for is Zinc Sulfate in a solution. The process she chose is to re-dope some glow powder so that it can be excited by the field around an AC current. In her video (embedded after the break) she talks about the chemical properties she’s after by detailing a cubic lattice of zinc and sulfur atoms with an added copper atom (adding that atom is a process called doping).
The quick and dirty synopsis of the experiment starts by washing the glow powder with dish soap to acquire zinc sulfide crystals. Then she combined copper sulfate and zinc shavings from the inside of a modern penny to yield copper metal and zinc sulfate suspended in solution. That was mixed with the zinc sulfide from the glow powder washing and doped with a little more copper sulfate. The excess liquid is poured off, the test tube is capped with glass frit, and the whole thing hits the kiln to start the reaction. The result glows when excited by alternating current, but could have been improved by adding chlorine atoms into the mix.
We’re excited every time we see one of [Jeri’s] new chemistry hacks. We’d love to see more so if you’ve come across interesting chemistry experiments during your Internet travels, please let us know about them. Just make sure you have some idea of what you’re doing when working with chemicals… safety first.
[youtube=http://www.youtube.com/watch?v=pmQqdYrn9g8]
















nice..
reckon this can be used to make multiple colours?
apparently doping with other rare earths changes the colour.
I’m very surprised EL generates light by pretty much the same mechanism as LEDs albeit on a different scale and in different conditions.
Marry Me, Jeri! LOL
am I the only person who finds it really irritating when projects are documented solely as youtube videos? I’d much rather text + images, way easier to skim through and faster to pick up.
anyway, this is cool, I’ve got some chemistry-related projects I’ve been cooking up, this could fit in well :p
nave.notnilc: Hmmm. You could watch the video to write some text and capture some images. Then post them here in the comments so that people like you could skim through and pick up faster.
Congratulations, Jeri. You are awesome and deserve the success.
Zinc sulfate could easily be made in large quatities with Zinc metal and Sulfuric acid.
Or you could buy the mineral Wurtzite (Zn,Fe)S and crush it up(not an easy task I imagine).
Zinc Chloride, for additional doping, can again be made by dissolving zinc metal in HCl. Both acids are readily available in a variety of concentrations and purities.
To anyone else who was wondering apparently ‘rather high voltages’ are from ~50-200V according to a response on her youtube page.
A fascinating little factoid:
So, a young lady I know was given a great deal of jewelry of the faux-diamond variety; specifically, artificial diamonds. I explained to her that they were basically extra-large LEDS without the wrapper.
She scoffed at this. Two wires and a bit of bias current later, I made one of her earrings glow quite brightly. She was impressed, and said “Cool! Can you show my mom?”.
Naturally, I ended up blowing a tiny hole in the earring during the second demo. It did not end well. I traded her a real ruby from thailand circa 1960 (and now fake ones are everywhere) for the earrings.
Jeri,
I know your interests run towards HV, but you might find ultrasonic bubble glow vaguely interesting.
Monty already did some experiments on ultrasonic bubble glow in Jeri’s lab, iirc. Fun times.
@bilbao bob What type of jewel should I search for if I want to reproduce this? Cubic zirconia?
@bilbao bob @LoneTech Yes Monty did set up his sono-luminescence rig and we made a video. He got the bubble to form, but it was either not generating light or the other light sources in the room were preventing us from seeing it.
i really like these vids, pretty cool.
one thing i was wondering about was, in that first diagram with the valance and conduction bands, are the arrows pointing in the wrong direction?
Jeri,
You have to work hard to get good luminescence. Even then, it’s not gonna look like the fusion reactor that seems to appear in american superhero movies.
OK.
I want to point out that the glow is a point contact glow – it is visible through the gem, but you won’t be creating a super-hero laser effect without a lot of work. On the plus side, it’s often green. Any impurites in the silicon will create different colors, but you already knew that.
The jewel is called moissanite, and it is rapidly going out of fashion for a variety of reasons. JC Penny in the US was a major retailer, but I don’t know who else sold/sells this stuff. The company is going down the tubes as far as I know.
Moissanite is basically the same old circa 1900 silicon carbide material used for radios, and it essentially was the ancestor of diodes and silicon circuits in general. I think an american found the glow, but a russian physicist published a paper or two on it. Olav? Olev? Something like that.
You basically grip the material in an alligator clip or any kind of metallic holder (I used the earring base, which was crimped on but had holes underneath) and then using a cat’s whisker/needle/whatever, apply the bias current to it.
The physics are almost identical to ordinary LEDs, as the material is the same. Current limiting is a good idea.
You can crush up moissanite and build your own low quality LEDs by creating the correct shape, inserting it into the plastic leaving a gap for the electrons and then sealing it. Vacuum pumps help, but not necessary.
They typically use a metal substrate and then apply a very small particle of silicon carbide or carborundum to save material. The gems are the same thing in large purified form.
The plastics used in old LEDs are recoverable through temp controlled melting, btw. Any transparent plastic should be usable for light devices.
Finally, if you watch the currents, you can have multiple anodes/cathodes on a single gem. It isn’t easy to do.
I blame this experiment on absinthe. It’s way cooler to use jewelry. Good luck!