We humans like to think of ourselves as the pinnacle of evolution on the planet, but that’s just a conceit. It takes humans roughly twenty years to reproduce, whereas some bacteria can make copies of themselves every 20 minutes. Countless generations of bacteria have honed and perfected their genomes into extremely evolved biological machines.
Most bacteria are harmless, and some are quite useful, even tasty – witness the lactofermented pickles and sauerkraut I made this summer. But some bacteria are pathogenic nightmares that have swarmed over the planet and caused untold misery and billions of deaths. For most of human history it has been so – the bugs were winning. Then a bright period dawned in the early 20th century – the Era of Antibiotics. At last we were delivered from the threat of pestilence, never more to suffer from plague and disease like our unfortunate ancestors. Infections were miraculously cured with a simple injection or pill, childhood diseases were no longer reaping their tragic harvest, and soldiers on the battlefield were surviving wounds that would have festered and led to a slow, painful death.
Now it seems like this bright spot of relief from bacterial disease might be drawing to an end. Resistant strains of bacteria are in the news these days, and the rise of superbugs seems inevitable. But is it? Have we run out of tools to fight back? Not quite yet as it turns out. But there’s a lot of work to do to make sure we win this battle.
Artificial Selection in Action
![Alexander Fleming [via Wikipedia]](https://hackaday.com/wp-content/uploads/2015/09/alexander_fleming.jpg?w=400)
In the 75 years since then, huge advancements in antibiotic therapy beyond penicillin have been made, and literally millions of lives have been saved by the tetracyclines, sulfonamides, cephalosporins and quinolones that have followed it. Each antibiotic class has its own method of action, and each medicine has a particular niche that it exploits – penicillins disrupt bacterial cell wall synthesis, for example, while tetracyclines inhibit protein synthesis in bacterial ribosomes. But each little trick that we exploit to kill off bacteria has an unintended consequence – it leaves survivors behind. Random mutations and genetic reassortment lead to the chance that a handful of cells in the infection will be able to resist the antibiotic being used. When the susceptible cells are killed off, the resistant cells are not only left behind, they’re left with an open playing field and tons of resources to exploit. The survivors reproduce, pass their resistance on to the next generation, and pretty soon you’ve artificially selected a population of superbugs.
Obviously it’s all a little more complicated than that. Our immune systems are hard at work during an infection, and once the susceptible cells are culled it generally does a pretty good job of mopping up the small number of resistant bacteria. And resistance does not necessarily mean “antibiotic-proof” – it might just mean a cell is able to put up more of a fight to the antibiotic of choice. This is the reason for long courses of antibiotic therapy – knock the infection back with the first dose or two, then keep the levels of the medicine up to keep up the pressure on the tougher bugs so the immune system can mop up. Quit the medicine early, and you’re just giving the resistant bugs a chance to explode in population and cause a real problem.
Misuse of antibiotics has also contributed to the current state of affairs. Patients generally want to walk out of their doctor’s office with something to show for the effort and expense, and being told to go home and wait out a viral infection often doesn’t sit well. Going home with a useless course of antibiotics used to be a common practice and has no doubt contributed to the selection of the resistant strains of bacteria we’re seeing today.
Ancient Wisdom
But isn’t there a disconnect here? If we are so susceptible to bugs, and we only figured out how to fight them off less than a century ago, how did we even get this far as a species? While it’s true that modern antibiotics are only a recent development, traditional treatment of infection goes back a lot further. Traces of tetracycline, which strongly binds to the mineral matrix of bones and teeth, can be found in human skeletal remains from 1700 years ago. It’s not clear whether they were purposely dosing themselves or just picking up tetracycline from naturally occurring soil bacteria, but they were exposed and probably benefited from it.

What’s Next?
As fruitful as it may prove to tap into ancient wisdom, the fact remains that the microbes will always be one step ahead of us. Whether we throw a new synthetic molecule or an ancient potion at an infection, random mutations and generation times measured in minutes will combine to quickly evolve resistance to the next therapy. But what if there was a way to strip antibiotic resistance from bacteria? Then no matter what new tricks they learn, we’d be able to sensitize them to antibiotics and maybe gain a little ground in the fight against infection.
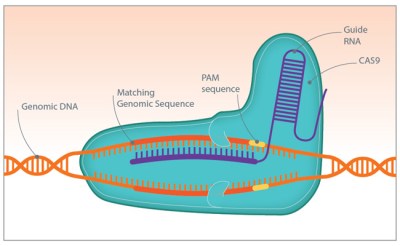
CRISPR is a really powerful tool for gene editing, and one that has applications for overcoming antibiotic resistance. In an ironic twist, researchers are packing CRISPR/Cas systems into phages and using them to attack bacteria. The CRISPR system is programmed to search for and destroy the sequences that code for antibiotic resistance, like the beta-lactamase protein that confers penicillin resistance. The bacteria are then vulnerable to antibiotics they had previously been able to stand up to.
There are limits to what CRISPR can accomplish in the fight against infection. One obvious problem is finding a phage that attacks each bug you want to kill, and being able to genetically modify it with a CRISPR payload. And the bugs still have the evolutionary upper hand, since they can rapidly try out random mutations that may overcome the CRISPR attack. But it’s at least another tool for us to use, and one that allows us to rapidly respond to new threats. Moreover, it’s a very approachable technology in terms of equipment and instrumentation. Indeed, CRISPR has become the hot new technique for biohackers who seek to create everything from better yeasts for craft brewed beers to vegan cheese. Perhaps some of these biohackers will turn their attention to the problem of antibiotic resistance too.
Even though the evolutionary deck is clearly stacked against us, I don’t think we’re done for quite yet. While the bugs have the numbers, we have our own evolutionary gift – a great big forebrain. If we put enough of them on the problem, some breakthrough will happen. So while the Era of Antibiotics isn’t necessarily at an end, it probably is at a really interesting inflection point.
[MRSA SEM Image via Wikipedia]















What’s the difference between “biohacker” and, say, microbiologist?
I would use the term “hacker” when the person has no professional education/training in the area.
Which site hosts the article about him/her.
I think the same as between “hacker” and an “engineer”.
Antibiotics will be the end of us all. They kill off our friendly gut bacteria which leads to an epidemic of obesity and inflammatory diseases. Plus, the target “bad” bacteria just keep adapting, and ever will it be so. There has to be a way to live with our microbes instead of engaging in this ridiculous antibiotics arms race. We will lose.
Yes, there has to be a way to live with microbes specifically adapted to exploit our own bodies to propagate themselves at the expense of our health and lives. Enjoy your free-range, organic staphylococcus aureus, I guess.
For an actual, non-snarky response: fighting disease is inherently an ongoing arms race, regardless of whether you do it with drugs, genetic engineering, or natural remedies (reminder that penicillin came from mold). We’ll never “win,” but if we don’t try we risk a return to the days of people dying or being permanently disabled in huge numbers by what would otherwise be unremarkable infections. At the very least people would suffer needlessly.
I think the way forward is to acknowledge that there are some problems that free markets and personal responsibility can’t or won’t handle, at least not before it’s too late. There comes a point where the public is sufficiently resistant to education and the tragedy of the commons builds enough momentum that the only reasonable solution is to outlaw prophylactic antibiotics for livestock, and legally coerce doctors into refusing antibiotics to individuals not actually suffering from a bacterial infection.
IIRC, in a number of countries, a decade or two ago, antibiotics were available to the public without a doctor’s prescription. Hopefully that has changed, but a lot of damage was done in this regard through the misuse of antibiotics. (i.e. it is not just the doctor’s or farmer’s fault)
Yeah actually I was thinking of gut bacteria which actually produce bacterocins that kill staf and c diff. Antibiotics are what make them more deadly. For example, currently the only way to help people with recurrent antibiotic resistant c diff is to give them FMTs from healthy donors and to let the bacteria from the FMT kill c.diff. So here we have a situation where antibiotics are no longer effective, but bacterocins produced by living friendly bacteria are saving human lives.
The problem for all of us is that we have evolved in symbiosis with microbes over the millenia, and the antibiotics we have developed are a double-edged sword. On the one hand, lives are saved and people live a little longer. The cost is that each time a course of antibiotics is finished we kill off large swathes of our human microbiome be it skin, gut, respiratory, urinary, oral… this leads to lower diversity of helpful microbes which ping our immune systems, keep pathogenic bacteria in check, and produce metabolites which stave off certain types of cancer, and chronic inflammatory diseases. So the cost is not just antibiotic-resistant genes in s. Aureus or c.diff. The cost is also that each of us ends up with less critically important bacteria than we started out our lives with. I really do wonder a lot what the general health of people in western societies will be like in the future as each successive generation of women with antibiotic impaired microbiomes goes forth and attempts to bear healthy children.
Beyond antibiotic resistance in human bacteria, there is also the issue of livestock fattening with antibiotics and the excretion of these antibiotics into waterways and soil, and how this affects soil and water bacteria we don’t even know about. How does it affect our food chain and food security?
Hopefully I will have shuffled off this mortal coil before the c diff and s.aureus hits the fan.
human bacterial culture is far from that stable nor uniform, on top of that we have naturally eaten antibiotics for quite a long time in cheeses, as with anything else it is far from as simple as antibiotics bad, own bodily function good.
Eating “active culture” yogurt after an antibiotic regimen only replaces 1 or 2 species of bacteria out of the hundreds that were killed off. It is better than nothing, but FMT replaces maybe “all” of the beneficial bacteria.
One thing anyone can do is to boycott factory farming. Ideally, they should ban the use of antibiotics in factory farming.
There are a number of ancient antibiotics which have not been explored by “biohackers”. Case in point: The ancient Israelites where known by others as the “garlic-eaters”. It appears that copious doses of garlic (Allium sativum) tends to be a prophylactic to infectious diseases. And if taken during an infectious attack it tends to reduce the ill effects of the microorganism. Also large doses of Ascorbic acid (Formula: C6H8O6) tends to have a prophylactic effect by reinforcing nitrogen in cellular membranes effectively thwarting cell membrane breach by bacteria and viruses. But this is only prophylactic and not reactive in benefit. It appears that the use of rose hips during the black plague was on the right track. However, some only used the rose petals which proved worthless. The hips have large amounts of Ascorbic acid. However, mega doses tend to only be eliminated in urine quickly and more doses are indicated. Side affect could be liver damage in rare cases.
Another overlooked chemical agent to mitigate diabetes millitus in rats is Capsaicin (Formula: C18H27NO3 – found in hot sauce). Arguable this is off-topic as it does not deal with “war on antibiotic resistance” – per se.
And last but not least is: Ethanol (Formula: C2H5OH) has the ability to dissolve like a solvent non polar substances like microorganisms. How doctors think that ethanol can be resisted by a particular strain is ludicrous and a bit disingenuous. If you are non polar you will be dissolved no matter what strain of bacteria or virus you are.
I think the proper noun “biohacker” is just a cool nickname for microbiologist. Bio is in both nouns.
P.S. – My pet theory is someone designing a customized super T-Cell (or T lymphocytes). A natural killer that could focus on say a HIV and destroy it and only it. AIDS could become a thing of the past including several forms of cancer.
Where is the “Like” button?
quote Where is the “I’m trying to fit in” button? end quote
FTFY
Likewise we have seen a resurgence of packing wounds with sugar in Europe. This is something that goes all the way back to ancient egypt and probably before, Honey when packed in a wound is very effective as an anti bacterial.
Honey yes, sugar no.
It has nothing to do with the sugar content of the honey.
Also there is another chemical in a honeycomb called “Royal Jelly”. Allegedly it is used to maintain the health of the queen and it’s larvae. People have found some interesting medical advantages to ingesting Royal Jelly. I do not know too much about it. I’m going to research it. It is created by the honey bees. I have no idea of it’s molecular breakdown.
The difference between the ancient hackers and today is that we utilize a tool called “empiricism” or better known as “science.” Ascorbic acid (vitamin C) has been studied in prospective randomized trials for cancer and cardiovascular disease and never been shown to be of benefit(http://ajcn.nutrition.org/content/100/3/915.long and http://jama.jamanetwork.com/article.aspx?articleid=1028653). All it has ever been shown to do is increase the risk of kidney stones (http://archinte.jamanetwork.com/article.aspx?articleid=1568519). I’d love to see your data on its role in infection prevention.
Your comment on HIV is confusing, do you mean a t-cell or a natural killer cell? They are separate things. The problem so far has not been killing HIV, we have meds that will quickly wipe out any circulating virus and make one have undetectable viral loads in 3 months. The issue is going after latent HIV that has inserted itself into host genomes. A t-cell or natural killer cell would not be able to do this. Medications like histone deacetylase inhibitors are currently being investigated to go after that reservoir of infection.
I would think the “ancient hackers” as you say were using the epitome of empiricism through direct human experimentation. I don’t think the late Dr. Linus Pauling would agree with your negative assessment of Ascorbic acid (aka Vitamin C or VC). I have personally seen a person achieve full remission form massive doses of VC. Many other anecdotal stories were told to me of others. Maybe the scientific VC trials are defective or poorly planned out? I would imagine that Big Pharma has a financial vested interest in the trials being allegedly inconclusive. Also I did say VC is totally worthless in doing anything against an active infection. However, it works wonders as a prophylactic measure. I think Dr. Pauling showed that the cellular membrane is reinforced by nitrogen making it almost impossible for a microorganism to penetrate and doing it’s thing, making you sick.
Natural killer T cells (NKT cells – not to be confused with natural killer cells of the innate immune system).
I’m surprised the article didn’t talk about how promising phages themselves can be used to attack an infection. There’s been some promising research that demonstrates bacterial phages can be engineered in the lab to attack certain strains of bacteria.
Granted this is a bit unsettling through… a virus engineered in a lab with the intent to inject in to a human. Sounds like the beginning to a zombie apocalypse movie.
Of course, it’s “a virus engineered in a lab and injected into a human… that relies on a totally different sort of cell boundary than eukaryotes have, and a somewhat different genetic code…” – I’d put it at about the same level of worry as “I ate a plant that had a virus”.
Yep, phage therapy is what the USSR developed while the Allies were figuring out antibiotics… it is crazy how much they got developed, and then ALMOST got forgotten. Check out the book “The Forgotten Cure: The Past and Future of Phage Therapy” it is a great read!
Near the end of the 1954 movie “Apache” (starring Burt Lancaster as an Apache warrior), he is wounded by a gunshot
and rubs dirt in the wound. I always wondered why he did that, maybe it helps as a clotting agent, but maybe ancient wisdom incorporated the tetracyclines.
@Ren – “The colloquial medical advice “rub some dirt in it” appears to have some merit. Researchers at Arizona State University’s Biodesign Institute have been experimenting with different clays, and it appears in research presented in the journal PLoS ONE that they’ve come across a family of antibacterial clays capable of killing pathogens ranging from E. coli to methicillin-resistant Staphylococcus aureus, otherwise known as hard-to-kill MRSA.
Clays have been used as medical tools for ages, appearing in ancient medical texts going back as far as 3,000 B.C. Topically, they were used to treat wounds, a practice that became common in the 19th century. Early practitioners of clay therapy noted that clays tended to aid in healing, in reducing inflammation of wounds, and in pain management–though they couldn’t have known why exactly.
It turns out that’s probably because some clays–particularly clays rich in a certain group of metallic ions–work as antibacterial agents. In their study, the ASU researchers tested a variety of different clays with similar mineral composition but ranging compositions of metallic ions against E. coli and MRSA. They found that five metal ions–iron, copper, cobalt, nickel, and zinc–could fight the two bacterial strains, both of which are increasingly difficult to kill using standard antibiotics and antibacterials.
That doesn’t mean these clays are silver bullet or any kind of antibacterial panacea. Not all clays are created equal and some lack the necessary concentrations of the necessary metal ions. Moreover, clays can contain other metals as well, like cadmium, lead, mercury, and arsenic (if you weren’t paying attention in chem class, these are not metals you particularly want to introduce to your bloodstream).
But the researchers are optimistic that medicinal clays could find widespread use in certain therapeutic roles, particularly as bandaging agents as their absorptive and adhering characteristics make them somewhat ideal for sealing out external pathogens as well as absorbing and removing unwanted particulates or devitalized tissues from wounds–all while delivering a dose of antibacterial ions to the affected area.” – Source Popular Science May 22nd, 2013
US NAVY Seals do this for gunshot wounds too.
Thanks for the response!
I think one helpful action would be to stop feeding antibiotics to livestock just to fatten them up (not as a treatment for a disease), exposing that many bacteria to so much antibiotics has got to give the little buggers a leg up in the evolutionary arms race.
@Hirudinea – Also need to get cattle ranchers to stop feeding heifers (female cows) chopped up heifer uterus. This also fattens livestock. However, since estrogen and progesterone is introduced to the livestock and human females ingest the livestock (i.e. steaks, hamburgers, etc.?), it appears that fibroid tumors are enhanced and grow much larger in the human female’s uterus. They cause all sorts of gynecological problems leading to embolization,fibroidectomy, or a hysterectomy. The last one ends child bearing. All so cattle ranchers can fatten up their heifers to make more money.
The easiest thing to do as an individual is to boycott factory farming.
The “biohackers” are a bunch of pathetic morons. There is no way anybody can make anything useful in their home lab without it becoming a real lab. These tools are available to real trained molecular biologists who use them and know how they work. Just some guy becoming fascinated by the possibilities isn’t enough. One needs proper training. Unlike engineering, biology is extremely non-deterministic and complex.
If they want to contribute, they should get a degree and do proper research instead of messing around with pathogens at home and creating god-knows what.
Very similar to somebody right out of business school who wants to form an internet startup without any actual tech knowledge.
And where would the world be if only formally trained individuals were allowed to experiment?
That’s hardly the point. Everybody should be allowed to experiment. But there are limits when releasing antibiotic resistance genes and pathogens are on the agenda. And also, my main point is that biohackers are totally overhyped and they are extremely unlikely to achieve anything that the guys in the lab haven’t been able to do.
Let me explain: Biohacking is not like rigging an arduino to a sensor and making a tweeting doorbell. That level in biology has been passed about 100-200 years ago, when you could discover antibiotics in your lab’s trash.
Today biology operates at a level far beyond chip manufacturing. So how many people are exactly making functional and useful ICs in their kitchen? Exactly.
The Crisper stuff the guy in the Nature tabloid article describes is a joke. That’s the level every biology student does in their introductory practical course. Whoever thinks they are onto something because they can replicate it at home should – at best – invest their time into studying and doing it properly or are – at the worst – Dunnig Kruger idiots. Molecular biology is not electronics!
Meh, you’re pretty out-of-the-loop it seems on biohacker/DIYbio terminology/ideology… it turns out that a hefty portion of the biohacker crowd /are/ professionals in the biosciences… the difference is that they continue to tinker and experiment outside of their primary professional position.
Also, you haven’t lurked on freenode IRCs #homecmos… even Jeri Ellsworth did a video on ICs at home… Ben Krasnow too.
I personally work on lab-on-a-chip devices in my spare time (the little I have)… I started as an undergrad in my Biotechnology B.S. program, and quickly began leaning on the online DIYbio community when my Professors’ limited attention spans fell off. Not all professors are geniuses that can think in cross-disciplinary terms… at least not when those kind of things aren’t their primary focus. I called myself a biohacker… because I was doing a lot more hacking than my fellow classmates. Now that I primarily do computer programming for my day-job, I still refer to myself as a biohacker because it isn’t my primary job. Even if I was professionally working in biotech… I’d still have my personal projects and would still identify as a biohacker.
I indeed am working on cutting-edge tech, and have found myself at the seat of many pro-grade equipments. I picked up a used SEM a few months ago and am working on getting it back up and running. I could go on for some time, but I need to get back to biohacking some more now.
I have (and continue to) work with many PhD’s, and I will say that they are (usually) very knowledgeable in the subject they studied, but they can have very limited knowledge of other subjects.
Genes (and genetic material in general) is of no use and will do no harm unless incorporated properly into a living organism. There is no release of pathogens occurring. E. Coli. is a common organism found in the colon of most humans, and is not considered pathogenic by the CDC. E. Coli. is commonly used for genetic modification studies, and I have worked with it myself (I am an EE). The other organisms mentioned in the HAD article are being studied under proper laboratory conditions.
The guy in the nature article is not interested in manipulating organisms to inhibit anti-biotic resistance. That was simply a whimsical title for the HAD author to draw attention to the possibility of a “bio-hacker” working on such a complex problem. He himself said he had not mastered the technique yet, and was interested in a group endeavor to modify yeast for casein production.
The cost and complexity of making changes to genetic material in a controlled and predictable way using the CRISPR technique are beyond the means of an individual, but often the individual can talk to the nearest university and may get some help with the project.
No-one expects a “bio-hacker” to make any contribution to science. But it is always interesting when the layman can at least re-create the work of a scientist while using no expensive laboratory equipment.
The point of the nature article was to demonstrate the ability of the layman to grasp new technologies, not to show how this particular person would change or contribute to the biomedical field.
Everyone starts somewhere in their quest for knowledge. As he gains knowledge, his home lab can grow into a real lab as he buys equipment. He doesn’t need to change the world. He is just trying to learn new things, because that is one of the many things that make life interesting.
Your comment assumes all things from all previous levels of complexity and technical skill have been exploited and exhausted. The simple rediscovery of previously forgotten and useful medical knowledge from times before proves that this is assertion is as false as stating that because I will never have the photolithographic technology to manufacture my own MCU or CPU that I can never create something novel or useful with CPUs or MCUs the I have access to.
This assertion isn’t just elitist it’s pretty ignorant.
In his later years, Alexander Fleming was shown a modern biology laboratory with the latest equipment, and sterile conditions.
The guide asked him, “Now just what couldn’t you discover if you had a laboratory like this?”
“Not penicillin.”, he replied. (The penicillium mold probablyblew into his lab through an open window.)
We are developing this technology for more than 17 years, focusing on staph. infection. To Whom It May Concern: http://www.immlab.com