True graphene is a one-atom thick layer of carbon. It’s incredibly conductive, transparent, and of course thin. It’s one of those materials that, if it were only cheaper, would be used in everything from batteries to water filtration. Researchers from CSIRO in Australia have found a novel, dirt-cheap, and simple way to make graphene, and it’s hacker-friendly, for certain values of hacker.
The method is to take a sheet of polycrystalline nickel foil, spread a thin layer of soybean oil on it, and heat it up to 800° C for three minutes. It’s cooled off, slid off the foil, and it’s done. While 800° is a lot hotter than a standard toaster oven, their setup isn’t really all that much different. Notably lacking are things like esoteric gasses, partial vacuums, and the like. The nickel foil has some kind of catalytic role in the process — you should read the original if you’re more of a chemist than we are.
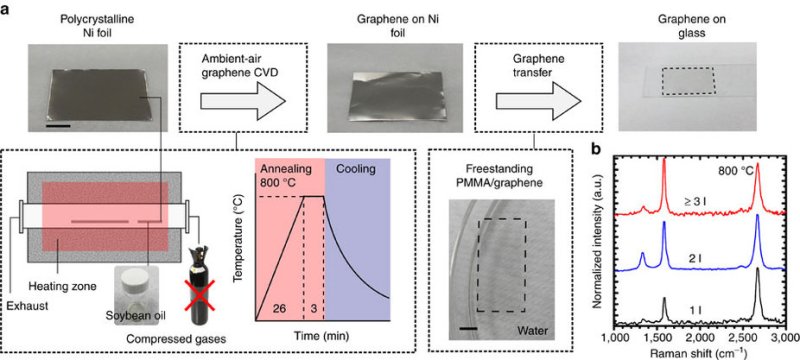 The resulting graphene isn’t perfect. According to their spectroscopic peeking, they are getting sheets with one, two, or three atomic layers. Lower temperatures result in sheets that aren’t completely fused, while high temperatures result in even thicker sheets. They also report a sweet spot in the cooling rate. In short, you’ll have to experiment around. But if you don’t have soybean oil on hand, butter will do the trick as well.
The resulting graphene isn’t perfect. According to their spectroscopic peeking, they are getting sheets with one, two, or three atomic layers. Lower temperatures result in sheets that aren’t completely fused, while high temperatures result in even thicker sheets. They also report a sweet spot in the cooling rate. In short, you’ll have to experiment around. But if you don’t have soybean oil on hand, butter will do the trick as well.
We’ve already seen a simple way to make graphene with a DVD player, but the precursor is graphite oxide, which we don’t have in our kitchen. (Although we do have a spare DVD burner…) If you’ve already made some graphene, and you’re wondering what to do with it, try Silly Putty.
Thanks [RandyKC] for the tip!
















Right on! One of the few things to come out of graphene research recently that isn’t pure hype. mtixtl.com sells polished polycrystalline nickel substrates as well. An anoxic reducing environment in an commercial or electric furnace would probably be sufficient for this synthesis.
Maybe. Making graphene is one thing, making quantities of it in usable forms is another as is doing so cost-effectively. While this looks like a step forward in those areas, they have a long way to go before this can be scaled to commercial production, and many places where things could go wrong (and likely will) along the way.
Yah I read you, sheet resistance was 324 ohms per square cm which is disappointing. Not many applications with that figure. Just excited to see the facile approach.
What about 324 ohms resistors, is there a market for such things?
this process is quite suitable for scaling up, i don’t know if the quality is quite there yet though.
one could use a a pure throughput type plant, with what looks like a total process time of just around 1-2 hours it wouldn’t even have to be that long or complicated, most of the machines to do it already exists.
from the graph the critical heating and annealing phase is around an hour, perhaps a bit more on the cooling side, the rest consists of very quick processes.
then scale would be a matter of parallelization and width of the feedstock.
A first glance it looks like it might scale, however I’ve seen too many cases over the years that I was in industry where great initial promise doesn’t come to fruition because of problems that manifest late in development. It is never all that clear to anyone, at the stage this project is at, if cost will be low enough and yields high enough to make this a viable process.
all true, fabrication isn’t trivial and i imagine the base technology isn’t either, since it seems both butter and olive oil works my first avenue would be a bespoke liquid for the coating, that might take out a bit of variability and might even allow for some novel materials with doping.
point is, there is surely a lot to learn just about this specific process, but at least it has scalable base mechanics going for it, not having that is what kills a lot of otherwise fancy tech, even if it doest outright kill it it slows down adoption, electric car batteries is a prime example of this.
Look I’m not saying this is worthless research, I’m sure many things will be learned. My issue is with the breathless assertions (mostly in the popular media) that this is it: the problem has been solved, and next year you will be able to buy it by the linear metre at the local building supply big box hype, that attends these sorts of announcements. These guys have a very long way to go.
never said you did :)
i agree on the subject of hyperbole as well, odd that it is so effective, one would have thought we would wise up to it over time but it seems the opposite might be true.
Since you seem to be the resident supercapacitor expert… could the graphene simply be left on the nickel foil and stacked and rolled with another graphene sheet to make a supercap?
No planar graphene is worthless for supercapacitors.
The purpose of using graphene in a supercapacitor is to have an extremely thin electrical conductor. Leaving it on the nickel would defeat the purpose.
mtixtl also sells the $50k furnaces that professional labs use for making really good mono graphene.
please don’t act like you’ve come up with something new. this entire process was optimized > a decade ago.
” But if you don’t have soybean oil on hand, butter will do the trick as well.”
If I strap it to the back of a cat, I’ll have a perpetual Graphene generator.
Careful. Doing that you end up with catalytic graphene, which has sharp points and a temperamental disposition.
[darren] wins One Internet.
(do not be alarmed when a small black box with a flashing LED on top arrives at your door)
How about using high frequency currents on the nickel foil, maybe pid regulated with radio feedback on the impedance of the oil to generate the graphene in a one atom thick 800 degree wave. The problem isn’t generating graphene, the problem is that in order to make a sheet as big as a cellphone with usable conductivity is basically harder than winning at lottery.
I really realty don’t get why it’s still so hard, I mean so much more complex things were refined so much quicker, this is a simple thing and should have been solved a few weeks after discovery.
I guess people aren’t as smart as they used to be.
why don’t you make some high-quality continuous monolayer graphene yourself if you’re such a genius at cvd?
“much more complex things were refined so much quicker” it took decades to integrate transistors into silicon wafers cheaply.
https://www.youtube.com/watch?v=LozJSTjrvek
Nah, if you use old ladies as the carbon source you end up with grannyene which is the opposite of a shape memory alloy as it is always forgetting.
Just ran across this technique http://www.seeker.com/a-lucky-lab-accident-results-in-bucketloads-of-graphene-2230364207.html looks even more promising
There are hundreds of promising ways, and yet nothing is industrial-scale.
all that gives you is carbon flakes and dust, moron. might as well just buy some graphite.
I’m skeptical about “Nano” materials in terms of health hazards. How do you prevent them from entering our biosphere? How do you responsibly dispose of them (provided you can actually reclaim them in the first place)? I’m no Far-Left Science Truth Luddite, but I’d like to see more studies about how these materials affect us long-term before they are deployed on a massive scale. Case in-point, the micro/nano plastic particles already used in consumer products like shampoo. These particles have already been detected seen in disturbing quantities in sea water samples, which seem to make their way into the Human food-chain.
I’m worried about nano-particles of plastic. Stuff gets everywhere food-chain wise.
They’re in the process of being banned where I live
The rule of thumb is that if you evolved with it in your environment it probably isn’t that dangerous, or it is obviously dangerous, but if it is novel in it’s behaviour or structure then you can’t predict how it will interact with natural biochemical pathways.
For graphene, it seems that the C-C bonds are easy enough to break, just by the difficulty of making it. And, for the most part, biology knows how to attack those C-C bonds. C60 buckyballs have shown that biology isn’t always capable of it though, but graphene has an edge carbon to attack first, tubes and balls do not.
As for those plastic “sand” beads that were in body wash and everything else, I have no idea what people were thinking. I mean, I like powdered luffa in soap, and Lava soap really does wonders for grease, but who thought “hey, this man made stuff that doesn’t biodegrade, let’s grind it to powder and replace those natural abrasives. Cheaper, and we can claim to be recycling!”
I already thought, if this micro plastic could not be replaced with ground up wood, like fine sawdust, for similar mechanical (abrasive) properties, but sure biodegradability. Or some selected mineral stuff.
A large belt of nickel foil in a loop could allow for continuous production of rolls of graphene on a huge scale, however down at that level reality isn’t as solid as we like to think so you can’t eliminate the noise in the process totally, which leads to defects. Still I would not be surprised if within a few decades that we can have affordable wall sized 300 ppi screens that are a few mm thick and use ambient light for much of their power.
Would single layer graphene be strong enough that you could peel it off the foil and roll it on a drum?
Graphene is pure carbon so eventually someone will invent a way to isolate and herd the atoms into the hexagonal shape. We already know that graphene can be self healing when there are gaps in the lattice and individual atoms are brought into contact near the gaps so the formation and growth of the crystal shouldn’t be that complex with the availability of free carbon atoms. This method of heating oil is interesting but appears to produce low quality graphene and graphite. Maybe they should consider using a magnetic field to produce more uniform sheets of monolayer graphene.
Are there any Ovshinsky amorphous silicon photovoltaic cell production machines going unused? I bet one of those could be repurposed for continuous process graphene production.
Has there been any more news on the ‘Tang-Lau method’ bottom-up synthesis method? This method sounded very approachable – basically cooking sugar water under pressure to produce a GO film, which can then be heated to reduce to graphene. But I haven’t seen anyone claim success reproducing it since the Robert Murray-Smith video in which he gets a film of “something” floating atop caramel, and just kind of assumes it. Has anyone confirmed this to work since?
no, because it’s a bullshit technique.
I haven’t seen anything since that original 2012 study. It’s been cited a lot but nothing concrete really.