Autoimmune diseases occur when the immune system starts attacking the body’s own cells. They can cause a wide range of deleterious symptoms that greatly reduce a patient’s quality of life. Treatments often involve globally suppressing the immune system, which can lead to a host of undesirable side effects.
However, researchers at the University of Chicago might have found a workaround by tapping into the body’s own control mechanisms. It may be possible to hack the immune system and change its targeting without disabling it entirely. The new technique of creating “inverse vaccines” could revolutionize the treatment of autoimmune conditions.
Identification: Friend or Foe
The body’s immune response is normally a good thing, defending us from various pathogens. Indeed, it’s crucial to our survival; individuals with suppressed immune systems must take all kinds of precautions to avoid harm on a regular basis. In autoimmune diseases, though, the body’s immune response gets misdirected, attacking the body’s own cells. Suppressing the entire immune system can solve the autoimmune problem, but leaves the body with precious little defence against actual pathogens.
A new method of molecular hacking may be key to solving this problem, according to a new paper published in Nature. The idea is simple: it aims to reprogram the immune system, instructing it ignore certain targets that it shouldn’t be attacking. It’s basically the opposite of how a vaccine works, hence the name “inverse vaccine.” They’re intended to advise the immune system on what not to kill.
The immune system determines what to attack by focusing on antigens. These are molecular markers, like proteins or strings of amino acids, that the immune system can recognize. For example, the COVID-19 coronavirus features prominent spike proteins. When presented with these spikes, our immune system typically identifies them as foreign and unfriendly, and scrambles to attack them. This happens more quickly once we’ve had a vaccine or previous exposure to the virus, as our body remembers how to recognise those antigens. The problem is when our immune system gets confused, and starts identifying our own cells as targets for attack. In many autoimmune diseases, it’s unclear why the body begins to attack itself, but research continues into theories around genetic predispositions, environmental factors, and responses to viral and bacterial infections.
To try and reprogram the immune system, scientists developed a polymer that was glycosylated with N-acetylgalactosamine, known as pGal for short. Attached to this polymer is the antigen causing the immune response, connected via a “self-immolative linker.” Once the polymer is ingested by an immune system cell in a process called endocytosis, this linker breaks away, releasing the antigen inside the cell’s regulatory environment. Once this happens, the immune system ceases to treat the antigen as a threat.
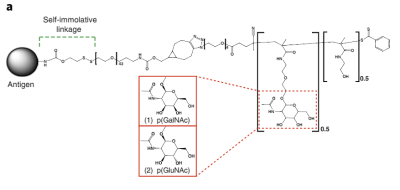
The idea for this method came from the body itself. Normally, when a cell dies naturally, the immune system does not overreact. This is because the liver uses pGal as a tag for molecules from dying cells, which tells the immune system to ignore these molecules. The researchers simply leveraged this existing mechanism as a way to get the immune system to ignore other targets as desired.
Early testing showed that the technique could be used to prevent Type 1 diabetes from occurring in a mouse model by preventing the autoimmune response to insulin-producing cells. Later work showed that the technique could also potentially be used to treat autoimmune diseases that had already occurred. This was achieved by demonstrating that an immune system could be programmed to stop attacking the myelin coating on nerves in animals suffering a multiple sclerosis-like disease. The nerves regained function in time, and the animal’s symptoms were reversed.
Lead author of the study, Jeffrey Hubbell, is buoyant about the possibilities. “In the past, we showed that we could use this approach to prevent autoimmunity,” said Hubbell, adding “But what is so exciting about this work is that we have shown that we can treat diseases like multiple sclerosis after there is already ongoing inflammation, which is more useful in a real-world context.”
Phase I safety trials of an antigen therapy based on this work have taken place with patients suffering from celiac disease, along with a further Phase I safety trial for treating multiple sclerosis. As of yet, no “inverse vaccines” are yet approved for clinical use, but there is strong potential for this technique to improve our ability to treat and manage autoimmune conditions.
















What if someone develops a virus that produces an inverse vaccine for itself?
In fact, why haven’t viruses already evolved to do exactly that?
Evolution works by random chance, and maybe it’s just an extremely low chance that a virus sticking antigens onto a massive polygalactoseamine has ever happened. Viruses have as little DNA/RNA as possible, far less than needed to successfully replicate, so they work by hijacking the cellular machinery to replicate themselves. The mechanism shown here is orthogonal to replication. There’s no evolutionary forcing mechanism to amplify something that’s just close to it, and there’s significant cost to carrying around the genetic material required to do something like this, so evolution is going to generally select for simpler viruses.
Unless it’s a virus that happens to target liver cells, and hijacks the mechanism to subvert the immune system.
Either way, there’s two options: either the trick doesn’t work very well and there are countermeasures for it, or we are at the verge of creating a killer virus by accident or intent.
I was thinking exactly this. However, I am holding off on my doomsday attitude until I understand this whole process better.
They do. AIDs does it.
No it does not. AIDS infects immune cells (said differently, it breaks the tools used to break the illness), it doesn’t reprogram them to ignore its own marker.
HIV does take some of the immune cells protein as part of its shell. This is one of the reasons it’s so hard to fight off and why the immune cells can’t attack it: they see HIV as one of them.
But that’s not what the article is about. HIV doesn’t reprogram the immune system, it attacks it, or hides from it, we already have a host of drugs that attack the immune system called immunosupressants, and viruses cloaking themselves from immune response by using cellular proteins is far from unique to HIV.
This is completely unrelated to what you’re talking about
AIDS is complication of HIV. AIDS do nothing – it is a condition.
I think “does nothing” is dangerously close to the denial seen in the 80s, tbh. Many people died from AIDS. Yes, along with something else mostly, but some did die in hospital from AIDS itself. 40 million dead.
It’s also dangerously close to the covid denial going around, arguing that everyone does from “complications arising from covid” rather than covid itself.
Both are stupid – millions are dead who otherwise wouldn’t be.
Because of evolution rule number ONE of viruses, You will never go that mad that you will completly disable or kill your host within a short timespam, otherwise you will be also self eliminating yourself, without a host.
That’s just by elimination. A virus like that can evolve, then proceed to kill its hosts, and disappear in the lack of new hosts to infect.
With a high enough r value, it could be plenty deadly and not worry itself (if a virus could worry!)
This has got nothing to do with viruses, Inverse vaccines are a completely new and novel type of drug for treating autoimmune diseases, which is are medical conditions where a persons own immune system attacks a class of cells in their body. Examples of autoimmune diseases are asthma, multiple sclerosis, cystic fibrosis, rheumatoid arthritis, lupus, inflammatory bowel, just to name a few particularly nasty and common ones that could be treated with this approach.
Don’t be confused by the name, this new class of therapeutic drugs could be live changing for hundreds of millions of people worldwide who live with currently incurable medical issues worldwide.
As for viruses, people don’t produce them, nature does. The best we can do is cut and paste bits of existing viruses together, and when people do that there are obvious telltale signs, analogous to the stitch marks and skin tone differences of people who have had limbs grafted on. There’s never been a reported case of a human made virus in the wild.
I guarantee you we’d never hear the end of it if there was though – people got hysterical about the slightest hint that Covid might have come from a lab, they’d be screaming it from the mountaintops if there was irrefutable evidence of a man made virus outbreak.
If a virus somehow came into existence that produced an inverse vaccine, presumably it would spread from one animal to the next, curing them of whatever autoimmune condition the inverse vaccine was for. That sounds like a good thing and a possible way to eradicate auto immune diseases.
All true, except that covid actually does now seem to have escaped that lab in Wuhan. Of course, they didn’t build it from scratch, they took an existing one and boosted certain things, so your overall point remains true.
dangerous levels of hubris on display here
I think you’re completely missing the virus related risk here. The pGal tags don’t cure an autoimmune condition, they tell the immune system to ignore some protein. If the tags are synthetically attached to whatever cell type the body has previously been attacking then that seems to cure an autoimmune condition. The speculation is that if these tags were attached to a virus, they’d cause the immune system to ignore the virus.
As a Type I diabetic, this looks REALLY interesting.
I’ve seen some studies that indicate that IF the beta cells can be sufficiently protected from the immune response that is killing them, they may regrow. IIRC there was one company working on a novel immunosuppressant + growth stimulator to do this.
The problem is that immunosuppressants that can protect the pancreatic beta cells have so many negative effects that it’s generally better to stay with the stressful regimen of CGM + daily injections or pump, etc. This, however, is an extremely narrow-target immunosuppressant that might be absolutely key for such a treatment.
Yeah, also I believe they’ve already been able to transplant pancreatic beta cells, it’s just not worth it because you have to go on immunosuppressants or they just die off eventually too.
Vertex have also successfully made artificial beta cells that appear to work, they just again need to suppress the immune system.
This inverse vaccine really could be the missing piece of the puzzle for a lot of things.
Well, now that the djinn is out of the bottle, an inverse vaccine for something as common as a simple influenza virus could be devastating. Of course, I have no idea how to deliver that to significant numbers of people.
Put a foil lined red hat on probably a good place to start
Stop mocking caution.
Relax. All he’s saying is that paranoid mall Santas are somehow more vulnerable to weaponized anti-vaccines
At 1:11 am… and these are the thoughts in your head?
It would probably be a easier to create a traditional vaccine that triggers auto-immune disease, yet even that is unnecessarily over-complicated.
The simple fact is these things are really hard and expensive to create. It would be far easier and reliable to just create a traditional virus bio weapon, than go through such a long, convoluted and expensive process. Bad actors want simple and reliable and there’s already a wealth of those routes available.
This has nothing to do with viruses, it’s about a new class of drugs for treating autoimmune diseases. You need to read the article and not just the title.
Or maybe they should do some real, serious science on the vaccines that are in use… Many autoimmune conditions are linked to or suspected of being caused by vaccines.
Re: No one of cons. – Autoimmune link
This gives a totally skewed impression of the facts.
Getting a viral infection causes immediate damage (scarring) and may have possible long term effects (think Shingles). Yes, even the Flu, not just Polio, etc.
This falls along the risk evaluation by people who buy lottery tickets. Subconsciously, it goes like this: “Well, I’ll either win, or lose. So… Chances are 50/50!”
Problems caused by vaccines are nowhere near to what not having them would be. You might be smart enough to know the orders of magnitude difference in these two choices (I hope). Some readers of your comment may not. This is a disservice to them and society.
Well, actually check out three years of data on the difference in death rate between those inoculated and not.
How DARE they potentially cure a load of serious long-term debilitating conditions, amirite?
This statement presumes that a) such research is not being done, and b) it’s an either-or situation. A quick search on Google reveals a number of research papers on the former, refuting the former. And the fact that there are many thousands of medical topics being researched at any given time refutes the latter.
Really? Please point to some credible peer-reviewed articles by people with relevant qualifications that says that any autoimmune condition is connected to immunisation. The vaccines that are in use are the product of “real, serious science” – the type of science done by actual scientists with actual qualifications from actual universities, not pseudoscientific nonsense spouted by crackpots with qualifications from schools of naturopathy and homeopathy to promote snake oil treatments or conspiratorial books and videos.
Not only do you know nothing but the paranoid bullshit you’re spreading about vaccines, you didn’t even read the article.
A few things missing from your post:
– “Suspected” by whom?
– Based on what evidence?
– How could something like that get past all the testing and monitoring?
– Links to high quality peer-reviewed studies. We don’t need any Wakefield-style carefully selected, cherry picked and faked anecdotal evidence that e.g. ‘vaccines cause autism’. Incidentally, new research can detect autistic traits at a few months of age, long before vaccines are given; to be a cause, vaccines must travel backwards in time. Now that’s impressive…
+5, informative
Only quacks think that taking a vaccine with a 1 in 100,000 risk of a single adverse effect is worse than getting the disease that causes that effect worse in 1 in 10,000 people.
The “vaccines are bad” crowd simply ignore all the lives saved. Then of course it’s worse! “If 1 in 30 million die from the vax, it should be banned!!!” sounds fine until you look at the fact that the vax *saved huge numbers of people from death* and, often, lessened the impact if they did later get it.
This is actually a pretty novel treatment modality and passes the sniff test from me. It that it means much.
Good write up and good intro to basics of immune disease.
I’ll underline that the immune system is thought of as defense against external pathogens but it is a lot less appreciated that it is heavily involved in apoptosis (as mentioned) and general own-self cell turnover.
When immune cells go awry you get lymphoma and leukemias. To a lot of the public this is probably surprising that the immune system has such a strong link to cancer.
The money on this one is in developing a variant that blocks/repairs arthritis, one of the most common and debilitating auto-immune diseases. a Medicare approved cure for arthritis would be bigger than Viagra.
Yeah that would be huge. Hope it works out
Not if it cures it. Only if it treats it.
Don’t worry, there’s still altruists outside the US “for profit” medical system.
How many millions will it cost?
And will any health insurance company cover it?
As someone that suffers from MS, and is on
Ocrevus, which costs $200,000+ a year for treatments, it’s already a yearly fight to get the health insurance company to cover it…
They can’t deny your coverage, but they can just raise your premiums (I pay $2000 a month for healthcare).
The expense to treat your current condition might be the thing that actually makes it worthwhile to insurance companies to cover, and possibly why they seem to be targeting MS with it now.
It seems crass to reduce your condition with very hard real world struggles to an economic equation, but if you currently cost them $200k a year, and they expect you to live more than 5 years, then a cool million might not be a bad investment for them I guess?
I think Shonky is right here. The Hepatitus C treatment that was developed costs on the order of $200k, but it must save more than that as a lot of insurance companies are covering it now. It’s completely changed the life of a friend. Hope we find an effective therapy for your condition soon!
As someone that worked directly on my thesis for HCV antivirals and was training at a hospital that was participating in the next-gen antivirals this comment is correct.
The crackpot argument that treating or curing a disease somehow makes money for drug companies or healthcare or anything else is bananas. Not treating them *costs* money long term. HCV in particular was a huge cause of liver transplant which is both super expensive short and long term and even if not transplanted chronic liver disease till death is expensive too.
The real issue isn’t actually economics it is disparity, access, distribution, compliance etc AKA public health/epidemiology.
Yeah of course there is money in curing diseases. But if you have experience in pysch and the perpetual subscriptions of medications that fill millions of peoples medicine cabinets, you may have a different opinion.
I dropped out of medicine because of this and many other reasons.
Access is key, I want to start a home health service to address this.
The counter to the idea that drug companies don’t want to cure diseases, only treat them, is that the equally powerful and greedy insurance industry wants to see cures so they don’t have to keep shelling out money to drug companies forever, this makes one-shot treatments that cure diseases basically just as valuable as ones that simply treat them.
If company A makes a treatment for asthma that will generate $100k in profit over a lifetime, and company B comes along with a cure that costs $110k, the cure is the one that makes the most economic sense, since the insured person may not follow the treatment regimen in the long run or may develop related conditions, whereas a cure will reduce that persons risk in that area to the same as a normal person by definition.
Also, a cure is likely to be more profitable than a treatment, because you only need to produce one dose per patient, and all those profits come at once rather than spread out over the lifetime of a patient and at constant risk of being superseded by a better treatment. CEO’s don’t prefer setting up revenue streams for future CEOs they want those profits to show during their own tenure.
It seems intuitively like you would make more money on treatments than cures, but the reality is that the drug companies set the pricing of cures to reflect the value of them, and treatments become dramatically less valuable in the face of cures.
Yah, great. Now a version of this can be developed to allow all those dumbass anti-vaxers to undo their vaccine. Enjoy your ventilator!
Well, actually check out three years of data on the difference in death rate between those inoculated and not.
For 65 and up? Yeah pretty good. For everyone else? Basically no change. And the studies on transmission show basically no effect. So it presents more of the qualities of a targeted therapy rather than a population inoculation.
Systemics have been out of favor for ~15 years. Biologics are used far more frequently depending on the disorder…. I have taken biologics for years and they work well without tanking your immune system (I was on cyclosporine and methotrexate for a while, full immunosuppressants, those were nasty with terrible side effects).
The downside of biologics is they are very temperature sensitive, and hard to manufacture. I am very thankful I have such great health insurance because my $30 copay is far less than the $1500 a shot which is required once a month. That is a cheaper biologic too.
I will be watching this one with interest
I’m simply untreated because the side effects are too bad with the old stuff and I can’t really get access to the new stuff. This sucks. So I’m quite interested.
A lot of biologics do have income based payment deals with the manufacturers, eg you make this much you pay this amount. So there can be considerable savings off the retail price. Something to consider/look into.
But yeah don’t bother with the systemics, my hair was falling out, I was jaundiced and felt like death all the time….and they didn’t do what they were supposed to either.
Exactly, I said this in my own comment, the situation isn’t as bad as the article makes it out to be, there are other options than general immune suppressants and biologics have been used for decades and are only getting more popular and more numerous. Most of them only target specific chemicals, or hormones, or whatever else so are a targeted approach and don’t come with all the side effects of systemic steroids or immune suppressants. Biologics do have side effects too but most people will never notice them and they do have potential long terms issues like some kinds of cancer but they are rare and it varies from drug to drug. Biologics are the best option for a lot of people the only issue is cost, but the cost has also come down a lot, especially when biologics patents expire.
I’m just glad I’m in the UK, they have deals with the drug companies and the cost is covered by the NHS. The cost difference with some drugs between the USA and UK is huge but you do get some manufacturers that help out lower income people in the USA.
Could this treat arthritis?
Rheumatoid arthritis? Yes, because it is a chronic (long-lasting) autoimmune disease that mostly affects joints.
If this really works it could be a game changer for organ transplantation!
What does a pig need with a human heart. ;-)
WOW!!!
Why does that feel like a bioweapon.
Hmm, so you read an article about a therapy for autoimmune diseases and think it sounds like a bioweapon…
I don’t know, it could be because you’re a paranoid tin-foil hat wearing lunatic with poor reading comprehension who also thinks that vaccines kill people rather than being responsible for saving more lives than any other invention in human history.
I can’t rule out other possibilities like mental illness or trolling though.
Not a smart choice of words, vaccine is from vacca, cow, and is a reference to the early cowpox virus being used as an active inoculant, and that has nothing to do with this technology.
Try *GAT* for short.
A simple but accurate word or phrase to refer to “Synthetically glycosylated antigens for the antigen-specific suppression of established immune responses” is *Glycosylated Antigen Therapy.*
This term is accurate because it describes the key features of this approach:
* The antigens are synthetically glycosylated, meaning that they have been modified with artificial sugar molecules.
* The goal of the therapy is to suppress established immune responses, which means that it is designed to treat autoimmune diseases and other conditions where the body’s immune system is attacking its own tissues.
The term glycosylated antigen therapy is also simple and easy to understand, which makes it a good choice for communicating with the general public.
So giving smallpox to a cow would be a reverse vaccine?
No, you gormless plonker. Giving a Glycosylated Antigen Therapy to a cow would be a reverse vaccine. Giving smallpox to a cow would be, uh… Giving smallpox to a cow, and nothing to do with the article or the comment above.
I can’t understand how someone who clearly can’t read is able to write a reply, did you get your caregiver at the group home to do it for you?
Even a German would get that that was a joke..
Perhaps it is YOU who needs to be checked out?
RV: “These aren’t the proteins you’re looking for.”
TC: “These aren’t the proteins we’re looking for.”
…
RV: “Move along.”
TC: “Move along, move along.”
Reverse vaccine? So an empty syringe pulls fluid out of your arm, and supposedly makes you ill?
Antivaxxers should be thrilled. Or divided. Or inverted.
This isn’t reddit, you’re not going to get dopamine points for clever non-sequiturs.
This isn’t reddit, you’re not going to get dopamine points for clever non-sequiturs.
But facts DO matter. Check out the Covid death rates of those inoculated and not. How about the eradication of several diseases?
Some people just always have a perception “There must be more to this! I’m so smart and thorough, I bet I can find some aspect nobody has even thought about …”
Vaccines aren’t perfect. Neither is washing hands, masks, …
Do you care if your surgeon follows hygienic procedures? A fact you don’t like, is still a fact.
Vaccines are a man-made blessing. You have no right to endanger the gullible and ill-informed amongst us by telling them they shouldn’t protect themselves and others.
You’re responding to the wrong post, bot.
Hehehe … lame … no defense?
There IS a difference between a tried vaccine and an experimental type of which there is a real economic and political need to hide/misrepresent the effectiveness and side-effects.
Also, stop touching your face with your hands, have you learned nothing?
About f**cking time. I’ve been wondering when technology would be developed to erase immune responses. Some diseases like AIDS can do it, so we already knew it was possible. This will be a game changer if they can commercialize is. That’s really exciting!
The only people I have seen suspect vaccines as being the cause of autoimmune diseases are either quacks who have had their license revoked for malpractice, or those with no medical training or background and haave patreon accounts to make money off antivax loons. Sure its *possible* a vaccine can slightly increase the risk of such an event, but given just how many vaccines are in use, and the time they have been used – we would have seen an indication of any significant issues. You comment implies that there are lots of people who think there is a link – and perhaps even implies they are professionals in the field, when neither is really true.
Exactly, I have seen no evidence that vaccines cause autoimmune conditions either. They still don’t really know what exactly causes autoimmune conditions but it is well known that infections and other things can trigger them, I have never seen any evidence that vaccines themselves trigger autoimmune conditions though.
There have been instances, but they are thankfully very rare. Pandemrix, a vaccine for pandemic 2009 H1N1 flu, was associated with a number of cases of narcolepsy in Europe, apparently via an autoimmune mechanism. It was not used in the US.
That said, the benefits far outweigh the risks with most vaccines. When they no longer do (because the disease became so rare that the potential benefit shrunk away), they are no longer used, or replaced with safer, but often less effective vaccines. This was the case with the Salk attenuated live virus vacccine for polio, long replaced in developed countries by a dead virus vaccine.
That’s a very good example. Safer but slightly less effective meant the risk of a really bad result was lessened, without much negative effect elsewhere.
Vaccine deniers *always* ignore the lives saved by the vaccine when pointing out that “the vax killed 3 people!” – ok, but that’s from 10 million doses, and the death rate otherwise was 1 in 50. That’s 200,000 – 3 lives saved.
A large number of very smart and highly respected medical scientists suspected vaccines might be linked to certain autoimmune conditions.
That’s why they did giant systematic studies to check. For the most part, they don’t seem to related.
There’s been found some weak correlation with chronic fatigue conditions between certain virus infections circulating with high immune system activity including vaccinations, but it’s a long way from understood yet. Looks like having a severe cold seems similar risk.
You leave out a very important other treatment for autoimmune conditions, there are more options than just suppressing the whole immune system. Biologics are an increasingly popular option yet have been around for a while, they target specific things rather than affecting the whole immune system and they have very effective results in a lot of people.
It is always good to get more treatments and possible cures (of which there are none just now) but the situation isn’t quite as bad as the article implies. Yes you do still have to be careful on biologics to avoid people who are sick and to get extra vaccines but it isn’t as bad as taking general immune system suppressants.