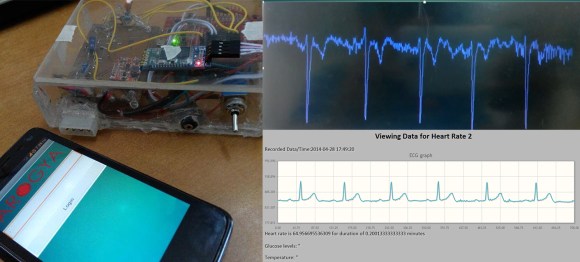
The title of [Nuclearrambo’s] post says it all, “Android based wireless ECG monitoring (Temperature sensor and glucometer included).” Wow! What a project!
The project is built around the HC-06 bluetooth module and the Stellaris LaunchPad from TI, an inexpensive ARM developer kit. Building an ECG is a great way to learn about instrumentation amplifiers, a type of differential amplifier used for its extremely high common mode rejection ratio (CMRR). Please be sure to keep in mind that there are a myriad of safety issues and regulation concerns for medical device, and there is no doubt that an ECG is considered a medical device. Sadly, [Nuclearrambo’s] post does not include all of the code and design files you need to build the system, which is understandable considering this is a medical device. That being said, he provides a lot of information about building high-quality ECG instrumentation and the web interface.
It would be great if [Nuclearrambo] could post the Android application code and Stellaris LaunchPad code. Even with these omissions, this post is still worth reading. Designing medical devices requires a lot of know-how, but who knows, maybe your next project can save your life!















“Medical device” my ass. So it interfaces with a human body. Big deal in that, just two pieces of hardware connected together. Safety – just feed it from batteries (or from battery-operating laptop) and take care on the design level that no high voltage can pass to the more sensitive piece of hardware (said human body) and you’re on the go. Regulations? These are for corporate drones that don’t live on the tech frontier.
We’re the hackers, the makers, the elite of the world – not some wussies that need a disclaimer and three overly cautious lawyers in every project, and incomplete data “to protect us from us”. We know what we are doing. (Okay, not always, but we are usually at least aware of that possibility.)
We don’t need no stinkin’ regulations. Damn the torpedoes. Full-steam ahead!
That said, the project is fascinating! I for one hope it will go opensource and will be built upon. Possibly to go on kickstarter, be affordable even as “just a toy”, and be usable as standalone or as a component of a bigger biomonitoring setup.
The “elite of the world” ? puhleeeze.
Get over yourself ! Sounds like my brother (former F-18 Hornet driver and TopGun graduate). Jerk would think the entire world revolved around him because of his “elite” status among the common peasants.
Maybe you don’t have two nickels to rub together so nothing to sue you for.
The rest of us might have actual tangible financial assets that we don’t care to encumber fighting off litigation from some dimwit that electrocutes themself using a design we publish.
Yes, elite of the world and I mean it. Without us, the others would still be hanging on the trees and bickering about bananas. Somebody had to invent even the first sharpened rock, and rightfully scoff at the comments of the fellow monkeys about how dangerous it could be and how somebody could get cut with the edge.
Overly cautious attitude chokes progress, which depends crucially both on the willingness to try and the willingness to publish. And then there would be no fun left in the world, just overpriced commercial solutions wrapped in NDAs and without documentation to speak of. While what the world needs is lots and lots of cheap and opensource biomed and lab instruments. The official healthcare prices are mushrooming and a real competition is needed sorely. Of course this competition will not have all the permits and FDA paperwork, as one of their purposes is to increase the barrier to entry so the status quo is not shaken. You may not be able to legally make and sell it – but a friend in Shenzhen (or elsewhere with smaller amount of lawyers per capita) can, e.g. on DealExtreme, if you want to go commercial. Many countries (Africa is full of examples) won’t worry about paperwork; and people in your one can import from there on their own, due to the beauty of Fedex and customs that can’t recognize a circuitboard from another circuitboard if they ever get to look (and if you don’t like to lie to them, call it not “ECG” but “multichannel data acquisition board”). We got the globalization dropped upon us, we can at least leverage some of it.
And if you worry so much, you can always publish anonymously on a server in a country with unfavorable political relations with your one (so the IP address request will not be honored), possibly via a proxyserver or a friend. International friends are there for a reason.
The society turned into a bunch of pussies unwilling to take even slight risks. (And then gladly taking much higher risks daily, just because they are so common nobody cares anymore about that class. Or because it is The Proper Way, e.g. not being to afford a licensed instrument vs being able to afford a cheaper but “not permitted” almost-as-good one; this one part is my sore spot.)
Fewer innovations will come out of the USA with this attitude. The rest of the world will happily claim this eco-niche.
He might have used MIT’s appinventor to write the app
Yup I did use MIT’s AppInventor. I would be happy to post the code or the app itself. Whats making me stop is that its not fully done yet. I will update my post with the code soon.
Lol sorry! I briefly skimmed the article just before going to work. Noticed you mentioned amarino and I stopped reading there
CYA, and get on with the future. Disclousere won’t happen. Instead you will read about the next discovery or dev here.
I don’t want to stir up a hornets nest of stuff on healthcare, but lawyers are number 1 on the jettison list.
More power to the elite (1337)!
It is indeed a medical device. There are varying levels of medical devices, from tongue depressors and surgical screws to drug pumps and diagnostic devices to pacemakers. Different levels of scrutiny apply to different levels. At the lowest level, all you need to do with the FDA is say, “This tongue depressor is just like others on the market except mine are made of bamboo.” (well, a little more than that, go read up on the 510K process).
At higher levels, if you are making medical diagnoses, or in direct contact with the patient, you start worrying about safety and efficacy, and needing clinical trials to sell commercially.
But none of that applies for a hobbyist device like this. No FDA stormtroopers are going to raid your house unless you’re selling it as a medical device. FDA has recently issued guidance on medical smartphone apps, and taking a very reasonable stance: Health maintenance doesn’t need regulation (e.g. fitness tracking), but diagnosis and treatment (melanoma detectors, diabetes insulin schedulers) most certainly do.
On a medical device like this, safety is a tiny concern (battery operated, minimal voltage across the skin), but does it have the accuracy and reliability for medical diagnosis? That’s why it’s regulated.
If you want the electrical noise to go down, get better insulated electrodes and add highpass and low pass filters, also median filters will help. also the impedance has to be low, you got a pretty good waveline there. Congrats!
Thanks for your suggestion, I will definitely look into it. Its been a while since I worked on this project.