When I started writing my recent article on COVID-19 testing, I assumed that I would be doing a compare and contrast sort of article. Like many people, I assumed that the “gold standard” test would be the reverse transcriptase-polymerase chain reaction (RT-PCR) test that I described in some detail. And indeed it is, but it’s not without its problems, such as the lack of certified labs and the need for trained technicians to run the samples. I also assumed there would be another test, a simple serological test that could use antibodies to discern if there was an active or even a previous, resolved infection.
At the time I wrote the first article, I could find no indication of an immunologic test for COVID-19 (more specifically, a test for SARS-Cov-2, the virus that causes COVID-19). But almost as rapidly as the number of COVID-19 cases rises, the news changes, and it appears that simple, rapidly performed antibody tests are now or soon will be available. They likely won’t replace the gold standard RT-PCR test, but they do stand to be a game-changer for the front line providers and the victims of this disease. So it pays to take a quick look at immunoassays for infectious diseases, and learn how they work.
Innate and Adaptive
To understand how immunoassays work, it’s necessary to know a little bit about how the human immune system recognizes invaders and targets them for destruction. It’s a vastly complex system, of course, seeing as it is tasked with differentiating between self and non-self cells based on only the slimmest of clues, and a full treatment would take far more time than is needed to get the idea. So yes, this is an extremely stripped-down tour of the immune system, but it should suffice for the task.
Since the main job of the immune system is to recognize and destroy invaders ranging from bacteria to viruses and even other human cells, it has to have something to key on that allows it to be both extremely selective and very aggressive. Proteins, with nearly infinite variability thanks to being composed of 21 different amino acids, make a perfect target, and doubly so since the exteriors of most invaders bristle with dozens of different proteins.
An immune response starts with the innate immune system, a rapid-acting set of first responder cells. The innate system is non-specific; beyond the ability to differentiate between self and non-self, its cells will pretty much eat anything. When it does encounter a bacterium or virus particle, it envelops it and breaks it down, a process known as phagocytosis. All those proteins that were studding the surface of the invader are then presented to another set of cells that form a second immune system, the adaptive immune system.
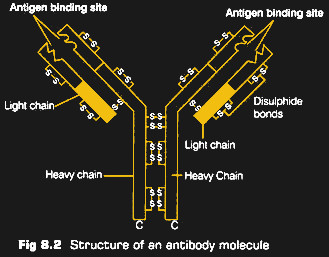
The cells of the adaptive system have their own proteins on the surface, formally referred to as immunoglobulins (Ig) but commonly known as antibodies. These proteins are always shown in diagrams as a Y shape, which is actually pretty accurate. The “arms” of the Y are highly diverse, with different antibodies having different sequences of amino acids. This produces a population of antibodies with varying affinities for other proteins, depending on the size, shape, and electrostatic charge of the amino acids.
When the innate immune cells present the bits and pieces of an invader’s proteins to the adaptive system, with any luck, one of the antibodies will randomly match and bind to the invader. This starts a cascade of events that cause the genes that encoded the matching antibody to be massively amplified in a process called clonal expansion. Cells of the immune system can take days to weeks to mount this defense, but once it starts, a huge number of cells go on patrol, armed with antibodies specific for the infection. Once the invaders are swept up, most of this army of cells is demobilized, but a few memory cells hang on to that specific antibody, ready to mount the offensive rapidly through the innate immune system if the same challenge arises again.
Going With the Flow
Monitoring the state of the immune system, therefore, can tell us not only if a patient is currently fighting an infection, but by digging into the institutional memory of the immune system, it can also tell us if someone has ever faced a specific challenge. All of this requires that we have a sample of the specific antibodies that have been produced by the body as it mounts its defense against a virus. Traditionally, that has been a slow and difficult process, where cells from animals challenged with the virus are painstakingly cultured and amplified through human cell lines, with the resulting antibodies purified before being made available for use in immunoassays.
Antibody purification usually takes months, but in the case of COVID-19, the first antibodies were isolated very quickly. An-Suei Yang et al at the Academia Sinica in Taiwan raised a monoclonal antibody (mAb) against the N, or nucleocapsid, protein of the virus. It took them just 19 days to isolate and purify the antibodies, using a process involving artificial intelligence models of the interactions of antibodies and antigens. Predictions from the model allowed them to create a library of DNA sequences coding for antibodies that should have specificity for the N protein, which can be produced and selected without having to run them through animals first. They managed to cut two months off the usual time to produce an antibody, and have gone on to create others that are so specific for SARS-CoV-2 that they will not bind to any other human coronavirus.
Putting the purified mABs to work in a usable assay is another challenge, and multiple companies are working around the clock to perfect their version of a test. Most of these tests, which are called point-of-care (POC) tests, use the lateral flow architecture. Pregnancy tests are a common example of a lateral flow immunoassay, and are very similar to the COVID-19 POC tests that are soon to hit the market.
Lateral flow tests work by taking a sample from a patient, probably blood for COVID-19 tests, and applying it to an absorbent matrix. The matrix is generally nitrocellulose, through which the blood will rapidly move thanks to capillary action. On its way through the matrix, the sample will cross a region filled with dried buffer reagents and a special mix of antibodies. These antibodies are not necessarily specific for SARS-CoV-2 proteins; they’ll stick to pretty much every protein in the sample. But they are conjugated to some sort of particle, like a colloidal gold microbead or a dyed latex particle. These will provide the visual signal later on in the test.
After crossing the conjugate region of the test, the proteins in the sample are now “decorated” with microparticles. The continue migrating up the strip until the cross lines containing dried mAbs specific for SARS-CoV-2 proteins. Depending on the test format, there could be two or three viral antibodies in these test lines, plus at least one control line that pretty much reacts with anything. Any of the decorated viral proteins in the sample will bind to the test lines, with the accumulation of conjugate producing a colored line that’s easily visible against the background of the matrix.
It’s important to note that there’s also a backdoor technique. Rather than raising up antibodies against SAR-CoV-2 proteins, cloned fragments of viral proteins can be used instead. In this case, the proteins would be applied to the test strips and would bind any conjugated-decorated antibodies in the patient sample. The main advantage here is that mAbs against the viral proteins are not needed, with the disadvantage being that different patients will express higher or lower amounts of antibodies depending on where they are in the viral infection cycle, as well as their general immune response.
Running and interpreting lateral flow immunoassays takes very little training, and the results are generally available within 20 minutes or so. Compared to the hours needed for a qPCR assay, not to mention the certified labs and the time needed to ship samples to them, the benefits of a point-of-care immunoassay are obvious.
Currently, there are at least nine lateral flow immunoassays for various SARS-CoV-2 proteins being developed. Some of them are shipping, some are in validation, and some are seeking approval from regulators. But all of them are based on the amazing complexity and adaptability of the immune system, the system that’s tasked with keeping humans alive in a world where challenges come is the smallest and most insidious packages imaginable.
















Very informative and enjoyable read. Thanks
All those references to PCR as the “gold standard” sound like a comparison of the two tests to me. But isn’t that sort of apples and oranges? The PCR test tells you where the virus is, who needs to be quarantined. The immunologic test tells you where it’s been and who might already have immunities. Both are valuable but in different ways.
Granted, there is some overlap among people who are infected and have been so long enough to develop antibodies but haven’t yet cleared the virus completely.
The PCR test is the gold standard because it detects the virus itself. The antibody test isn’t conclusive proof of infection because something else may have triggered the production of the detected antibodies, however unlikely that is (this isn’t helped by C-19 being a novel virus). As you said, its ability to function post-infection, does give it uses beyond PCR.
thanks for the effort.
Hemantha from Sri Lanka
“They managed to cut two months off the usual time to produce an antibody, and have gone on to create others that are so specific for SARS-CoV-2 that they will not bind to any other human coronavirus.”
Targeted payloads like some cancers.
this whole biology field is in the infancy state, not even reached the “transistor” state as the electronics did, for example if you go to a doctor with any non trivial problem they can do nothing with you, maybe you are given some pills to mask your symptoms, that probably will give you other problems as well
This sounds sorta like the process they use to make biologic class medicines and cancer treatments. As I understand it they take the monoclonal genetic code that’s matched to proteins in the cells for specified target area of the body and the code is spliced into the genetic code of a modified flu virus which is then infected in mice. This causes the mouse’s system to have an abnormal immuno response to the modified virus and results in the mouse developing a sort of leukemia that’s not fatal to the mouse but makes the mouse’s pancreas (not too sure about this part) begin to manufacture the proteins that were spliced into the rna code of the virus the mouse was infected with. Blood is drawn from the mice and the human MAB material gets filtered out. Then they mix it to attach different kinda meds or chems or radioisotope and infuse into patient in order to treat only the specific targeted part of the body with the biologic, chemo, or radio therapy and limit the exposure to the rest of the body. At least that’s the basic principal of the process, the real world process is much more complex and I probably butchered the explanation a bit (please correct me or let me know if I got part of it wrong or left anything important out) but I’m sure everyone will get the basic idea. It totally blew my mind when I was talking with my Dr about it lol. I understand the principals of chemical synthesis used in drug manufacturing but when you add a biological/ physiological dimension the process that’s just insane to me that someone was able to even cook somethin like that up in their brains much less be able to put to real world practice and cook up in a lab.
I asked my wife about rtPCR and how’s South Korea’s test is going on. She is working as clinical pathology at university hospital, and has master in biology. she is currently working on molecular biology.
In South Korea, rtPCR machines are common in clinical pathology, because of flu and MERS outbreaks. Most of hospital with clinical pathology can run can run rtPCR and a lot of agency and companies has machines. She said rtPCR machine is bit larger than micro-wave and just fit on desktop. Her hospital has more than dozen of them, only two are assigned for COVID19 tests.
Inspection is very cheap because the equipment is well supplied. If you have doctor’s direction, you can get it for free with national health insurance, but you can get tested under $100 on demand. (My wife’s hospital is charging $70.)
Commonly it said 24 hours for test result from external/outdoor inspection station, in hospitals you get faster result. My wife’s hospital is not designated hospital for testing and treat COVID19, but they run 3 test batch a day for ER and patients. It takes 3 hours of running but need more hours for prepare. So it commonly said 6 hours to get result. At the beginning it had to use two wells, but now it can be run on single well. So it run up to 90 samples in single machine.
She don’t believe in immunologic test, because it can’t detect early stages. It can be used for fast diagnose or tracing virus regional tracking, but can’t be used for quarantine or isolation. In South Korea, discharge decision is made if virus is not detected after 50 cycles of rtPCR between two days. That’s why Korea’s cure rate rises late.
The Abbott Labs ID NOW test that got approved last week uses an isothermal reaction to amplify the coronavirus RNA with a flourescently tagged primer. 5 minutes to get a positive result, longer 13 minute reaction to get a negative result.
There’s a much bigger perk to what you call the “backdoor” method of serological testing: you’ll be analyzing the bodies immune response to the antigen instead of solely it’s presence and if you design the assay to distinguish IgG from IgM you can get an idea of how long it’s been since the antigen challenged the persons immune system (IgM comes first then fades as IgG starts to ramp up)
qPCR tests are great to detect a viruses genome very early on in infection…but that doesn’t mean the virii are actually capable of infection, just that that genome is there; serological tests looking for antibodies will tell you that, albeit a few weeks post-infection. Neither are really a “gold standard”, both have tradeoffs and complement each other nicely in the clinic
I do hope that this column receives wider publication as, among the political babbling from too many quarters, there is a dearth of information that can be understood by anyone with a solid college level foundation in the sciences.
Hello,
i just have two questions if someone could elucidate me it would be wonderful..
Was covid 19 was isolated, purified and the gold standard created? Pardon if i didnt express correctly….But i have heard some doctors complaining in my country that they cannnot find the gold standard, and it should have one.
The PCR test is being used to detect the virus but i have also heard that it is running in 45 cycle and it might corrupt the sample. It will produce or not an amplification with too much noise, thus corrupting the sample?
Many thanks to all.