It wasn’t long ago that graphene seemed to take the science and engineering communities by storm. You can make bits of it with a pencil and some sticky tape, yet it had all sorts of wonderful properties. The key, of course, is that it is a single layer of atoms. Now scientists have done the same trick with boron to form borophene, and it looks to be even more exciting than graphene. You can read a pretty dense paper about the material if you want to dig deeper.
The new material is stronger and more flexible than graphene. It appears too that it could boost the performance of lithium-ion batteries. Computer simulations showed that borophene was possible back in 1990, but it wasn’t until 2015 that anyone was able to make any. The material is a good conductor of electricity and heat. It also exhibits superconductivity. Another exciting prospect is that it can be created in different arrangements, each with a unique set of properties. So you may be able to build borophene to be, for example, especially conductive or particularly strong.
Another surprising application could be hydrogen storage. Studies show that the material could store over 15% of its own weight in hydrogen, which is more than normal materials can handle. It also can act as a catalyst to decompose water into hydrogen and oxygen ions.
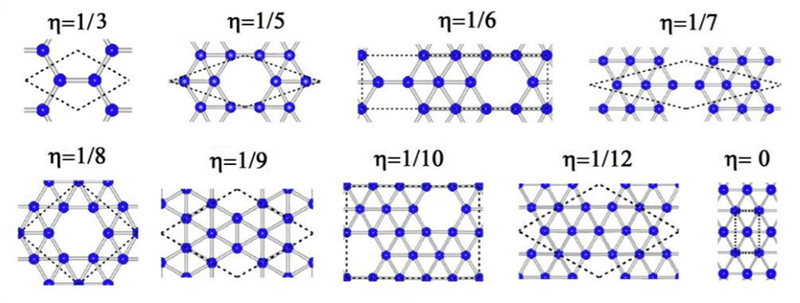
So is it all good news? No. Creating the material currently requires you to perform chemical vapor deposition of boron on a silver substrate. The arrangement of the silver atoms cause the boron atoms to assume a flat hexagonal pattern with a certain pattern of holes or vacancies that determine the material’s properties.
The other problem is that the material is highly reactive and subject to oxidation. So not only is the material hard to create, it is hard to handle, store, and use, as well. There are indications that the material may allow for some exotic sensors, too, something that graphene is also good at. Many hackers have come up with clever ways to make graphene, so now the search is on for a good way to make borophene, as well.















Tangible uses?
Please, oh please can ‘borophene’ be the cornerstone of marketing for the next few decades?
I would never stop laughing. We could have borophones, and borobikes, and borobatteries, and borophuel…
Ooh, ooh, borteries!
Set up factories in Boro Boro.
Would that not be Boring?
My molecule is also named Bort
I have so many molecules, it is difficult to remember each of their names.
I wish there was an easier way to evict some of my fat molecules.
How about something more exciting? This article is so boron’….
Perhaps “death by boro-boro”?
Maybe even a boroscope? Oh wait :-P
In Japanese, “boro” means something like “ghetto” (as an adjective).
That’s cool, it will be interesting to hear about boron material research. But I can’t get excited about any new material after buckyballs turned into such a total failure. They were going to revolutionize everything, now they are just toxic to fish.
C60 itself isn’t toxic at all (there were worries that it was toxic, but that proved due to be due to the solvent used in the production), but you don’t use pure C60. It’s the other stuff that can be toxic. There’s still plenty of fullerene research going on, it’s just the standard “overhype” problem.
I wonder would chemical vapor deposition of Nitrogen at 63.15K produce nitrophene, and would it also be more exciting than graphene.
I am sure that their are exceptions but the years of O-chem I was forced to take tells me that Nitrogen can only accept 3 bonds while boron and carbon both accept 4 bonds. The being able to accept 4 bond make them able to take a flat arrangement while Nitrogen with 3 bonds will have to take a tetrahedral arrangement.
Then we’ll just have to change the definition of “flat”.
B^)
You mean like the “flat” earthers?
Why exactly are we jamming a superflous ‘ph’ into the middle of the name?
Excellent question. Surely this is Borene.
I don’t think chemist’s scrabble sets are as big as the ones pharmaceutical companies have so their invented words lack the randomness.
Pharmacists have a spinoff of Boggle and Scrabble where they try substitute an amount of Boggle’s letters, determined by dice roll, with a previous player’s Scrabble word to form the sound of the cards from MadGab.
there is a special convention that chemists use to describe molecules. The “ene” at the end of the word has a special meaning so “borene” already describes something else.
Whatever borene is (other than the stuff this post is about), Wikipedia doesn’t know about it.
the way a chemical structure is described by its IUPAC name does not depend on whether it can actually exist.
Look up methane and borane, then look up methene. Borene cannot exist for the same reason methene cannot exist. But even though borene cannot exist a chemist can tell you why because of its name. So the name is already used to describe something else (even if it cannot exist).
Definitely needs the “ph”: borphene, borphite. Or maybe an “f”, borfite looks pleasing to the eye. Borf.
Probably was, until graphene got popular.
Superconducting under tensile strain and hole doping @ 34.8 K, so not some magic room-temperature superconductor.
Just before anyone gets too excited.
Pretty interesting though!
Thanks for pointing that out. Superconductivity at this temperature is nothing special at all.
Not had a chance to read the paper but from vague memory of my boron chemistry from university; don’t boron structures have a tendency to be quite easy to screw up? The difference between something innocuous and something mind-bogglingly toxic was being a bit lax with the synthetic conditions.
Just let me say it first,
borophene and graphene layered to make a new super [insulator, conductor, semiconductor, medicine, weapon, toilet paper] !
Oh, I forgot to include Blockchain…
borochain, or blockphene?
It might turn out to be a great discovery.
After all, why were/willbe Kirk, McCoy, Scotty, and Uhuru mining boron on Vulcan during their exile with the Klingon ship?
because as everyone (else) knows – boron is good for absorbing neutrons (hence it’s use in control rods)
And as Spock said a bit later in the future.
There was a flirtation with nuclear fission in the latter 20th Century.
I wonder if it is possible to do chemical deposition of monoatomic layers of other planar simple chemical compounds, basically, 2D crystals, for a wider range of different physical and chemical properties. E.g. compounds already containing oxygen in an ordered lattice. It seems that interesting properties of new materials come from constrains on their spatial geometry. After bashing metals as inferior to monolayered elements, they might have a return with a revenge, with their own monolayers.
They have recently been doing many things like this unsure if the reactive situation ur hoping for is available though far as the recently proclaimed time crystal tho it’s far from might be a good read for you
It’s only cause they now have a mass ability to produce graphene they say oh no it’s too readily available for the masses let’s make it specialist made only and control the amount on the market. Eh graphene itself is just now getting into production goods and that took way too long boro on the scale required is just under fantastic. especially when the extremely cheaper graphene can do the same things on a much better time and expense. Let me see it made in tonnes at a time and we may have a workable deal but as they already have 80°f supercaps at high pressure and this is Soo reactive under any exposure it’s unlikely to see any real uses unless it’s combined better
Hello,
Just a small update as of 1 April 2019 ( No not a joke !)
Thousands of companies ‘claim’ to make graphene but with no world wide standards many potential users have been ‘hoodwinked’ into wasting time testing hundreds of samples just to be disapointed
and sceptical as to it’s acclaimed benefits.
The word graphene has become nothing more than a ‘marketing tool’.
The first graphene company in the world to successfully complete the Graphene Council’s ‘Verified Graphene Producer’ programme has now arrived on the scene, named Versarien.
The trade name of the product is ‘Nanene’.
The news can be found here: http://www.versarien.com/investors/rns-announcements/
Versarien PLC – Completion of Verified Graphene Producer Programme
1st April 2019, 09:00
also a new company named Paragraf. They are producing large scale graphene sheets on a roll . They have patented 3 products . A computer chip, a fitration system and are working with a solar cell company to improve the efficiency of todays solar cells by over 50 percent. Funded by a a few venture capital companies and 2 very rich individuals this company will be one that will be very big in the graphene business for years to come.
Located in the UK and was formed in early 2019.
These 2D materials will keep evolving and exhibit exceptional properties. The cause of concern is commercializing them. None of them have been able to make a significant dent in the electronics industry as predicted in the late 2000s which coincided with the discovery of Graphene. Let us see what happens now with borphene and silicene