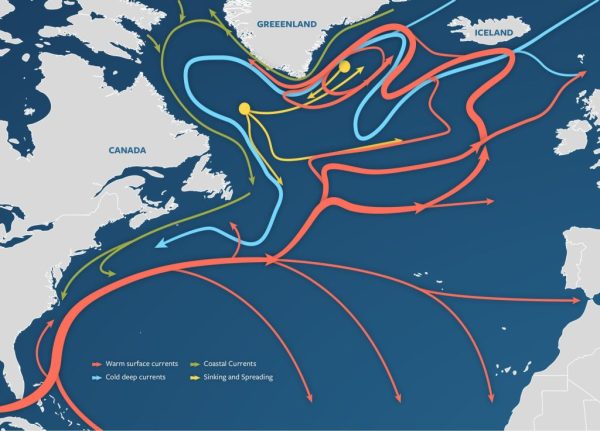
Although it can be hard to tell from looking at the often placid waters of the Earth’s oceans, their currents carry immense amounts of water around the globe on a daily basis, underlying a dynamic system that – much like the Earth’s atmosphere – plays a major role in everything from weather systems to local climates and ecosystems.
Of all these ocean currents the Atlantic meridional overturning circulation (AMOC) is perhaps the most famous, as it is basically the sole reason why Europe has the mild climate that it does today, courtesy of it carrying thermal energy from the equator all the way to the coast off Scandinavia.
Although collapsing an ocean current seems as improbable as stopping the jet streams in the upper atmosphere, it’s actually significantly easier due to how much ocean currents rely on factors that we can fairly easily influence. Over the past decades we have seen worrying signs that the AMOC is indeed weakening, with the million-dollar question being what scenario we’ll be looking at.
While collapsing the AMOC within a decade may be theoretically possible, current models seem to point towards a weakening by about half by the end of this century, with a recent research article by Valentin Portmann et al. in Science Advances going over the various statistical models to come to this conclusion.
Continue reading “AMOC And The Planet-Wide Impact Of Ocean Currents”