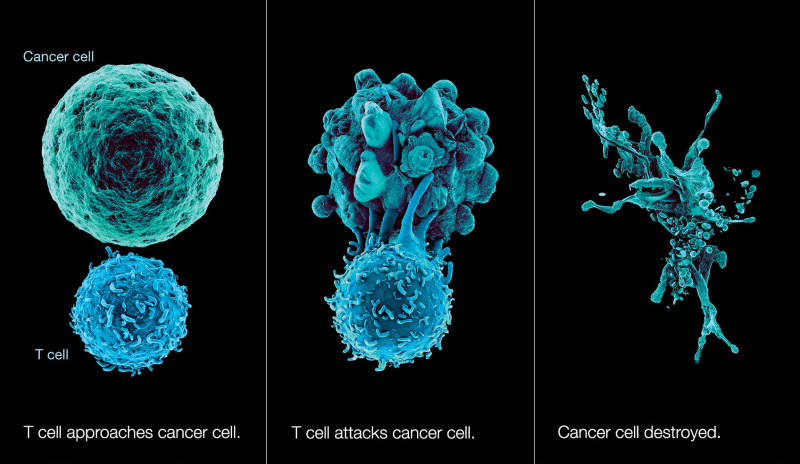
One of the human body’s greatest features is its natural antivirus protection. If your immune system is working normally, it produces legions of T-cells that go around looking for abnormalities like cancer cells just to gang up and destroy them. They do this by grabbing on to little protein fragments called antigens that live on the surface of the bad cells and tattle on their whereabouts to the immune system. Once the T-cells have a stranglehold on these antigens, they can release toxins that destroy the bad cell, while minimizing collateral damage to healthy cells.
This rather neat human trick doesn’t always work, however. Cancer cells sometimes mask themselves as healthy cells, or they otherwise thwart T-cell attacks by growing so many antigens on their surface that the T-cells have no place to grab onto.
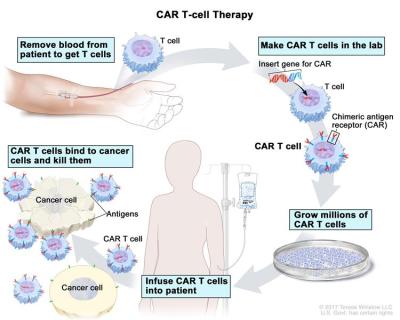
Medical science has come up with a fairly new method of outfoxing these crafty cancer cells called CAR T-cell therapy. Basically, they withdraw blood from the patient, extract the T-cells, and replace the blood. The T-cells are sent off to a CRISPR lab, where they get injected with a modified, inactive virus that introduces a new gene which causes the T-cells to sprout a little hook on their surface.
This hook, which they’ve dubbed the chimeric antigen receptor (CAR), allows the T-cell to chemically see through the cancer cells’ various disguises and attack them. The lab multiplies these super soldiers and sends them back to the treatment facility, where they are injected into the patient’s front lines.
Protein is Key to Unlocking the CAR’s Potential
Currently, T-cell therapy only seems to work in blood-based cancers like leukemia, and is ineffective at fighting solid tumor-producing cancers. But a team at Cardiff University has discovered a new T-cell that could change the game. This T-cell interacts with a certain protein called MR1 that appears on the surface of every cell in the body. When it analyzes the MR1 proteins of cancer cells, it can tell that the metabolism going on inside the cell is distorted, and reports this miscreant cell back to the immune system.
This discovery is still in its infancy, but it has great potential. Editing T-cells to interact with MR1 proteins specifically could one day kill a wide range of cancer cells in human patients. For now, the trials are limited to the lab, but the outlook is good: researchers have succeeded in destroying 10+ types of cancer, including many solid-tumor types.
CAR Is Still a Bit Rickety
CAR T-cell therapy is still quite an expensive, bleeding-edge cancer weapon, but it’s far less ravaging to the body than chemotherapy, though low doses are sometimes necessary to make room for the gene-edited cells. And there are still some kinks to be worked out — patients can have serious side effects, some of which cause neurological difficulties like confusion, and trouble with speaking and understanding language.
Another issue is the timeline for the CAR T-cell therapy process — it takes 2-3 weeks to get from initial blood withdrawal to injecting the personalized dose back into the patient. In June of 2019, trials began with an off-the-shelf version of CAR T-cells made from healthy cell donors and stored. The downside to this off-the-shelf solution is that the donor’s cells may react poorly with the recipient’s, but not always. All in all, this seems like progress to us.
Main image photo by @nci on Unsplash















Team here used CRISPR as a mechanism to generate a panel of new mutants for screening. But in treating patients this isn’t the typical way to get the modified cells. Better is to grow up a whole lot of a patent’s cells in one vat, and a whole lot of retrovirus in another. Put the two together and retrovirus induces the new protein in the T cells.
What retrovirus is really, really good at targeting human T cells? HIV. Encode CAR in the RNA of the retrovirus, then let the virus do the work getting the chimeric protein made in the T cells.
It’s possible that a poxvirus might be a better way to do this: their polymerases have better proofreading activity so you may get higher-quality results. HIV shotguns its way in.
Great pics, nature at its brutal best – eat and be eaten and all the time too. We just do the same thing with layers of abstractions as part of nature’s primal imperatives; food, shelter and sex – not necessarily in that order ;-)
I understand there’s a growing hobby gene splicing community growing in western countries, you can upload and download DNA sequences even for kids to inject into their favourite bacteria – was on a documentary about splicing the gene for spider silk into goats mammary glands so the milk produced had some proteins to make the silk, showed a line of the spider silk being pulled out of the milk onto a spool !
Thanks for post, could set up a lab for only about as much as a Tesla car – decisions decisions…
Coming soon to a Harry Potter fan forum near you, bombardier beetles spliced with lobsters for real Blast Ended Skrewts.
Not soon enough to save my son Mason, sadly. I hope this works and ends cancer everywhere for everybody else.
Sorry for your loss.
Thank you for your kind words.
My condolences,
I agree this area of medicine needs heavy investment, get out of the lab to the real world as soon as possible.
Thus, the expensive treatment phase will be shorter in the coming decades.
Mason had a bone marrow transplant at the beginning of last year. His leukemia came back in spite of it. We were hoping hard that we could get him to a CAR T study in January 2020 (about now). But he passed away only four months ago. Thank you for your kind words.
Sorry about your loss. This MR1 seems to be the best hope for cure right. Needs to go into trials as soon as possible. No delays in 2021.
I’m so sorry to hear about your son Mason. From what I can gather from your blog I see that he loved to tinker with electronics with you and I’m sure you two made some great memories with one another.
Yes, we had a great time making things together. Thanks for your kind words.
Please, tell me that this research is *not* taking place in a lab under Raccoon City…
So they remove blood, infect the current T-cells with a inactive virus (hopefully not a mutagen called the Tyrant virus or T-virus) to modify them, and then re-inject the new custom T-cells++ back into the patient.
I don’t know it all sounds perfectly fine as long as it is not being developed by Umbrella ( https://ie.linkedin.com/company/umbrella-corporation-uk ), and even if it is we can all sleep well at night knowing that it is custom made T-cells for each patient, as long as none of them are allowed to donate blood, sneeze, and keep their saliva to themselves (There are white blood cells in your spit) – all is well.
No, it is being conducted deep in the very foul heart of Wyvrnn, astride the river Selis, in the Ambrose Caverns.
By an Elf with a pet ham.
https://poignant.guide/book/chapter-4.html
It either cures you, or turns you into a Star-Monkey.
No, this is being developed by Black Mesa Labs with systems integration by Aperture Science’s Applied Nanotechnology Division.
We’ll be fine. All these worries and whistleblower allegations are Fake News.
The operative word being “could”
Sounds great. I’m diagnosed 3 years ago with CLL (chronic lymphatic leukemia). No real symptoms yet but if I can get rid of it better soon than late. This proofs to me again what a great site hackaday.com is !!! Always at the avant garde !!
And yes, let’s hope Harry Potter succeeds in fighting the evil cells so I can get my cure. Expecto Guariscum…
Currently, FDA will only allow a patient’s own cells, “unprocessed,” (meaning separated is fine; enzymes, CRISPR etc. no) to be used in treatments. Stem cells, platelet-rich plasma, collagen, fat injections for body sculpting, etc., all fall in that category. Modifying existing cells requires approval of the specific process, with clinical studies, etc.
And if it’s not industrial-scale manufactured, it will be difficult to get biopharma companies interested in something that is done on single-human scale, as there’s no profit in it.
Other countries are less stringent, meaning there are clinics getting set up in Caribbean islands, and other countries looking for medical tourism dollars. Bruce Sterling wrote about this years ago in one of his stories, positing that Indian Reservations would become biotech therapy centers.
Do you want zombies? Because this is how you get zombies. :P
Jokes aside, this is impressive work.
“neurological difficulties like confusion, and trouble with speaking and understanding language” are still being called “side effects”. For real zombies this must be much further developed, but it looks promising.
Maybe a few years?
I’m totally not being snarky here… I’m more or less an idiot who knows some vocabulary words. Can someone explain how this differs from Pembrolizumab or Cetuximab?
They use mouse antibodies.
I still find it fascinating, I am on Ocrevus for MS.
And it works the same way.
Mouse antibodies programmed to attack your antibodies, so your antibodies can’t attack the central nervous system.
So could modify an embryo so that their T-Cells already have that gene? Or could you make this like a vaccine? Every few years you go back and get a treatment to know out cancers before they get a chance to really got started?
Why not, the human genome (and I guess just about every other genome) has multiple tumor suppressor genes in it naturally, so why not start putting artificial TS genes in too?
There are all sorts of ethical issues with modifying the human genome. Ethical and moral vs practical are the issues at hand.
I think that the moral issue was solved years ago with the simple statement about abortion, “My body, my choice.” And as for modifying embryos, if you can legally suck them out and trash them why in the hell can’t you legally modify them?
Aside from the fact that the statement in question solved nothing and this is still a huge ethical/legal conflict, if it is “my body” and “my choice”, how are you going to reconcile that with imposing your genetic modification choice on an unborn child? What about the child’s choices? Where will your limits be? Just extra T cells, we’re preventing cancer! But… will you chose to make your child short or tall? Will you change its hair/eye color? What about intelligence? Will your child be an Einstein? Or maybe you prefer a sports star? “Other people think it is ok” and “well it’s legal” does not answer any ethical/moral questions.
Well the predicate of there being legal/moral questions of that type in the first place depends on how we view ourselves and others from our solipsistic like perspective. It’s clear we start out as comparatively minimal sets of simple numerical DNA patterns (amino acids easily forming naturally from sterile atoms) that develop the notion of choice over the long term with some presumption of free will, when of course that’s an illusion as free will and it’s degree inextricably dependent on numerous educations which is not a static by any means. Eg simple religious ideas only mess with emotional certainty and inevitably cause conflicts by varying levels of divisions in hierachies protecting survival of the few echelon at the top, for the most part.
Do we have anything definitive that, we either as bodies or minds and the emergence thus attained in a conjunction offers some unity of self have real choice ? Are we anything more than messy borderline chaotic chemically developed feedback paths giving us the fleeting comfort of elusive consciousness whence our inherent sense of awe (of mere existence and self observation) is exploited by religions under the guise of a soul and the flawed expectations that presumes arrogance ?
Until those issues are resolved, which is highly unlikely then who are we to ever restrict the range of permutations nature has started out with ? The incessant mobility and change of DNA and it’s patterns eg viruii modifying our gonads so we carry 7% ancient viral DNA in our genome suggests we can do what we like with our DNA and that includes our progeny and for a cohesive stable society that means before birth. Since after birth they are an effective individual with means to work with others, before then we are totally responsible and shouldn’t have that aspect removed as it is partner nature at its core.
Fwiw. Evolution marches on apace and in chaotic acceleration. For the last 40 years or so we’ve added approx 50,000 new chemicals each year and many in great number impacting all life. In the soup of chemical existence change is then not only certaintly quicker but far more varied than we can ever have imagined, it’s far sooner than you think…
I recall an old Sufi saying, pre islam:- “Our descendants may well deny lineage from us as we have from our distant ancestors”
IIUC, it’s not “your body” or “your family” with modified embryos– it’s the entire gene pool and your species’s future. Well, you could just keep them from reproducing “somehow”. I would love to be that person, summarily blacklisted from the beginning even while necessarily innocent. If society ensures that the person will never be forced to exist in such a circumstance by firmly forbidding the circumstance, then someone is doing something right.
Most commenters need to read the article or maybe this version https://www.genengnews.com/news/skeleton-key-t-cell-receptor-works-across-multiple-cancer-types/
Note specifically the HLA vs MR1 to understand the differences between traditional CAR-T and this, and why its a very interesting piece of research! (hold promise because it did work in a mouse, and with human cells in culture. Not proof it will work in human models, but way more promising than a mouse model alone!)
This is a few years old, but if you’re interested in this kind of thing, you’ll be really interested:
https://www.microsoft.com/en-us/research/video/engineering-artificial-intelligence-into-the-human-immune-system-for-cancer-therapy/#!description
Spoiler: the trials he’s talking about in the future tense did happen, and results were amazing, at first… but for some patients the modified cells didn’t stick around as intended, and for some patients the cancers evolved to evade the modified cells. Bottom line, this wasn’t the slam dunk everybody was hoping for, but it does still look promising.
The description is cringy. Please consider having a biologist proofread!
Haven’t you figured it out yet, hackaday is just crowd sourced proofreading for one of those $200 a month, invitation and subscription only, “Exclusive Executive technology insights” newsletters.
If by CAR-T cells being a “fairly new” treatment you’re referring to the 80s, sure. It’s a relative term, after all. But I feel like you’ve misidentified what is new here.
This is very interesting. I’ve been reading up on CRISPR and its impact on fighting cancer.