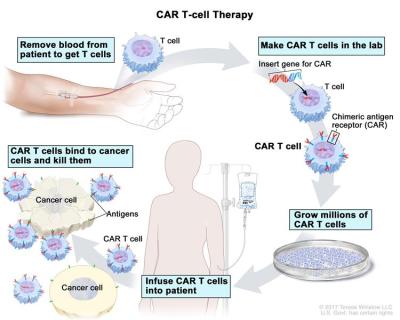
With immunotherapy increasingly making it out of the lab and into hospitals as a viable way to treat serious conditions like cancer, there’s a lot of pressure to optimize these therapies. This is especially true for therapies involving chimeric antigen receptor (CAR) T cells, which so far required a cumbersome process of extracting the patient’s T cells, modifying them ex vivo and returning the now CAR T cells to the patient’s body. After a recently published study, it seems that we may see in vivo CAR T cell therapy become reality, with all the ease of getting a vaccine shot.
We covered CAR T cells previously in the context of a way to prevent T cell exhaustion and making them more effective against certain tumors. This new study (paywalled) by [Theresa L. Hunter] et al. as published in Science demonstrates performing the CAR manipulation in vivo using CD8+ T cell targeting lipid nanoparticles containing mRNA to reprogram these T cells directly.
In rodent and non-human primate studies a clear effect on tumor control was demonstrated, with for auto-immune diseases the related B cells becoming effectively depleted. Although it’s still a long way off from human trials and market approval, this research builds upon the knowledge gained from existing mRNA vaccines, raising hopes that one day auto-immune or cancer therapy could be as simple as getting a cheap, standardized shot.