As the world pulls back from the acute phase of the COVID-19 pandemic, it enters what will be perhaps a more challenging time: managing the long-term presence of the SARS-CoV-2 virus that causes the disease. In the roughly two-century history of modern vaccination practices, we’ve gotten pretty good at finding ways to protect ourselves from infectious diseases, and there’s little doubt that we’ll do the same for SARS-CoV-2. But developing a vaccine against any virus or bacterium takes time, and in a pandemic situation, time is exactly what’s at a premium.
In an effort to create an effective vaccine against this latest viral threat, scientists and physicians around the world have been taking a different approach to inoculation. Rather than stimulating the immune system in the usual way with a weakened sample of the virus, they’re trying to use the genetic material of the virus to stimulate an immune response. These RNA vaccines are a novel approach to a novel infection, and understanding how they work will be key to deciding whether they’ll be the right way to attack this pandemic.
Stimulating by Simulating
The immune system is remarkably complex, consisting of a collection of cells and tissues that are capable of recognizing and eliminating invading organisms and developing an “institutional memory” of the conflict so that future infections can be dealt with rapidly. It can be a little hard to wrap your head around the terminology of the immune system, and so if it’s been a while since high school biology, you might want to check out the previous article I did on immune system testing for COVID-19, which covers the basics of the immune system.
At its heart, the immune system functions by responding to the proteins present in invading microbes, and it does so in a two-pronged attack. Things start with the innate immune system, a “rapid-reaction force” of cells that bind to and engulf any cell bearing non-self protein markers that manages to get inside the body. These invaders are broken down and the remaining proteins are presented to cells of the adaptive immune system, which then begins the slower process of creating antibodies that bind to the invader’s proteins with great specificity. These antibodies are greatly amplified to deal with the current infection, and when the threat is over, cells bearing these antibodies will remain at low levels, ready to mount another attack should the invader make a return visit.
Vaccination is a jump-start on the adaptive immune system’s attack. By challenging someone with either a killed sample of a pathogen, or a live version of it that has been weakened enough to not cause disease, the immune system learns what the disease looks like. By simulating an infection, vaccination primes the adaptive immune system, bestowing a degree of immunity without actually having to go through the disease process. There are, of course, caveats. Not every vaccination results in lifetime immunity; this is especially true with seasonal influenza viruses, which tend to mutate frequently and present different signatures to the immune system, requiring a new vaccine every year. But for the most part, priming the immune system through vaccination works well enough that dozens of once-deadly diseases are no longer a threat, and several have been eradicated.
Traditional vaccine production is a painfully slow process, though. Creating a version of the pathogen that correctly stimulates the immune system without causing the disease it’s meant to prevent is a tricky business. For viral vaccines, the first step is isolating the virus and growing large quantities of it. For influenza viruses, this is done using chicken eggs, to the tune of millions a year. Other viruses are grown in mammalian cell cultures, which tend to be very fussy about growing conditions. Once enough virus has been grown, it needs to be isolated from the growth medium, purified, and added to other ingredients that will make the vaccine suitable for injection. And that’s simply the manufacturing process; add in research and development and the time needed to conduct safety and efficacy trials, and time to market for a new vaccine can easily be measured in years.
The Vaccine Factory Inside You
To shorten the development time of a new vaccine, manufacturers are attempting to leverage genetic engineering by essentially making the patient the manufacturing facility. RNA vaccines work not by introducing viral proteins into the patient but rather the instructions for making those proteins in the patient’s own cells.
Recall that messenger RNA, or mRNA, is produced in cells by the process of transcription, where sections of DNA inside the nucleus of a cell are copied into the single-stranded nucleic acid RNA. This transcript moves out of the nucleus of the cell to be used by ribosomes to create strings of amino acids in a process known as translation. These polypeptide strings then fold up into complex shapes that dictate their function as finished proteins. For a bit more detail on the process, check out my article on protein folding.
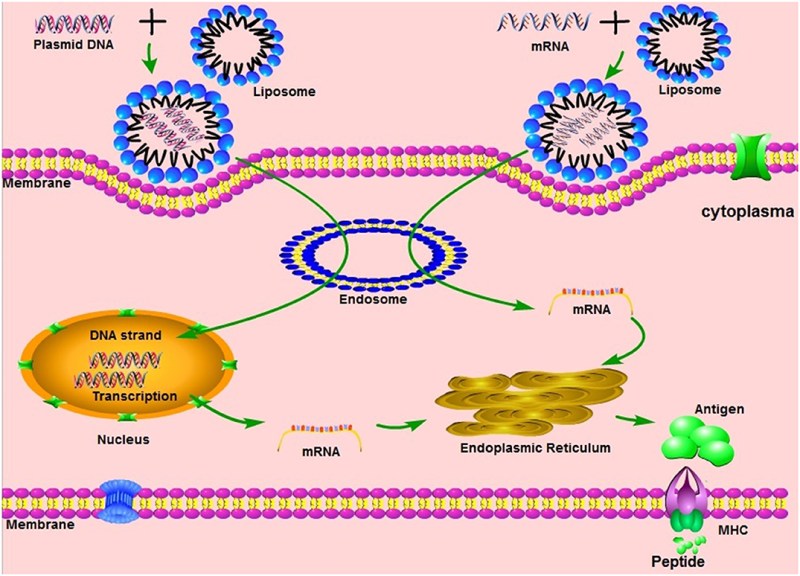
An RNA vaccine, then, seeks to place a specific RNA sequence into cells in such a way that it will be translated into proteins by them. It’s not at all unlike the actual process of viral infection, which results in the genetic payload of the virus hijacking the cell’s machinery to make more copies of itself. But where a virus injects a complete genome into the host cell, an RNA vaccine’s payload is very limited, and has no potential to destroy the cells it infects, which is what causes the symptoms associated with a viral infection.
Once an RNA vaccine has been introduced into a patient, things progress much as they would for a viral infection. The cells that take up the RNA will dutifully translate the genetic code it contains, producing the protein that the sequence codes for. In the case of SARS-CoV-2, most researchers seem to be concentrating on the viral spike proteins that stud the exterior of the virus and allow it to bind to receptors in the human respiratory and digestive tract. By producing large quantities of this protein, the patient’s own cells are doing the job normally done in chicken eggs or cell culture, with no need for expensive and time-consuming purification of the final product. And once the target protein, or immunogen, is created by the cells, it is readily available to train the adaptive immune system, which hopefully produces extended immunity.
Pros and Cons
The RNA vaccine process sounds reasonably straightforward, but as with all things involving biology, there’s more to the story. While some “naked RNA” vaccines have been tried, simply injecting a solution containing fragments of RNA won’t necessarily work. Anyone who has ever worked in a bio lab will tell you that RNA is notoriously delicate stuff. Unlike its double-stranded and relatively robust cousin DNA, RNA’s single strand leaves it open to attack by ribonucleases (RNases), enzymes that non-specifically cleave RNA into fragments. In the body, RNases play a crucial role in regulating protein production by limiting the lifetime of mRNAs, and chances are good that some or all of a vaccine consisting of just naked RNA would be degraded before producing any useful amounts of viral protein.
To work around the delivery problem, some companies are focusing on delivering RNA vaccines via lipid nanoparticles. This is exactly what it sounds like — an RNA sequence packed into a greasy little ball of fat. The idea is for the lipid container to protect the RNA from degradation and to make uptake by the patient’s cells easier, delivering the genetic payload exactly to where it can be used. Some manufacturers are even working on customized lipid packages that can target specific cell types within the body.
No matter what the delivery vector for RNA vaccines looks like, the bottom line is that we’re essentially building artificial virus parts — genetic payloads that hijack the cellular mechanism. The intention is therapeutic, of course, but the fact remains that by leveraging what our cells already do well, we might be able to get vaccines for COVID-19 and other diseases, perhaps even cancer, to a waiting world much faster than by trying to do it the traditional way.















Amazing!
Eggsactly what I thought!!1!
Congrats, we been treated (“dispensed”) some much worse articles here about this topic. It´s accurate, clear and relatively in-depth. I wish quality articles be the biggest share here.
Hmmm.
I believe that’s called “Boneless Chicken” in some places.
Dan, you’ve done a really wonderful job breaking down a complicated topic into a beautifully clear and concise overview. Thank you so much for contributing!
I see in the article you said that mammalian cell cultures are difficult to maintain and I assume chicken eggs wouldn’t be useful for all human viruses so I want to know if anyone has tried using the HELA cell line (or similar cell lines), they seem to be easier to grow and keep in the lab and are human cell lines, so what’s the deal?
The HELA line has a dubious ethical state, for starters. Also, potentially injecting people with fragments of another human cell line, with its own oddities, may also be an issue.
Well yes there may be issues with HELA cells but there are other immortal cell lines, as for the fragments, well, that’s what research is for. Still good article.
The person who did the unethical thing involved in HELA cells (taking them from a patient without explicit permission) is dead and buried. It is consummate absurdity to view HELA cells, which have benefited so many people, as tainted somehow because of their origins. I agree that an injustice was done; an appropriate remedy could be, for example, providing compensation to Ms. Lacks descendants. Shunning the benefits of this cell line would only compound evil upon evil; stopping the use of HELA cells will not benefit anyone.
“RNA vaccines via lipid nanoparticles” AKA a genetically engineered synthetic viron. What a shame that so many people are paranoid about GMOs, it is as silly as fearing all books just because they may contain something you don’t like. How many people have to die and how much economic damage needs to be done, not to mention the psychological suffering, before humanity will be allowed use genetic technology in a mature and sensible way?
That will happen AFTER humanity shows it can/will use genetic technology only in a mature and sensible way.
If humanity could only use technology AFTER it had shown it can/will use it in a mature and sensible way we would still be wandering the savannas of Africa, naked, digging up roots and grubs with our bare hands. Humanity can only invent technology and then figure out it’s costs and benefits because time has an annoying habit of only going in one direction. (Cronus f**king loves Harry Styles!)
A lot of it is done to prevent overuse of pesticides and supplemental chemicals to crops which as we already know go into the ground and water system and our bellies. I would argue that it is a mature use of the technology and has been around for decades. If you want widespread famine and folks with soft skulls and joints exploding in their bodies then by all means stick to that way of agriculture. It will never be perfect but that is the key as our knowledge evolves of genetic code and better ways to implement the good over the bad. As far as immunology is concerned, there are always gonna be good people trying to cure things and bad people that want to exploit those cures or techniques. It is a tool like any other. A hammer can drive a nail or split a skull. A computer can help us not crash or a bad actor can use it against us to unlock it and steal it. Bill Gates may be a better analogy as most of yall seem to hate Microsoft but look at the good the Gates foundation has done with malaria. Jobs would just be trying to sell them shiny facemasks that were given to celebs for free to market or buying a new immune system after he paid to jump in line. Anyway it was a good and interesting article as others have stated. Hope we see more like this on HaD :)
Maybe because GMO foods that are designed to tolerate repeated dousing with glyphosate in order to kill weeds absorb large amounts of said glyphosate, deemed by many health organizations to be a carcinogen. Hybridizing has of course been going on for decades and has produced superior plants in many cases. Designing seeds that allow poison in our food supply and poison the farmers using it has tarnished the concept of GMOs. Monsanto also wanted worldwide control of seeds. Blame them, not health conscious people.
Is there currently an RNA vaccine?
Nope. Still in development.
Edit: More specifically, there’s one in testing right now. Which means that they’ll get results around the end of June, and allowing for further certificatory stuffs, it could be available as early as fall, which would be a miracle, or maybe later. _If_ it works as well as it might.
e.g. https://biontech.de/science/pipeline, and look for BNT162. There may be other firms doing similar. This is just the one I found.
I assumed John meant another RNA vaccine for some other (not COVID19) disease… In other words, are RNA vaccines already a reliable well understood vaccine mechanism?
Or maybe not. But that’s what I would like to know :-)
Thanks for a good article with informative diagrams.
Oh, and for anyone wondering, HELA cells are an “immortal” line of human cells, which were cultured from a tumour without the consent of the human they were derived from. Wikipedia has a good piece at https://en.m.wikipedia.org/wiki/HeLa
And how can we be sure that injecting synthetic genetic material in our bodies that directly messes with cell translation to produce an nih patented spike protein won’t have long term effects on human health?
Smallpox vaccine, something I got without a blink over fifty years ago, is in the news due to monkeypox.
But it’s risky if I was to get it today. I have little immune system, deliberately by way of a drug. So little protection, and vaccines don’t do much for me. But the smallpox vaccine, unlike the mRNA vaccines, actually introduces a tiny bit of smallox into your system, which means I could get sick as a result.
So mRNA is safer than oldstyle vaccine.