When you’ve got a diabetic in your life, there are few moments in any day that are free from thoughts about insulin. Insulin is literally the first coherent thought I have every morning, when I check my daughter’s blood glucose level while she’s still asleep, and the last thought as I turn out the lights, making sure she has enough in her insulin pump to get through the night. And in between, with the constant need to calculate dosing, adjust levels, add corrections for an unexpected snack, or just looking in the fridge and counting up the number of backup vials we have on hand, insulin is a frequent if often unwanted intruder on my thoughts.
And now, as my daughter gets older and seeks like any teenager to become more independent, new thoughts about insulin have started to crop up. Insulin is expensive, and while we have excellent insurance, that can always change in a heartbeat. But even if it does, the insulin must flow — she has no choice in the matter. And so I thought it would be instructional to take a look at how insulin is made on a commercial scale, in the context of a growing movement of biohackers who are looking to build a more distributed system of insulin production. Their goal is to make insulin affordable, and with a vested interest, I want to know if they’ve got any chance of making that goal a reality.
That’s a Lot of Pigs
To understand what’s involved in making artificial insulin, the best place to start is with a look at natural insulin. Insulin is a hormone involved in the regulation of blood glucose levels, secreted by the pancreas. Specialized cells, called beta cells, sense the level of glucose in the blood, which typically spikes after eating a meal, and secrete insulin into the bloodstream in response. Insulin quickly makes its way around the body, interacting with cells by stimulating their glucose transport system to take up blood glucose for their metabolic needs.
In Type 1 diabetes, though, the ability of the pancreas to produce insulin has been destroyed. The reasons for this are unclear, but in general it’s thought to be at least partially related to autoimmunity, where someone’s own immune system recognizes the pancreatic beta cells as foreign cells and destroys them. Type 1 diabetics therefore lack the ability to sense and respond to rising blood glucose either partly or in total, meaning they have to inject insulin regularly to survive. Type 2 diabetes is a completely different disease, caused in part by the body becoming insensitive to insulin. It’s treated with a wide range of other medications, although some Type 2 diabetics take insulin too.
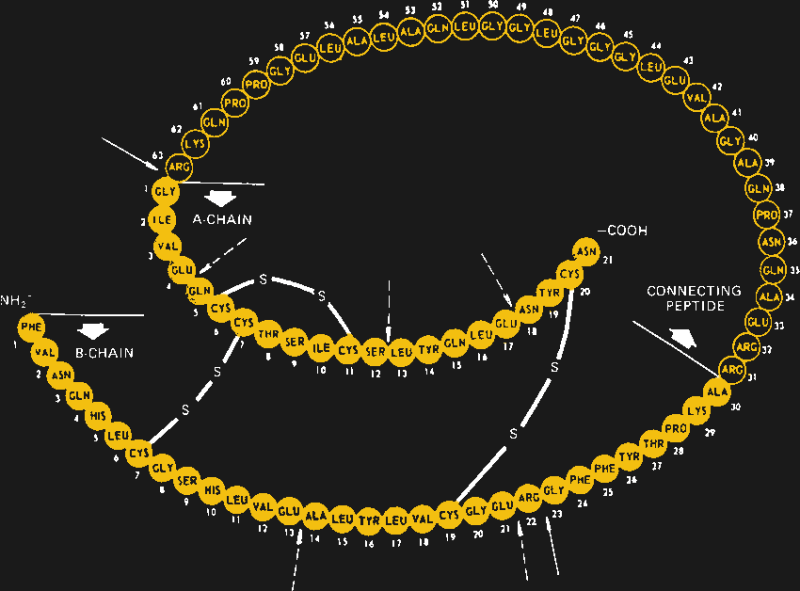
When first identified as a treatment for diabetes in 1920 by Canadian doctors Banting and Best, it came as a surprise to learn that insulin is actually a protein. Up to that point, it had been assumed that all hormones would be small molecules, so the discovery that insulin is actually a protein composed of 51 amino acids in two separate chains that are linked together in three places by disulfide bonds came as quite a shock. It would also open the door to the current recombinant insulins on the market today.

But, for the first 60 years or so of its commercialization, insulin was derived strictly from animal sources, chiefly from the pancreases of pigs. The amino acid sequence of insulin is highly conserved across species, and even insulin derived from fish has a clinical effect in humans. Porcine insulin differs from human insulin by only a single amino acid, but purifying it was a difficult process. It took two tons of slaughterhouse offal to create just 250 ml of insulin, through a thoroughly disgusting process of grinding, mincing, and extracting the insulin, and purifying it to the point where it was free enough from contaminating substances to not raise an immune response when injected. And yet, sometimes it still did.
All that changed in the 1980s with the introduction of recombinant insulin, made in genetically engineered microorganisms. By putting the human insulin gene into a small loop of DNA, called a plasmid, and coaxing those plasmids into bacteria or yeast, cell lines were created that were capable of producing vast amounts of insulin, in a way that was easy to scale up and far easier to purify than from mammalian tissue. It also had the advantage of being identical to natural human insulin, down to the last amino acid. And, it opened the door to tweaking the amino acid sequence with genetic engineering, to produce different kinds of insulin with specific characteristics.
Scaling Up
Today, most insulin is produced in the common bacterium Escherichia coli, although some manufacturers prefer to use the yeast Saccharomyces cerevisiae for their processes. Regardless of the microorganism, the process of producing a batch of insulin is pretty straightforward. Genetically modified cells are used to inoculate a large volume of liquid culture medium in a device called a bioreactor. This is basically a huge stainless steel tank to hold the fermenting culture, along with heaters, mixers, aerators, and an array of sensors to control and monitor the growth process. There are often multiple bioreactors in series, with the output of a smaller one being fed into a larger one, to step up the volume of culture gradually.
The genetically modified microorganisms are allowed to grow until they’ve reached the maximum growth rate, at which point they are harvested from the bioreactor. The first step is filtration, to separate the insulin-filled cells from the spent growth medium. The cells are then split open with a high-pressure homogenizer, leaving behind a mixture of cell debris and insulin inclusion bodies, which are sort of like crystals of the hormone that formed in the cells during fermentation.
This is the point where things get touchy. Up to now, the insulin has been pretty safe inside the cells. Once the cells are cracked open, though, all their proteins, including enzymes that degrade and recycle other proteins (proteases), can start acting on the insulin, degrading it and reducing the yield. Avoiding this is a matter of choosing the right buffers, adding the proper protease inhibitors, and keeping everything at a low temperature. Acting quickly is important too — the longer the insulin is exposed to the cellular debris, the more chance there is for degradation.
After a series of solubilization steps that further separates the insulin from cellular debris, a round of post-processing may need to be performed. In the body, insulin is expressed as a single long chain of amino acids, called proinsulin, which folds back onto itself and forms a tertiary structure between distant cysteine residues. Once that structure is established, the loop is cleaved in two places, leaving the active two-chain hormone. Manufactured insulin needs to replicate this structure; depending on the manufacturer, this may be achieved either by growing the A- and B-chains separately and combining them chemically, or by producing proinsulin and processing it after the fact.
Either way, the almost finished insulin still needs some final purification. This is often accomplished using affinity chromatography, where the insulin produced by the microorganisms is provided with a “tail” of protein. The tail allows the protein to bind to antibodies, which are attached to a solid-phase matrix of some sort. When a solution containing the fusion protein is allowed to flow over the matrix, anything not bearing the protein tail flows right out with waste, leaving the insulin fusion protein stuck to the matrix. The pure fusion product is then freed from the column by tweaking the pH of the solution, cleaved from the protein tail, and given a final purification with reverse-phase chromatography before going through rigorous QC checks and final packaging.
Smaller Might Be Better
While commercial insulin production is certainly complicated and expensive, it’s not because the process itself is difficult. Creating and maintaining transgenic cell lines, expressing fusion proteins, and purifying the end product is something that’s done by grad students in biology labs around the world every day. The complexity of commercial insulin production comes from the degree to which it must be scaled up to be financially viable, leading to huge labs with rows of bioreactors, endless miles of stainless steel piping, and an extremely complicated command and control system to make sure it all works. Comparing a modern biologics plant, which would make not only insulin but other biologic drugs like immunotherapeutics, to a semiconductor fab, is apt: both capture the essence of the problems and the potential for scaling — while it’s entirely possible to make semiconductors in your garage, it’ll never be commercially viable to do so.
Embracing this brutal reality is the key to solving the insulin supply problem, at least in the eyes of the Open Insulin Foundation. A team of biohackers with a wide geographic footprint, OIF is a non-profit focused on creating community-scale systems that can produce safe, affordable insulin. Over the past six years, their volunteers have been busy building transgenic bacteria, working on culturing processes and equipment, and dealing with the fussy purification steps, and they’ve succeeded in seeing the procedure all the way through to finished product.
Now that OIF has the science figured out, the next step is to scale it appropriately. In a presentation from 2020, Louise Lassalle, the group’s communication manager, gave a breakdown of the equipment needed to make insulin on a medium scale — in bigger batches than are possible on the benchtop, but far, far smaller than what commercial biologics plants are capable of. It’s not cheap — about $1 million worth of gear. But, that investment would produce enough insulin for 14,000 diabetics, meaning that a functioning, community-level insulin factory could conceivably be funded for about $70 per person. And that factory would produce insulin at around $6 a vial, taking into account everything from raw materials to salaries, rent, and utilities.
Granted, there are huge and potentially insurmountable legal and regulatory hurdles with this plan. It may well be that the insulin industry, with a vested interest in keeping prices high — at least in the United States — will let fly their legal dogs of war, and shut this group down if they ever get close to realizing their goal. And they’ll certainly have to deal with the Food and Drug Administration if they ever expect to actually use their insulin on humans. On the other hand, the efforts of the Open Insulin Foundation and other biohackers working on alternative sources of insulin might just win in the long run, by showing that making insulin just isn’t as hard as we’ve perhaps been lead to believe, and that maybe scaling everything up to massive proportions isn’t always in the best interests of the consumer, even if it’s profitable business.
This leads to another, possibly more important point. If the last year and a half have taught us nothing else, it’s that long supply chains need to be looked at with skepticism. When a pharmaceutical company builds a mega factory for financial reasons, it creates a single point of failure that probably doesn’t need to be there. We’ve seen how easily supply lines can be pinched, and it seems incredibly foolish to engineer into a system the potential to shut down the world’s supply of any medicine, especially one as vital as insulin. OIF’s vision of distributed mini-factories for insulin production seems smarter right now, and will probably only seem like a better idea as time goes on and complex systems become less and less reliable.

















A lifelong dream for me. I have an old bookmark html that had my “FOS Insulin” folder.. Time to dig that up..
I always found it astounding how the patents were made and how this market came to be how it is now.. This civilization’s MO is not sustainable.
That html is from 2009.. When I gave up.
The current prices are artificially high. Nice write up, I was wondering about the whole process.
It’s not artificially high. It’s a substance that is mandatory for some to live. Given that you can ask the price you want by being a bit lower than extortion rate.
Any price above break-even pricing is artificially high. The fact you can inflate it up to extortion-tier because it’s indispensable doesn’t change that.
This is just quibbling over the definition of the word ‘artificial’. In a society with healthcare regulated more by capitalism than the government, the ‘natural’ price for something is as much as the market will pay. See also: rent, where the price of owning and maintaining the item in question is far, far lower (in most markets) than the price being charged for the item.
If it is a natural consequence of the system you’re living in, it’s not artificial. Or it is artificial, because that system itself is artificial, which leads to questions like what to do about it.
Corruption:
“American Insulin Prices Are Off The Charts”
https://www.statista.com/chart/23127/average-price-per-standard-unit-of-insulin/
This is legitimately cool, but why is there even a need? Insulin is cheap/free and provided by the NHS or your country’s equivalent. Is this more of a “in case things really go south” scenario, or is it a case of wanting to produce things locally for the sake of resilience?
“who cares about mad cow; meat comes from the supermarket!”
Insulin is not cheap/free in the US. It goes for $150 to $300 a vial depending on which kind someone needs. People in recent years have been under-dosing themselves to save money, putting their health or lives at risk.
That’s terrifying. I should read up on it.
How much is in one vial?
It depends on how you buy it. I use U-100 Novolog. It’s a 10ml vial with 100 “unit” per ml. One vial lasts me about 10 days in my pump.
A problem that could be solved with the stroke of a pen. Instead people risk lives (by under-dosing or some day home-brewing) to work around an artificial barrier. Not unlike desperate immigrants risking their lives in hope of a better future. The state of healthcare in the US is sad.
I want to help immigrants via my government using my tax dollars for humanitarian purposes instead of wasting my tax dollars on two-decade wars that accomplish nothing.
The insulin market is very confusing to people who live without the to buy insulin for a loved one.
People like to complain about the US drug market, but they ignore many other options and focus on ‘list price’ and brand-name insulin, assuming a person has no insurance coverage and has a specific medical need that requires the latest innovation in insulin.
In America, it is estimated that 12% of the population lacks healthcare coverage (40 million out of 320 million), and many of them would qualify for subsidized/free coverage, but opt not to apply. So that leaves 88% of America covered by health insurance.
As far as insulin goes, for those without a prescription plan, there are discount plans like GoodRX which negotiate vastly lower prices for prescription drugs – in one example, found on their website, I found “1 carton (5 flexpens) of insulin aspart 3ml” for as low as $110 from a National chain pharmacy – that’s a cash price, no insurance required. Now, being a non-diabetic person, I’ve no idea how that relates to the $385 item people like to complain about, but it seems to me that with just a little bit of effort, significant savings can be found in your own neighborhood.
If you have severe diabetes and are uninsured, sign up for coverage – it certainly cost less than buying name-brand drugs a list price – and you can’t be denied coverage, due to PPACA (Obamacare).
If you must buy insulin retail, explore all available options, and discuss with your doctor the possibility of using a generic form of insulin., rather than a name-brand.
The vast, vast majority of Americans currently have healthcare coverage, and every American is eligible for coverage – every American.
Please, provide a citation for your $150-300/vial insulin, I found generic insulin in a five-pack for $112 at my local pharmacy (Walgreens) through GoodRX:
“1 carton (5 flexpens) of insulin aspart 3ml”
https://m.goodrx.com/insulin-aspart?r=504
I have no idea about the insulin market, but it seems that the worst-case price for a five-pack of vials, got transformed into the worst-case rice for a single vial, then it morphed from worst-case to average price.
The reality of the PPACA act (ObamaCare) means that every American can get insurance, and that insurance would absolutely reduce the cost of insulin – I can’t explain why some 40 million Americans choose not to be covered.
There is a difference between having “healthcare” and coverage and what that coverage in-tails. Affordable healthcare act or Obama care as you put it (really it’s a republican think tank idea), Affordable healthcare act is a shit program that forces you to be insured by private companies for coverage that really only covers you in a catastrophic accident. whilst the private company reduces their operating cost, Also most insurance provide even to employees thru employers can cost at the upper end around $950 a month (subsidized in part by the employer, and the employee pays $75, $150 , x) is shit, the employee is still paying about 4000 to 6000 a year out of pocket before hitting deductible then the 80 coverage 20 you pay rule. you think a for profit model is tailored for the well being, prosperity, and health of humanity. hey f@uck it though right it’s a live and let die kind of world (it’s not or you would see what the really happens) . there is a debate to be had about profits greed ethics and when it’s OK and to what limit, that debate should not be had on basic human needs. that;s not to say an individual provided that social (communal service)should be abusing that service. healthcare in America is a joke.
I span the political spectrum ,so pin me where you think I am because you will be wrong.
also i don’t know where you fall on the political spectrum and i don’t care. my goal is a better system and world for the benefit of both of us and all humanity.
note this comment like my last will be removed by mods after a short time. got to keep the system in check. no ghost in the shells
Insurance notwithstanding, I’m sure you’re aware that there are many types of insulin that a person can be on. The one you’ve chosen is a fast acting insulin, often given as a rapid action to deal with meal times. But a lot of people are also on long acting insulin. Check the prices for insulin glargine, insulin detemir, and another fast acting, insulin lispro. Ie, costs add up.
I spent about $1,200/month in total for ACA insurance for my partner and I, Humalog insulin, supplies for my insulin pump, test strips, and necessary health care visits. Every American has the possibility of “coverage”, but not everyone has the opportunity to afford to use the coverage they buy. It’s absolutely a clusterfuck and was one factor in wanting to leave the country for me.
@ Ken said: “The reality of the PPACA act (ObamaCare) means that every American can get insurance…”
I was forced onto ObamaCare. I lost coverage for me and my employees as a result. It was a U.S. based company primarily working outside the U.S. My superb Global health insurance company (Netherlands based) said they could no longer insure us in the U.S. because of ObamaCare. After a couple-few years the Netherlands based provider did find a way to do it by working around all the intentional obstacles ObamaCare introduced to drive Americans towards a Socialist health care system. But by then it was too late for my company. I had to close it and return to the U.S. for the first time in many years. What a revelation. I returned to a U.S. in rapid decline with a taxpayer-funded education system that indoctrinates American children to hate America. (How did we let that happen, and why is it still going on?) So now I’m stuck with Obamacare; almost $2K per month for me alone (in good health) with a ~$7K per year deductible – and that’s for what I call “bare-bones” coverage. Plus I am locked into a horrible HMO system now. I can no longer choose my Doctor. The deductible is so high I must forego even sensible preventive care. ObamaCare destroyed my life.
Obamacare is not the problem. The fact that we don’t have socialized medicine is. Too many companies trying to make a buck off the back of Americans who only want to be healthy. This includes insurance companies, health organizations, pharmaceutical companies, Doctors, lawyers, and of course lobbyists and politicians. Lets face it, medical care is big money and we are all just chumps, waiting to be fleeced.
Personally, as a retired person (living on a minimum SS check) and a Veteran, I get my primary health care from my local VA Hospital. I’m lucky because my VA Hospital is closely affiliated with OHSU (Oregon Health Science University) one of the premier medical schools on the west coast. I would say that my care is equivalent to what I would get if we had socialized medical care. Yes, sometimes I have to wait for an appointment but who doesn’t. My drugs are cheap with only a small copay. If I have an emergency I get in immediately and don’t have to worry about going bankrupt. Frankly, I love my VA. I get great care and the people are wonderful. I wish every American had access to this level of care
If the American people would quit the political infighting and just plain whining, and insist on a decent public health care system, they could force politicians to make the changes necessary. The problem is, too many people actually listen too and believe the false propaganda they hear on certain news shows and usually, right wing radio.Too many people are too willing to believe disinformation campaigns and conspiracy theories ladled out by self serving pundits whose only goal is to make a buck by any means possible. When Americans get tired of being screwed, things will change. Not a moment sooner.
BTW, I really like the idea of locally produced insulin, using an open source model. It makes perfect sense economically but will likely will never happen. This model would cut into the profits of large, well connected corporations that make big money milking their dependent clients. Pharmaceutical companies prefer to treat the symptoms of a disease, rather than cure that disease. And that hoopla about the high cost of research is also somewhat of a fabrication. A lot of the basic research and medical breakthroughs are publically funded through research grants. After the research is completed the findings are released to private companies who turn the fundamental research into commercial products. The aforementioned big investments, usually go to FDA approval trials, production facilities, advertising, and the like. Few significant medical breakthroughs are made by big pharma. Mostly big profits. I would like to see the American people keep a portion of the profits from medical breakthroughs funded by government grants, but that would be socialism, government interference in capitalism. Besides, we can always recoup our investment through income taxes. Oh, that’s right. Most big corporations don’t pay that much in taxes anymore. Oh well, there’s always trickle down. Have you been trickled on lately?
I just wanted to chime in on this to help you understand why 40 million Americans “choose” not to have health insurance (in Oregon at least). A single person can expect to pay $700-$1000 a month for terrible insurance if their employer doesn’t offer coverage. If they make more than $1400 a month they do NOT qualify for free coverage. Minimum wage comes in around $2000. When you consider cost of living, its pretty obvious why they “choose” to opt out….
Then this is technology being mis-applied to solve a societal problem that it seems only the USA is afflicted with.
Depending on corporations to produce it in large volumes is not a comforting thought when questioning how long it will be there.
“Insulin is expensive” … in some countries, the price of drugs is shared between the citizens …
Yes … I talk about social security which is a good starting point.
Next step is to have drug formula be opensource: I don’t agree that big pharmaceutic companies make huge incomes with the COVID pandemic, for example. First they said “price is high because we need to pay for all the R&D” … next they increased the price by 25% (the R&D was supposed to be already paid …).
In the Netherlands the price of 3cc is around 6 to 7 euro. With a normal insurance, which is mandatory for everyone here, the first 250 euro you spend on medicines per year you have to pay yourself after that the insurance company pays. A normal insurance, costs around 125 euro per month.
If drug companies weren’t allowed to make profit then almost zero drugs would ever be developed. The list of drugs developed in capitalist countries is far longer than the ones developed in communist ones. I’m not against the idea of open source drug development, but I’ve not yet seen any evidence that it will deliver anything like the output of ‘big pharma’. The same can obviously be said of many technologies. Software seems to stand out as the main exemplar of successful open source development.
As it is, the patent situation only gives a certain time period for the inventing company to make profits (I think 20 years from first filing), after which the drug can be made and sold by any generics company that wants to.
That’s a fallacy. Why do you think the little girl who wanted to heal people, that became a young woman going to college to learn how to heal people, that became a researcher looking for ways to heal people did all of this? For profit?
Capitalistic reasoning is a cop-out. Capitalist thinkers are just small minded and ultimately greedy. And as long as capitalist thinkers rule, society as a whole is ultimately doomed. Sure capitalist countries have developed more, faster. But to what end. Altruism? Never! Profit and growth are the only goals. Companies build under capitalism as a unit could care less about a (or a million) human life(s).
A modified heritage check system would be a great start to a truly altruistic society.
Hah! The “Capitalist” Big Pharma boys payed to put Biden in office. The Biden Puppetmasters are all corrupt Marxists.
Wait, you’re saying Big Pharma are Marxists? That is a strange, strange claim.
Drone, I hear you droning on. You are just regurgitating a large steaming batch of right wing propaganda. I think you managed to include at least 4 talking points in only two sentences. Good job. Have you ever had an original thought?
Remember that the ‘little girl’ probably needs to buy textbooks, food, pay rent, buy lab equipment, hugely expensive production equipment, people to run clinical trials, etc. etc. if she is going to bring drugs to market.
I am very much against the Martin Shkreli types. I do however think that we need corporations (perhaps modified) in order to focus the resources needed for drugs to be discovered, tested and produced.
The state could easily offer to buy patents, giving big pharma the choice of instant profit and then either allowing use of the patent to any domestic manufacturer for no charge or some reasonable agreement for outsiders…
Literally no state does this, not even those with mandatory insurance (read – all of EU), where it would save considerable amount of $$$ from the collective insurance.
You can easily feed the wolf while keeping the sheep alive, yet nothing has been done. Ever.
IIRC it was attempted in India for AIDS’s drugs. India decided no to pay for the outrageous price asked by Big Pharma, and announced that it won’t respect the patent on the drug and produce itself. The Big Pharma decided to counter attack against India at WTO but IIRC they’ve lost. As a result, India is now able to heal its own people for a price that would not pay for paper and pencil in such companies. India treatment is $350/year/person while it’s more than $6400 in US.
Go look at Cuba, a leader in biopharmaceuticals and vaccines.
https://www.ineteconomics.org/perspectives/blog/how-cuba-became-a-biopharma-juggernaut
And then tell me how it has to be based on profit.
It all goes down to what we value more: Human rights or property rights.
The lega/regulatory hurdles are indeed steep. Bioreactor facilities need to be built, inspected, then mothballed until the product is approved. Clinical studies have to be run to prove bioequivalence — less onerous than a ‘new chemical entity’ but still significant. FDA approved the first interchangable biosimilar, meaning the pharmacy can substitute it instead of the doctor specifically prescribing that version just this last month — and it was for insulin.
https://www.fda.gov/news-events/press-announcements/fda-approves-first-interchangeable-biosimilar-insulin-product-treatment-diabetes
In Canada we are celebrating the 100th anniversary of the discovery of Insulin. With a set of $2 coins.
https://www.mint.ca/store/coins/discovery-of-insulin—commemorative-collector-keepsake-2021-prod3800021
Yeah, but what have we done lately? I’m far from a socialist (usually because of the smell of body odour and patchouli) but we never should have sold Connaught Labs!
https://en.wikipedia.org/wiki/Connaught_Laboratories
US healthcare is sick.
I love the spirit of this but I fear this is going to get someone killed. I certainly hope they come out with a way to easily test successful production.
It very likely will. That’s unfortunately an inescapable part of the deal when making drugs. This is why clinical trials exist: so that if a product is gonna kill someone, it’s hopefully limited to just the people who knew the risk (or at least signed a piece of paper saying that they did.) But how many lives could be saved by having access to affordable insulin? To say nothing of the number of lives significantly improved by access. It’s a morbid math problem I readily admit, but I think it’s one worth answering.
It doesn’t even have to directly be taken to save lives. If they manage to scare the pharma industry enough, the industry will lower prices to try and keep them out, helping everyone. As long as the open source insulin threat remains viable the prices could be held below the current monopoly-level heights.
(Literal) million dollar idea: prove price collusion in insulin market, file class action lawsuit. Your cut is a few percent of a few billion.
Corporation = Legal person without a conscience.
Walmart Relion insulin for $25 a 1cc vial. No presciption required in some states, do not scrimp on your insulin.
Opps thats $25 for a 10cc or 1000units of U100 Walmart Relion rebranded Nono Nordisk
Being a professor in this field I as well find this very worrying… producing recombinant pharmaceuticals requires very much knowledge on the downstream processing which next to constantly audited processes, very good analytics. During Insulin production for example non-canonical amino acids can be incorporated making some of the insulin not usable. I firmly believe that there should be a political solution rather than homebrew.
“…there should be a political solution rather than homebrew.”
This!
It would be interesting to know the profit margin on the production of insulin. Is insulin a “cash cow” for pharmaceutical companies? If insulin becomes cheaper by mandate, what does that do to the prices of other medical drugs?
“Being a professor in this field I” …should perhaps assume the people working on this are learning as much as they possibly can to make it work correctly and safely. And/or the people working on this are scientists, physicians, etc. who working in the field now with current knowledge and training.
Political solutions are not always solutions, and political solutions based on intended altruism are often counterproductive.
Here’s an alternative political solution: open pharmaceutical markets in other countries so they are not subsidized by patients in the US.
Do need to allow for “oh no, the power was out for half an hour… Oh, it’ll be okay” or “got a GREAT deal on those reagents!” (because they are lower purity, or contaminated and were rejected by incoming inspection elsewhere) or just plain typos.
All too easy to cut corners when you’re close to bankruptcy, or don’t understand why it matters (or don’t want to), or just HAVE to get that promotion. Humans have weaknesses, and any system needs to allow for that.
Nope nope nope. Pharma manufacturing must follow documented quality controls. Any change to suppliers, environmental conditions, etc, must be approved (major changes) or notified (minor) to the agency, subject to bring fined or shut down.
I think the issue worth focusing on is the “lots of little factories” alternative to a few mega-factories producing all the world’s insulin, but I’m doubtful of not-for-profit pharmaceutical factories. The demands of producing safe, quality medical-grade drugs is an expensive proposition, requiring top-skills that are highly-sought-after by Pharma companies, and are well compensated – do you really imagine all those workers would take significant pay cuts to choose to work for a not-for-profit drug company? My point is the significant cost savings you imagine could be realized are an illusion – producing smaller batches is more expensive than big batches (reverse economies of scale) – and the huge profits you think you’re going to eliminate really aren’t there (it doesn’t cost $6 to make a five vial pack of insulin, store it, and ship it to a pharmacy, where the cost explodes to $350). There may be some savings, but I don’t think they will be massive. The real impact a number of smaller insulin plants might have on the market would be to drive prices down through competition. Until your doctor, fresh from lunch with their big pharma drug rep tells you that you absolutely need the latest, greatest, brand-name insulin for “medical reasons.”
“The demands of producing safe, quality medical-grade drugs is an expensive proposition, requiring top-skills that are highly-sought-after by Pharma companies, and are well compensated – do you really imagine all those workers would take significant pay cuts to choose to work for a not-for-profit drug company?”
Like say…a public defender?
10Ml per Vial, there are different concentrations ranging from U-100 to U-500. U-500 is basically 5x the power of U-100.
My last invoice for $2100 through the mail for 10 vials should be more than enough evidence. I’ve paid the cash price in a pharmacy and I’ve paid for black market versions as well as with insurance (oddly enough the black market folks were easier to deal with than my pharmacist, go figure).
“Plant-based vaccines for COVID-19 and other viruses | COVID-19 Special”
https://youtu.be/-HkM2ipSD4k
…And for obscene profits, consider the cost of those little strips to measure your blood sugar so you can get the right dose. They’re also about 20% accurate, according to my endocrinologist.
Yes, Insulin can be purchased for about $25 a vial at Walmart in the US.
Like any pharmaceutical, new formulations that work better, faster, longer, or “are just different” have been developed – and it’s those new varieties that cost a bunch more. An it’s the new varieties that we seem to think are medically necessary.
Walmart sells “Regular” and “NPH”. (The NPH starts working a bit faster). Both are now considered a last resort because they require a lot more careful diet and more discipline to use than the more modern stuff , but they both work and will keep you alive.
I’ve become a bit tired of manipulative news reports showing a grieving individual claiming their relative died because insulin is too expensive. If I couldn’t afford Novolog, I would read the directions, and learn how to use the NPH.
So best case is about $1000/year give the rate of 1 vial for 10 days quoted above. I’m sure there are plenty of people who dont have $1000/year of disposable income, meaning they have to sacrifice this, or some other necessity.
If you think NPH is nearly as safe for the average user to regulate as glargine, you’re either retarded or have never know a diabetic (or both). Yeah, it CAN save a person. It can also destroy a stable A1C and put a person in a comma after 3 days. Brother in laws type 1. His doctors straight up yelled at him for even trying it. 3 different doctors, all say only for animals and only out of desperation. Even so, made my cat loose his teeth.
Insulin comes with directions, people’s metabolisms don’t. NPH, and to a lesser degree Novolog, have highly varied interactions with different people. Even so, my brother can easily eat a vial every couple of days. $250-300/month, for a dangerously inferior product, just so people like you can continue not caring about some of the highest insulin prices in the World?
Please. How about this, YOU regulate a diabetic CAT on Novolog for a year. Then, when your cat’s dead, you can get back on here and correct your worthless statements. Or maybe, someone who doesn’t know anything about actually caring for diabetics should refrain from laughably uniformed comments on it. Lord knows homebrew insulin’s gonna’ be safer than your expert ‘advice’.
I find it interesting that they have to destroy the yeast to get the insulin. Somebody (maybe a biohacker) should come up with a way to modify the yeast to simply excrete the insulin into solution from which it can be harvested.
There are organisms that do this naturally (Pichia pastoris) and E. coli and Bakers yeast can be made to as well. The batch cultures are still destroyed after expression because growing them continuously past a point is much less efficient due to nutrient limitations and pH changes which are hard to regulate
One relatively simple thing to do to determine if the price of insulin is reasonable, is to make an inventory of different prices for the stuff worldwide and over time, and compare those numbers with each other.
The best hacks are freedom hacks, anything that gets people off a dependency on big pharma and fragile supply chains is a very good thing, because if the right to repair your own hardware matters then that includes your not so hard wetwear.
bit offtopic, but I’ve been making my own liposomal vitamin c since 2016, based off of paul dazzler’s recipe.
I normally get my insulin free in the UK but had an extended stay in Brazil. A 3ml Lilly Humalog Mix 25 KwikPen (which normally lasts me a week) cost US$8 at the local pharmacy.
Production of pure insulin is covered here but I’m interested to know how the fill-finish into vials will work. They are nornally two separate processes with different equipment and regulatory requirements. We want cheap drugs but they also need to be safe. There are lots of critical manufacturing steps where degeneration can occur. As another comment says, we need a political solution.
As someone who works with one of the biggest insulin manufacturers on the planet, specifically on their insulin production lines, I can confidently say that the FDA will never, ever let operations like the ones described in this article come to pass. The sheer number of people and amount of man hours it takes simply to collect, analyze, and act on data as required by regulation is staggering. Think entire departments of dozens, if not hundreds of people. This is not an issue of scale, nor is it a function of how easy or difficult it is to make insulin. Whether you want to sell billions of vials a year or dozens, the FDA (and its equivalent agencies all over the world, depending on where you’re selling) will hold you to the same extremely stringent regulations. This is ultimately a good thing, since it prevents harm to patients, but it would be impossible for a neighborhood-sized operation to keep up with regulatory requirements and still operate at a profit.
Lots of people like to talk about the cost of manufacturing a vial of insulin as if the only expenses involved were the price of lab equipment and CEO salaries, but there is so much more that goes into making pharmaceuticals. I’m not saying the price payed by the patient shouldn’t be subsidized by governments or better healthcare systems, but none of that would change the requirements placed on an aspiring local manufacturer to keep regulators happy with their operations. If you can’t afford to hire an entire floor of regulatory compliance people, you can’t afford to make insulin, period.
And exactly how much after taxes profit did your company make last year? your CEO’s monthly paycheck is bigger than the cost of my house before stock options, or other perks.
Read my comment more carefully. Notice that I never said anything about the current price of insulin being reasonable. On the contrary, I actually indicated that the price payed by the patient should be lower. You are missing the point here. A local operation like the one described in the article could pay their CEO one dollar per year and they would still not have enough money to ensure regulatory compliance with the FDA or its equivalent agencies.
Even if one ignores the near impossibility of a backyard operation being compliant with regulatory requirements, insulin has quite a narrow therapeutic index, and an error in effective bioactive concentration of 20% would be enough to kill or leave someone a vegetable.
Actual Biochemist here. One thing that I have yet to see addressed by the Open Insulin crowd and biohacking in general is the painstaking, expensive and difficult steps of eliminating endotoxin contamination. Endotoxins are essentially any of the bits from the bacteria/yeast etc. that will trigger an immune response; often rapid and fatal. They are typically bits of sugar from the cell surfaces but any fragment of LPS (Lipo polysaccharide), protein, or glycan can cause this. This was arguably the most difficult part of producing recombinant insulin and what distinguishes all, “I’ve expressed proteins in E. coli” from, “I would definitely inject this into my arm.” There is absolutely no room for error, for injectibles endotoxin levels have to be less than 0.5 EU/ml sometimes lower. This corresponds to 50 pg (pico = trillionths) of a gram per ml of drug. To put it in perspective to something you can see. If you took an Ibruprofen tablet (200mg) and cut it into 100 pieces/, then cut one of those pieces into 1000 pieces, then cut one those into 1000 pieces then cut one of those into four pieces or so you would have about 50pg left. Or in other words dissolving one advil into 4 million Liters of water gets you a concentration of 50pg of advil /ml Thats about 1 advil in two olympic pools.
Sounds like that might be a suitable target for open source, DIY or cottage industry. Certainly easy to compare to the commercial test strips for accuracy. Any biotech folks know if this is feasable?
Where is it?