There’s something intrinsically simple about the concept of carbon (CO2) capture: you simply have the CO2 molecules absorbed or adsorbed by something, after which you separate the thus captured CO2 and put it somewhere safe. Unfortunately, in physics and chemistry what seems easy and straightforward tends to be anything but simple, let alone energy efficient. While methods for carbon capture have been around for decades, making it economically viable has always been a struggle.
This is true both for carbon capture and storage/sequestration (CCS) as well as carbon capture and utilization (CCU). Whereas the former seeks to store and ideally permanently remove (sequester) carbon from the atmosphere, the latter captures carbon dioxide for use in e.g. industrial processes.
Recently, Pacific Northwest National Laboratory (PNNL) has announced a breakthrough CCU concept, involving using a new amine-based solvent (2-EEMPA) that is supposed to be not only more efficient than e.g. the previously commonly used MEA, but also compatible with directly creating methane in the same process.
Since methane forms the major component in natural gas, might this be a way for CCU to create a carbon-neutral source of synthetic natural gas (SNG)?
Carbon Capture in a Nutshell
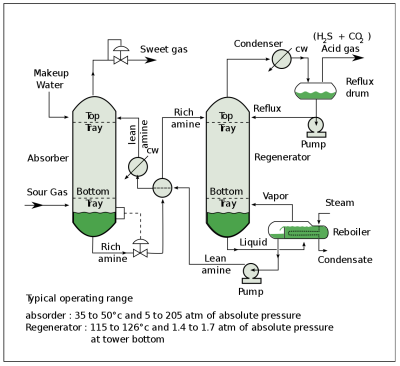
The most common type of carbon capture (CC) system is a CO2 scrubber that is used with fossil fuel power plants or similar sources of flue gas. This gas is led through the liquid solvent, typically amine-based. Amines are derivatives of ammonia, with at least one of its three hydrogen atoms having been replaced by a substituent. Monoethanolamine (C2H7NO, MEA) is a primary amine that in a water-based solution can efficiently absorb CO2 and H2S from flue gas.
When the resulting CO2 rich amine solvent is then led into a regenerator unit which heats up the rich solvent to about 118 °C at 69 kPa, it causes the absorption to be reversed and the gases to be released. Most of the MEA is recovered in this manner and can then be returned to absorb more CO2 from the flue gas.
Problems with MEA include the high water content in the solvent. Water has a high specific heat, which means it takes a lot of energy to get hot. MEA also reacts with carbonyl sulfide (COS) and carbon disulfide (CS2) to form heat-stable salts, which remove MEA from the process and require an additional process step to be removed.
After the CO2 gas has been captured this way, it is generally compressed before transport for use, storage or sequestration. The heating up of the rich amine solvent in the regeneration process, as well as the compression of the captured carbon dioxide all cost considerable amounts of energy. This is where the economics of CC are not very favorable, and prefer input gases that are already rich in the gas that is to be captured.
Over the years, various alternatives have been developed to MEA which require less water, and processes that omit or reduce the compression step. Examples of the former are for example Shell’s Cansolv capture technology and Mitsubishi Heavy Industry’s KM CDR process with its proprietary KS-1 solvent. These all have the same goals: use less water, reduce the amount of amine solvent caught in the flue gas that’s emitted into the atmosphere and improvement of the recovery rate of the solvent while reducing energy requirements.
None of these processes are suitable for something like direct air CCS, though. Although one can technically lead atmospheric air through one of these capture plants, the difference in CO2 content in the air versus flue gas is immensely different (>9% in flue gas versus 0.04% in air), leading to a very low efficiency rating.
Even so, with flue gas the capture rate of CO2 is generally above 90%, but <99% (claimed 98% for Cansolv, >90% for KM CDR). This means that although most of the CO2 is indeed captured, some of it still is emitted with the flue gas, along with amine solvents.
PNNL’s 2-EEMPA
In PNNL’s paper by Heldebrant et al. (2021) titled Integrated Capture and Conversion of CO2 to Methane using a Water-lean, Post-Combustion CO2 Capture Solvent a number of claims are made:
- >90% conversion of captured CO2 to hydrocarbons (mostly methane).
- More efficient than the usual Sabatier process (skipping the CO2 compression & transport steps).
- Process conditions are 170 °C and <15 bar H2 pressure with ruthenium catalyst.
- Better performance of 2-EEMPA than MEA.
There are a number of steps involved in this process, from absorbing the CO2, to getting it to the point where it can react with the hydrogen that is added to create the hydrocarbons. Heldebrant et al. first describe the Sabatier process, using a combined cycle natural gas turbine plant equipped with Shell’s Cansolv (using a 50% by weight amine solvent) process as example:
- CO2 captured in the absorber & released in the stripper (regenerator) at 2 bars of pressure.
- Pure CO2 is compressed and mixed with hydrogen.
- Mixture is added to methanation reactor for the Sabatier reaction.
- The Sabatier reaction runs at 350 °C and 30 bar with an Ru/Al2O3 catalyst.
- The exothermic reaction provides heat for the stripper unit and power generation.
The PNNL version does not use the proprietary Cansolv process, but instead its own 2-EEMPA (N-(2-ethoxyethyl)-3-morpholinopropan-1-amine). Heldebrant et al. claim a CO2 capture efficiency of >95% with coal-derived flue gas. 2-EEMPA-based solvent is projected to have a ~4% water content by weight in operation. At this water ratio, 74% of the CO2 captured by 2-EEMPA (as EEMPA-carbamate) is converted to hydrocarbons when hydrogen is introduced and with an Ru/Al2O3 catalyst present. Of these, 92% of these were methane.
At the same conditions, MEA showed a conversion ratio to hydrocarbons of <20%. The entire process chain can be summarized as in the following graphic:
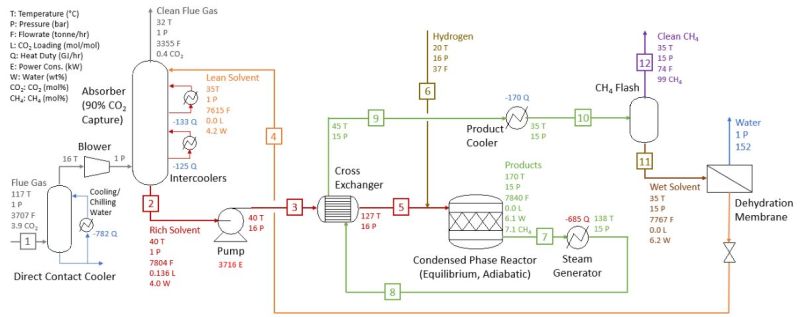
The benefits compared to the traditional Sabatier process are a lower reaction temperature (170 °C instead of 350 °C), lower pressure (15 vs 30 bar) and lower cost for constructing and maintaining the equipment.
A Story of External Factors
As alluded to earlier in this article, a major consideration with carbon capture is the efficiency of the process. If we consider that CCU as proposed here relies on a rich source of CO2, as well as a pure source of hydrogen, it would appear that the former would have to come from flue gas and similar waste streams from the fossil fuel industry. For the latter, things are more problematic if we wish to not create additional waste.
Currently virtually all hydrogen on the market is produced through steam reforming (SMR) of natural gas. This makes it not carbon-neutral: if it requires natural gas as input for SMR to create the hydrogen needed to create the SNG, one may as well directly use the natural gas.
This is further illuminated by Howarth et al., whose recent study details the effectiveness of so-called ‘blue hydrogen’, which uses CCS with SMR of NG and came to the conclusion that it’s more effective to just burn the NG directly. This leaves then only so-called ‘green’ hydrogen as viable input for this SNG process to conceivably make it carbon-neutral.
In 2020, only 4% of worldwide hydrogen was produced via electrolysis, some of it from low-carbon power sources. Even if all hydrogen at these SNG production facilities came from electrolysis facilities powered by VRE or nuclear power, that would still leave the CO2 as an issue. If this came from fossil fuels, then it merely postpones the moment this carbon enters the atmosphere to when the SNG is burned.
True carbon-neutral fuel is conceivable, but so far no viable source of carbon has been found. Carbon from flue gas costs about $7.5 per ton, extracting carbon from sea water as carbonic acid would cost about $50 per ton and direct air carbon capture between $94 and $232 per ton. This then leaves PNNL’s process primarily as a way to use the carbon from fossil fuels (coal or NG) twice, though at a considerable energy investment.
No Free Lunch
In light of these considerations and also based on PNNL’s own press release, it would seem clear that the ability to generate methane using this method is mostly transitional, to support the transition to low-carbon ways to power the modern world. The only likely exception to this is probably extra-terrestrial exploration, where in-situ resource utilization (ISRU) is likely to become a big thing.
One of the reason why SpaceX’s Mars-bound Raptor rocket engines are methane-fueled is due to the relative ease with which methane can be produced even on locations like the surface of Mars. When the nearest source of terrestrial methane is suddenly a planet away, the electrical and others costs of even DACC and electrolysis of water to slowly create a trickle of methane fuel for the trip back or to sustain a colony do not seem as outrageous any more.
One major benefit of water-lean solvents like 2-EEMPA is also likely to be the more efficient capturing of CO2 at fossil fuel plants. Whether this is enough to make big players like MHI and Shell sit up and pay attention is still anyone’s guess, but it’s hard to deny the benefit of more efficient CCS at fossil fuel plants.














while just a simulation, and involving nanofab techniques that likely do not scale (we need a breakthrough in this field!!!), this would be really good.
https://www.nature.com/articles/s41467-021-25426-5
I actually thought this is what the article would be about, and wondered if you’d cover the limitations.
I thought it said anime-based solvent. I need more coffee.
Same here, I expected to see Chim-Chim and Spridle making methane ala Speed Racer comedy routine.
Me too… nurse, more caffiene!
We are probably just better off using the CO² as energy storage.
Not such a bad idea. From memory CO2 needs about 800psi to liquify at room temperature. It is used as a refrigerant in some loops. In a closed loop system, CO2 could be very interesting to use as a working fluid in a power generation system. Even dry ice could be the state used to store it.
Not to mention compressed gas can be spun and used to pump heat, it can drive motors, in it’s liquid state it might be interesting to use it to propel a car or motorbike, maybe even a rain. It could be used to generate electricity in remote areas. Maybe we could reuse the existing natural gas pipelines to transfer compressed CO².
Don’t think that would work well – compressed gas over long distances for power transfer isn’t very efficient, and I don’t think the gas pipelines could take liquefied CO2, so its expansion at source provides more power for the same volume pumped…
I am however no expert on how natural gas distribution is done, or has been done on a grid scale… SO maybe pumping liquid CO2 is quite viable.. (seems like a lot of CO2 volume though)
It also takes a lot of energy to compress a large volume to 800 psi. I also hate the term “carbon capture” when what you are really saying is “carbon dioxide capture”.
If they combined it with an efficient way to convert H2O +CO2 to a hydrocarbon and Oxygen, then that could close the cycle better. It’s a pity that Chlorophyll isn’t easy to produce/use since that is basically what it does (ultra simplified).
It’d still require a lot of sunlight, but everything to split hydrogen from water requires lots of input energy.
The cheapest way for capturing carbon is to plant trees. And the byproducts can be used for construction, effectively storing the carbon in the most sustainable way.
Thank you.
Step 1 – grow something
Step 2 – don’t burn it, bury it
. . . That’s pretty much it. . .
I was thinking sinking the things you grow in the sea, preferably near a subduction zone, would do the trick.
You could grow algae at sea, compress it and weigh it down. We need a subduction zone near where there’s a natural upwelling of nutrients.
You can grow kelp on strings attached to buoys over deep waters, then once the kelp matures, it can be cut from the buoys and sink to the sea bottom to die. I’ve seen a few diagrams of people proposing this, not sure if it has been done though, or at any sort of scale.
See https://www.statista.com/statistics/1102545/tonnage-sold-for-demolition-worldwide/
There are on average 20M tonnes of ships scrapped each year. Ship scrapping is (I have heard) incredibly dirty and polluting and the ship owners have to _pay_ to have them scrapped (if doing it legally).
So instead, some of the larger ones could be strung together in the sea, with said kelp-growing ropes in between. You could also grow trees etc on the decks.
You’d need some desalination plant for fresh water, solar-powered and/or wind powered.
Probably a better choice would be a doldrum area where floating plastics accumulate naturally, you could then harvest these too and use them to make the kelp ropes.
The kelp would attract marine life and a nice fishy ecosystem, which could also be harvested, frozen and exported.
Quadruple-win (so far).
While not scrapping the floating ships, some of their contents could be salvaged to provide the ballast needed to sink the kelp parcels and tree trunks. This would also make the hulls more buoyant so they could take further weight of soil, trees and other infrastructure.
I think there is already a nice system of carbon credits we can tap to get some payments from the rest of the world. With a big enough raft of interlinked ships (say a few thousand square miles) this could make quite an impact. It might even generate its own microclimate – think cloud forest.
Bio decay produces a lot of methane which at sea depths produces fragile hydrates which when disturbed release methane back into the atmosphere.
That’s a good point, but these methane clathrates already exist in sediments all around the world.
What’s the big deal about adding to them? Yes they may be a little unstable but all I’m suggesting is accelerating the existing sedimentary accumulation of carbon.
My main concern is how much we can grow in a given sea surface area. There’s a new project off the coast of Scotland to farm kelp, I’ll see if I can get information from that about typical growth rates.
It has a tendency to breakdown if you bury it. But if carbonised, pretty much nothing can eat it. So perhaps bury low quality charcoal.
This might consume some energy but you gain the volatiles, some of which can be used as fuel. Further, if mixed into fields, may well have some positive effect on plant growth. Perhaps better than storing it in an easy to get back way as it’s more permeant if it’s widely dispersed and too costly to mine.
Unfortunatly growing something and burying it wont work, anything that is grown does capture carbon , However that carbon is still in the current ‘carbon loop’ because buried vegetation decays releasing the same amount of carbon as if we simply burned it ! not a long term solution and most growing things capture a little carbon over a long timefor a little time out of the enviroment, until cut or harvested then released slow or fast (buried or burned) So not a solution to a long term problem.
Humans have in the span of 150 years released from fossil fuels carbon that had been stored and acumulated over 150 MILLION YEARS, rasing the carbon compounds in our atmosphere to prehistoric levels (See where the fossill fuels came from!) In order to halt and maybe reverse global enviromental changes we must capture existing atmospheric co and co2, captured co2 must then be chemically locked into no biodegradeable, non soluable stable forms such that it is removed from and stays out of the current carbon cycle in the enviroment say calcium carbonate, like limestone. Taking captured co2 and turning it into methane is a crackpot idea! methane is worse as a greenhouse gas than co or co2 ! methane burned releases the co2 back to the air just moving the problem from power plant to other users NOT A SOLUTION, and thats before we add in the effects of the process to make co2 into methane heating, compressing , building the infrastucture to do this, the transport of the reactants , the transport and storage of the final product all will generate more carbon!! This doesnt count the release of methane after production via, leaks, incidental release when transfering it seepage , preignition release during use etc. NOT A SOLUTION if anything the plan as laid out would amplify the problem, which is global warming.
In order to prevent enviromental change to the point humans cant live here anymore we must stop using any fossil fuels whether synthetic or natural AND capture and sequester atmospheric co2 until the level returns to around the levels found pre industrial revolution.
Depends on where you bury it – in the right places it won’t go anywhere for millennia, and even in other places where it will degrade you have taken x years of that plant growing out of the cycle much of it for likely decades longer than it grew for – and because in its place you planted a new carbon capture planty object its a continued stream of putting more into the ground and the current plants than is being re-released – it tends in the right direction…
Taking Co2 and making methane from excess renewable electricity should be good for the greenhouse gas emission overall – you actively draw some from the atmosphere while reducing the demand for fresh oil products (which generally just burn off the methane as its too impossible to ship compared to crude) – and because you made it near the demand you don’t have all that shipping on it too, where generally fossil fuel travels miles before it even hits the refinery to be made useful, and then travels on again through who knows how many resellers before it hits the end user…
It certainly isn’t a magic save all, but synthetic fuels could be very useful during the transitional period towards a carbon neutral/negative globe, and has a place afterwards too – its a viable long term reserve of energy, and a good energy dense fuel for when you must ship energy somewhere – you don’t want to have to truck/fly huge batteries to the arse end of nowhere when power is needed there, and then have to ship the full weight back again…
and/or plant Kelp.
(Getting on SOAP BOX)
As one who grew up in spending a lot of time in Salish Sea an on the Oregon Coast with family in both the Fishing and Timber industries I get really annoyed when people only focus on land based reforestation an ignore water based reforestation. We need both and I think water could be argued to be more important as there is more water surface area.
(Getting off SOAP BOX)
The thing with oceans is vast as they are much of it is and always was pretty lifeless – the really green bits of ocean are much much smaller than the whole you think of – which is not to say I disagree people tend to ignore the oceans too much, just keep in mind much of it is effectively a desert…
see above – slowly rusting ship hulls provide your missing iron
Desert isnt the word I would use for something involved in producing at least 50% of all the oxygen we breathe.
Oh FFS even more uneducated tripe, please please get some oceanographic studies in biochem under yer belt :P
Pray tell, What utter dimwit numbskull told you this crap ??? Which website, did you bother checking anything ?
Unlike plants on land where nutrients in soil, in the sea all nutrients sufficient for plant growth in the sea as dissolved minerals, proteins, amino acids etc it’s a massive (borderline) grex like Soup in high state of dynamism – so called crystal clear waters replete with microbes !
So the evolutionary adaptation to tether into sediment as per land ie soil for plants far less important. In any case it’s a complex equilibria constantly shifting, what this means is a large proportion of prokaryotes which produce oxygen get their CO2 from that dissolved gas in the seas And due to far less need to tether roots into sediments meaning there are heaps of plants not needing to agglomerate anywhere near as much into floating beds of seaweeds though many species do historical survival niche, as I write it’s a widespread shifting equilibria and across millennia too heaps of oxygen even in “clear” water.
So “Foldi-One” your ambit claim of seas as a “desert” way off the mark, seaweed and other less visible type cells all over the place, as are bacteria some of which include cyanobacteria (many in Atlantic fuming vents as well and others) and in mineral ore voids and which produce oxygen too, plus trillions upon trillions of phage in each cubic metre of seawater as feedstock for “intermediate” micro organisms.
Ie. Grab a litre of sea water, place in a suitable recepticle, centrifuge for appropriate period and get about 20 billion phage & amino acids, do your PCR and get heaps of stuff – fodder for plants and many bacteria too, as I write it’s a huge dynamic, seas produce massive amounts of oxygen – even in areas if you don’t notice anything there, microbes are too small to see but, still there doing their chemical thing All The Time !
I did say the ‘really green bits’ are smaller and the rest is effectively a desert – as it is, deserts are generally full of life too but bugger all life compared to the really fertile populous land biomes!!!!
I never said the whole damn thing is dead, that is wrong, just that the really really full of life areas of the oceans are much much smaller than the whole – its huge, its complex, but there are vast areas of ocean that are very very much more lifeless than the rest!
I’m always wondering about the system-wide efficiency of this: like having to use gallons of water to wash the last of the peanut butter out of the jar, what is the carbon cost of manufacturing the solvent, providing the hydrogen, and running the plant?
Hmm, what a timely surprise, my eldest son just got PhD in Chem eng from Curtin University, Western Australia few weeks ago on CO2 & amines though not in the thermal based process instead electrochem. Tomorrow he’s flying off to MIT from Perth for a post doc position for 12 months, in lead up to northern hemisphere winter too, yikes whilst we are going to sweat worse in looming summer.
Interesting post, thanks. Good to see more effort into chemical engineering especially so regarding global CO2 problems and importance of pollution management, so many issues from excess CO2 since our human and plant adaptation to about 270ppm for many millennia now causing all sorts of perturbations as it’s risen in very short period and still on the rise.
Research of this type interests me for this reason:
I am of the opinion that as flashy as they look…as “futuristic” as they seem, electric cars–for anything but limited fleet applications–are really just a bad joke. Worse, having them rammed down our throats by idiot politicians (who have no engineering background, but a desperate desire to be seen as being “green” ) is a recipe for economic disaster.
Assuming breakthrough developments in battery technology (not something you can guarantee), significant expansion of the electric grid (not likely), and breakthrough development in primary energy sources (still waiting on YOU, fusion!), there is one physics problem you will never circumvent: How do you rapidly transfer huge joule amounts of energy from a charger to a vehicle in a period comparable to an existing “gas stop?” This requires either very large voltages (problematic from the standpoint of insulation and consumer safety) very large currents (problematic from the standpoint of I^2R losses and heat), or both. (I would add that an entire U.S. fleet of automobiles, dependent upon our grid, will result in endless rolling blackouts and utility prices that will make you look back fondly a $5 gallon gas.)
OR… we could simply accept that liquid fuels ARE the best way to transfer large amounts of energy quickly, and focus on fine-tuning HOW we use them to minimize potential planetary impact.
If through chemistry–whether direct synthesis or through harnessing alcohol or biofuel production–we produce our fuel with captured carbon, then burning that fuel, even in a 60’s era muscle car, is a zero carbon footprint proposition.
This allows us to to utilize our existing fuel delivery infrastructure (even if it requires minor modification), so we don’t have to reinvent the wheel.
Ultimately the best automobile of all, in my opinion, will be one with an electric drive train, fuel cell, and fuel tank. A car like that will be quiet, durable, reliable, truly emission free, and “rechargeable” in minutes, allowing for the same performance and cross-country capability as any existing gasoline-powered vehicle.
You are forgetting one key aspect of how most personal cars are used: The fact that they sit around with no one in them for 90% of the time.
The gas tank and battery are not equivalent in how they are used. With a gas tank, you drive around until it is empty, then you fill up. With an electric car, you come home every night and fill it up to 100% while you are sleeping. This means starting the day off with a full tank.
This means every morning you have the ability to go the full 400+ KM until you need to recharge. This works out great for the majority of commuters and drivers.
Having said that, there are some caveats that exist for drivers that don’t have access to recharge daily at home and at work, and for those that travel long distances (eg: road trips, long-haul trucking, or some sales people) where they’ll have to go out of their way to charge elsewhere, however for the large majority of people these things either don’t apply, or they can live with the downsides.
Agreed, Battery (or even Fuel Cell) EV won’t be the right fit for or deployable to everyone, but for most people they are a superb and cheap (to run) mode of personal transport..
And very green as long the worn out battery do get recycled and new/refurbished packs are available to keep that otherwise quite new car on the road in 10 years time…
Cheap – maybe not when the electrical grid becomes a bottleneck. Green – I don’t think battery recycling for lithium ion is what I would call green as of yet. Green – depends where that electricity comes from doesn’t it. By the way what is the resale value of a 10 year old electric vehicle?
On more thing. Look into the production of lithium and cobalt and then tell me more about “green” energy. It will take a major breakthrough in battery technology to make this anywhere near green.
Hint – it can’t involve mining for lithium and rare earth metals. If I use a form of energy (wind, solar, fossil fuel, nuclear, hydro) to create electricity that I then store in a battery that I use to power my car, am I not losing at every step of the conversion process?
Also creating the amines used in this process has a cost and environmental impact as well. Kind of like DEF used in diesels. Is the impact of the DEF (in all those great plastic containers, inside cardboard boxes) really less than the carbon particulates they reduce? I kind of doubt it.
Would love to hear how good we can get with recycling used fossil fuels… oh right, this article points out how freaking hard and “not green” it is. In most cases, the waste products are just dumped into the atmosphere. But let’s dismiss batteries because you wouldn’t call recycling them green… though they CAN be recycled!
And sure, the mining/drilling, transport, storage of fossil fuels is environmentally destructive (see recent CA coast oil spill for an example), not to mention that every single gallon/pound of fuel extracted must be replaced once it has been used, but let’s dismiss the environmentally problematic mining of lithium and cobalt even though every pound of these minerals that makes it into batteries can be used and reused and reused for 10 or more years, then recycled (whether “green” or not) for use in batteries for another 10 or more years.
The “well, batteries are not green either” argument is very weak.
Regarding losing at each step of the electrical conversion process… please tell me how efficient the process is of drilling for x gallons of oil, transporting those gallons half way around the world, refining them, then transporting them to a filling station for you to put into your tank and for you to burn those gallons of fuel until they are completely gone… only for the whole process to be required again for the next batch. I hope you’re not trying to argue that a power plant such as wind, solar, nuclear, hydro generating electricity which is sent to a home over wires and then used to charge a battery is less efficient than all that? Sure, fossil fuel powered plants inherit
the fossil fuel inefficiencies, but the amazing thing is if the fossil fueled plant is replaced with a non-fossil-fueled one, then every single EV consuming the electrical power instantly becomes more efficient (and more environmentally friendly!).
The grid need never become a bottle neck – for any car plugged in at home every night it can charge at such a low constant draw and still be full for the morning commute the grid wouldn’t even notice if every car charger in a city turned on at once… Its only the relative handful of users jumping onto the fast chargers and demanding that fast charge experince that can potentially pose problems, and putting in infrastructure for them isn’t that bad either…
Resale of a 10 year old EV is a tricky one, I expect it will be pretty reasonable once more of the current EV generations start getting that old so the second hand market starts to really exist. But there really isn’t a great way to tell till it happens, as guessing what people will actually be willing to pay/ the demand level that far in the future is all pure guesswork really…
It also really doesn’t matter where the electricity comes from for it being green, even from dirty coal an EV works out greener than a similar ICE – the EV’s onboard efficiency is so good, the grid is very efficient at distributing the energy and big dirty coal power stations are much better at cleaning their exhaust than any mobile platform… Where your petrol power has a huge amount of extra inefficiency driving fuel tankers around, and in the pretty woeful energy extraction efficiency so you have to ship around much more than double the amount of chemical potential you end up getting out…
Absolutely a greener source of energy makes it even better, but that is a rather hollow argument against EV.
You do have some point about the mining of lithium etc – but that is part of why I mentioned it needs recycling and refurbishment scaling up so its not all mining, though its not like Oil and coal extraction is at all clean either…
You are also correct you loose energy every step of the way, but you do that for every single form of energy transport – so what matters is the overall efficiency, and EV vs ICE the big number there is an EV is generally well over 80% efficient in using the energy its given, the absolute best ICE engines used in racing start to approach 50%, most cars are in the 20-30% range…. Then look at the efficiency of the infrastructure to provide that energy – all those fossil fuel extraction, and refinery steps we can call the same for both – assume the EV is entirely dirty fossil fuel powered just to drive home the point, but after that you are comparing massive trains shipping from refinery to power station (assuming they are not built close enough to just pump) and rather efficient electrical grid, able to fill your car at home (for many) to many hundreds of times more HGV fuel tankers, consuming vastly more of the fuel they transport than the train, and all the fuel the drivers have to burn getting to and from the petrol station!
Its absolutely no contest in use a Battery EV is much better efficiency wise no matter how much you try to stack the deck against it – the only thing that ICE clearly does better is constant long haul type jobs – and that isn’t because its more efficient, just because its not hugely inconvenient and expensive to build the infrastructure to park up HGV for hours all the time.. You can also argue ICE is the only viable option for those that can’t charge mostly at home or work – if your only method of charging will be the public fast charger you are for now better off with the 5 mins at the petrol station.
The only perhaps valid arguments against Battery EV for most users are the upfront cost (for now at least) and the longevity of the battery pack and its associated replacement cost – basically the one major maintenance cost – but as it stands it is a bit to early to really judge that to an ICE, as the ICE needs constant trickle maintenance costs in a way the EV doesn’t near as much, even the consumables like brake pads last much longer in an EV as so much of the braking is regenerative – so the overall maintenance costs might well end up lower on the EV even now, and almost certainly end up lower at some point in the future when more garages are doing the battery replacements as part of normal business and mass production of the refurbish/replacement batteries start ramping up.
> EV is generally well over 80% efficient in using the energy its given
Not when you count the embedded energy cost of the battery. That is also “energy it is given”. The 80% figure applies from the wall to the motor shaft at the ideal rate, and there are a lot more things to consider.
@Dude the embodied energy of a new car is huge no matter what, and your ICE engine needs constant flow of new energy into it to keep the moving parts working in a way the EV with its one major and basically maintenance free moving part does not – so even if you start counting all those costs its not significantly unbalanced between the two, the cost is just paid more upfront with an Battery EV.
I’d suggest the embodied energy of an ICE when you really take all the stages of production is infact vastly worse – sure its mostly basic steel and Aluminium metals, easy to recycle etc, but the amount of energy required to refine them to the grade required for each job is significant, then you have the massive amount of energy going into the foundry castings, and machining on those castings, then remember most of the fiddly little mechanical bits are probably not machined anywhere near the engine block foundry/machine shop so all the bits get shipped around who knows how many times before becoming an actual functional engine… Note I’m not saying lithium battery production doesn’t have similar problems, just that nobody ever really considers the full cost of the more established ICE, as those bits we buy in, their energy cost isn’t in our budget etc…
> By the way what is the resale value of a 10 year old electric vehicle?
We don’t have many 10 year old electric vehicles, but a 2012 Tesla Model S is going for $30k for the base version (https://www.edmunds.com/tesla/model-s/2012/), they were $57,400 new in 2012. 2011 Nissan Leafs can be found for single digit thousands, but the 2011/2012 Leafs aren’t known for having great batteries and only did about 85 miles at best (vs the Telsa Model S base starting at 160). Still probably less than a 10 year old ICE car will have depreciated. Used EVs made in the last couple of years seem to have barely depreciated at all.
Foldi-One says: The grid need never become a bottle neck…. ”
Nonsense.
Total gallons of gasoline consumed in the US in 2020: 123.73 billion gallons.
Energy content in a single gallon of gasoline: 33.56 kwh/gal.
Multiply those those two together—If you want to eliminate all gasoline-powered vehicles and replace them with rechargeable electrics of comparable capability, that’s basically how much additional electrical generation capacity you’d need.
I get 4.15 E 12 kwh
Oh! But electric vehicles are more efficient, right! OK, let’s be generous and presume for the sake of argument that they are TEN times more efficient than gasoline-powered cars (accounting for system-wide losses, they’re not, but I’ll give this to you for the sake of this argument). You then need this much electricity:
4.15 E 11 kwh
Reality check: The total electrical energy production for the US grid in 2020 was 4 E 9 kwh
Conclusion: In order to implement electric-car-Utopia and eliminate all gasoline-powered vehicles, we must first increase the existing grid’s power generation and delivery capability by TWO ORDERS OF MAGNITUDE.
This… mind you, in a time when you can’t build a new hydro plant (bad for the environment), you can’t build a nuke plant (bad for environment), and no amount of windmills (which kill birds and are insufferable to live next to, due to infra-sonic noise) or solar panels will make up the difference.
(By the way, where will we buy our solar panels from? The CCP?– with whom we are about to go to war with over Taiwan?)
When mandates cause sufficient electric vehicles to come on-line to consume what little reserve capacity our power grid has remaining, what will follow are nation-wide rolling blackouts. Many people will no longer be able to afford to light, heat or cool their homes. Expect that reports of elderly retirees freezing to death in Michigan winters or dying of heat exhaustion in Phoenix summers will become commonplace.
The price of all consumer goods produced in the United States—including food and water–will skyrocket.
Excessive energy costs will also work to drive out what’s left of our domestic manufacturing capability, to boot, so a good many of us will no longer have a paycheck.
Yes, that’s what happens in the 21st century when you mess with the energy supply.
Observer’s numbers don’t seem to match the official statistics: https://www.eia.gov/energyexplained/us-energy-facts/
Namely, electric power consumption already outstrips the power consumption used for transportation. Transportation is at best 30% efficient, power generation using fossil fuels is about 50% efficient, so the ratio doesn’t change all that much even accounting for transmission losses, so I don’t think Observer’s argument is correct.
I have no doubt the grid needs to grow and change to accommodate altered loads due to electric vehicles, but the situation is not so dire as requiring two orders of magnitude worth of investment. It’s barely a doubling by these numbers. No small feat either, but doable given the will.
@Observer, very sure your numbers are flat out wrong, but even if you take them as gospel its not a big deal – as the world transitions more to electric because its convenient and efficient the supply goes up, and the efficiency of the supply goes up. Yes as it stands right now most nations have nowhere near the reliable electric supply to replace all their fossil fuel uses if everything changed tomorrow (though IMO not everything needs to), but adding more capacity isn’t actually hard, and many folks will add in their own personal generation capacity as they can anyway…
Buy your solar tech from Germany then, or make them yourselves (I know you used to make rather good solar panels in the USA, I assume you still do. Germany definitely does)…
Also you fail to consider just how much of your fossil fuel consumption is moving fossil fuels around to get to the end user – include that and the vastly more efficient transfer of electric over distances is a clear winner – cutting down the total electric generation required to supply EV’s hugely.
You also have to remember that the stated energy value in fossil fuels is nearly irrelevant, in the case of road transport you are somewhat lucky to get 20% of that energy out on the road, the very very best ICE racing cars push 50% (in the ideal predictable conditions of a racetrack), so take something like 1/5 or 1/4 of your total stated energy consumed as petrol as the actual target the grid needs for EV’s – being very very much more efficient energy in to motive power out you don’t need as much electrical potential oversupply…
And even right now there is no need for rolling blackouts – you don’t try to draw max super fast charge in your cars all the damn time, and can as they have smarts in the charger control the demand to supply to some extent. As long you have all the cars connected monitoring the current the price of electric so they only charge faster when supply outstrips demand etc it becomes quite manageable – you can even sell your battery energy back to the grid when demand is too high. SO all the grid has to be able to do is supply enough on average overall – and it doesn’t even really need to do that, as most EV owners will probably fit solar to their and do most of their driving for free, with excess probably going back to the grid!
>This works out great for the majority of commuters and drivers.
Do the great majority of drivers own a home with a driveway/garage where they may install a level 2 charger?
Charging up a 400 km battery out of a regular 120 Volt socket takes about 60 hours, and even a 230 Volt 16 amp socket would keep you waiting for well over 24 hours at the fastest. In practice, the 16 Amp rating is not for continuous use and 10 Amps would be closer to reality, pushing the charging time up to two days. The effective rate at which you can charge is about 100 km overnight.
In a two car family having one hybrid and one full electric would cover any long distance travel. Even so if I arrive with no range left in the EV and spend a weekend somewhere I can still charge at my destination with a higher level charger for the return trip.
If you really need to charge up 400KM worth of battery drain very often at all I’d like to know what your job is… And off a wall socket even at puny American voltages you should be able to easily top off your normal daily drive worth overnight – 400KM is what anywhere from 5-10 hours driving at legal sort of speeds, that is one heck of alot of driving to do the day before, and then expect to do again the next day… Also a standard UK domestic socket would fill most EV battery capacities from absolute empty to over 70% over night – wild range of battery capacity options though so could be more or less… The point stands however, you are charging for hours, even the slow rate of fill is quite effective, while you slept your car by magic got back a good deal of mileage in a way that should cost you almost nothing – bitching its not a full tank coming back every night off the normal domestic socket is just daft, your ICE makes you drive out of your way every single time it needs a top up, and many of them don’t have ranges much different to the bigger EV, sure the fill up itself is faster, but you drove 15 mins out of your way every single time you needed it – the EV generally wont make you… Also being stuck at domestic socket charge speeds would I suggest speak to your stupidity as fitting a proper charger isn’t bank breaking, or misfortune to be stuck at the In-laws/Aunt/Sibling/etc who don’t have one…
And should you really, really need to grab the small amount of charge you are still missing for your next long haul you stop in at a fast charge at the start of your day for a ‘splash and dash’ style charge, or take a tea break at the charger en-route, you are driving for hours you are going to want to stop…
The only valid point you make is how many drivers have a space to park that they can charge at, which is a valid reason not to have an EV, however I just heard in London and Portsmouth lampposts are turning into chargers, apparently quite good fast ones (friend has been driving only EV/Hybrid for a long time now) – so the folks living on those roads don’t need a driveway, just a spot they can park at all (which in the UK in some areas isn’t at all a given I grant you)..
Also notice:
>With a gas tank, you drive around until it is empty, then you fill up.
>With an electric car, you come home every night
The “you come home every night” is the limiting factor. There was a recent study about EV adoption in California, and out of the people investigated about 20% had switched back to gasoline. The common factor with those who switched back were: lower income, dependent on a single vehicle for transportation.
Almost everyone needs to drive long distances and away from home/work sometimes, or have unplanned trips just when they’re low on fuel. If the EV serves you 99% of the time, that’s still 3-4 days a year it doesn’t, and it’s usually those times you would need it the most.
And you have to remember that EV ownership is still an upper/middle class hobby, and California has high subsidies for EVs and high taxes/prices on gasoline skewing the game even further. The 20% that gave up their EVs weren’t poor, just less wealthy. After all, they could finance a brand new car in the first place – something which 70% of the population find infeasible. Most people have to take a loan to buy a decent low mileage second hand vehicle.
The savings on fuel and maintenance that EVs are sold for are a total red herring when you pay 10-20k more for a car than you would for a regular economobile.
When somebody makes an econo EV its fair to compare upfront price to a ‘regular’ one.. For now they are all pretty much luxury end of the market, so have to be compared to the Merc’s BMW etc, where price wise they don’t fair nearly so badly, maybe even work out cheaper to buy.
If you need to go that extra distance 4 days a year you can hire a bloody normal car, takes no time at all, certainly no time compared to the mammoth drives you are obviously about to do if your EV can’t do it… It will also cost you pittance, more than made up for by the cheaper running costs in the rest of your motoring that year (assuming you actually do any real mileage at all), especially in places where fuel isn’t stupidly cheap the way it seems to be in the USA…
I do agree for now at least EV’s are not for the poor – but then no new car is for poorer in society, even most middle class are probably in the year or two old second hand market more than new… So some of them will probably end up in EV’s when the current crop of EV start being something around as old as their current second hand cars, though if EV’s will depreciate quite so fast is a question to which we don’t yet have an answer, it might take somewhat longer, but eventually cheap ones will arrive.
>When somebody makes an econo EV its fair to compare upfront price to a ‘regular’ one..
Dacia is supposed to come up with a cheap EV in the 15k£ range, at least it was couple years ago. Haven’t checked what came of it. However, the battery was tiny.
If you compare apples to apples, you can take Nissan Leaf vs. Nissan Tiida or whatever your local name for the model: same car, one made in electric the other with a petrol engine. The Leaf is 10k more expensive.
>If you need to go that extra distance 4 days a year you can hire a bloody normal car,
I can, but I own a car so I shouldn’t need to. It’s difficult to get a rental quickly.
Think of a scenario like, you’re driving home from work and your battery is going at 20%. Then you notice you left your wallet at the office and you have to turn back. Sorry, no-can-do.
The difference in price buys a LOT of fuel, and there are some very cheap cars out there if you simply need a car. Skoda, Dacia, Kia, Fiat… You can buy a Peugeot 108 for £12,785 or a Dacia at less than £10k. The cheapest EV I can find is a Smart EQ Fortwo at £20k and it’s not even a real car. If you want all four seats, A Renault Zoe costs you £29k.
People who talk about EVs mostly compare them to Audis, BMWs, Volkswagens, Volvos, etc. because anyone who really considers owning one will be squarely middle class. They aren’t even aware that there are cars at half the price.
> especially in places where fuel isn’t stupidly cheap
Even in the EU, 10k worth of fuel gets you about 150 000 km of driving, which is about the life of the EV anyhow. Ten years on the road and the battery is done. That, plus the amount you lose in faster depreciation of an EV on the second hand market.
The dirt cheap cars turn up once the tech is established and they can buy in the massively mass produced units to make it possible to build one that cheap… Getting there but not quite there yet. So you compare them to the more middle-high end of the market because that is what they are feature wise, they are not the tiny cheaply built junk with only the minimum equipment…
You also have to ask what are you talking about on life of the battery – some cars Like the early Nissan Leaf are really great at killing batteries, but most current generation ones are expecting vastly better battery lifespan, and showing it to be pretty beliveable – generally remaining at very high battery health after the year/milage they have on the road so far…
Also got to ask who on earth works so far away from home and drives?!?!?! Most driving commutes seem to be something like 40 miles, even the smallest battery EV do that easily… If you really must commute 100+miles and can’t afford the long range (so generally more premium) model EV, guess what an EV isn’t for you, yet…
Having a car at all used to be an upper class hobby. Now not so much.
Factor in sub arctic temperatures and you get even lousier ranges and longer charge times. You do not want to run out of charge in 20 below freezing. EV are jokes when it becomes cold. Only viable option is fuel cells.
For long haul trucks there should be an industry standard agreed upon so battery packs can be replaced. Instead filling up with diesel the whole pack gets changed out and then recharged for the next truck.
If motorcycle manufacturers can agree on a standard battery other companies can as well. Pretty soon electric dirt bikes will take over just as 4 strokes replaced 2 strokes. Especially for long off road races like Hare Scrambles and enduros if a pack lasts 30, 40 miles or so it would be plenty to make a couple laps and swap at pit stop.
With long haul trucks some of the battery capacity could be in the trailers themselves. Some trailers sit at a loading dock for days, it could be charging during that time and add extra mileage. Even put a motor in the trailer and you wouldn’t need a yard dog, the trailer can move itself and also add power when connected.
Of course some won’t want to share a pack with other companies, But look at a lot of semis they are basically the same parts so theres already some sharing of components.
Yeah, Elon Musk is one of those idiots shoving Teslas up your .. throat. Real shaky engineering, I hear. Strange it’s not an economic disaster. I heard the same arguments about how uneconomical solar energy was, for a decade, while I noticed that solar powered everything was showing up in infrastructure, because of actual economics.
That is to say, I believe that your characterization is off-target when you assert that the grid is unable to sustain a high enough power rate for the energy delivery required to equal the power and energy characteristics of a gas station stop. The grid does not have to deliver that much energy that quickly. The only rate that the grid has to deliver at is the same one that the energy is used at. You fill up your car once a week, on average. The grid only has to deliver power a little better than that, on average.
“Rechargeable in minutes” is a niche requirement. The majority of car usage was for commuting locally, under 30 miles a day. Overnight or at-work recharge is fine for that, but already a Tesla can gain 200 miles in range in 15 minutes at a supercharger station.
And things are changing, fast. A lot of people might never sit in an office again, so they’re not commuting. Distributed power generation is perfect for the way the electrical grid is laid out, although it is going to have some distributed intelligence to match the distributed generation in order to keep the grid stable. The markets will decide and optimize in our busy, hot world.
The actual problem, by the way, is not the energy refill rate, but the energy usage rate. One of my vehicles burns 30 pounds of gasoline an hour. That’s 200 kilowatts. I’m going to need a Mr. Fusion, mini nuclear reactor or micro blackhole to drive that thing around.
> there is one physics problem you will never circumvent: How do you rapidly transfer huge joule amounts of energy from a charger to a vehicle in a period comparable to an existing “gas stop?”
E=mc^2. Don’t move the energy, move the matter. In this case, you could swap batteries. This will require a kind of standardization on batteries we don’t yet have, but it’s possible.
To bo honest, I don’t we’ll go this way though. I think car ownership will become for collectors and we’ll instead use ride sharing with fleets of autonomous vehicles for most purposes. This is maybe 20 years from becoming viable, and it will probably take that long to deploy charging stations everywhere anyways. Then the recharge problem literally just goes away, as the fleet will need some degree of surplus capacity. The best engineering solutions are the ones you can define away.
Why do you need to? In general you really don’t need to at all! – As an electrical grid of some pretty significant power capacity exists nearly everywhere put some charge points (even rather slow ones) in and the EV can charge overnight/while you are shopping – with the range they can come with now you don’t need to really rapid charge at all – for nearly all users, and nearly all trips, you got to where you were going and while you were doing whatever you went there for the car magicked up heaps of miles to get you back without ever needing to dump a full tankful into it in 5 mins or more fairly 20mins – as remember you will often have to drive out of your way to find a petrol station, something you shouldn’t have to do most of the time with an EV as it filled itself while you were busy…
We burn hydrocarbons because converting a liquid with lots of covalent bonds into a gas with only a few covalent bonds releases lots of energy because of the vast increase in entropy. Anything we do to reduce the entropy of the result, like carbon capture, is going to be an energy sink, so it is inherently not going to be economic. Either we throw up our hands and resign ourselves to a future of flooding, forest fires, and mass migration as parts of the world become uninhabitable, or we address this the way we should by spending the money to deal with the mess we’re making. Solar, wind, hydro, nukes, and moving hydrocarbon combustion only to situations where the specific energy of chemical energy production is required. But waiting for or insisting on an economically competitive solution to carbon capture is only a way to abstract the problem into a different form that’s harder to argue against. It’s the same as our resistance to reprocessing nuclear waste: sure, it’s more expensive in the short term to the users, but it’s the right thing to do because the long term costs are intolerable.
Completely agree!
We already have “resign(ed) ourselves to a future of flooding, forest fires, and mass migration as parts of the world become uninhabitable”
We can’t stop that now, it’s way too late and already happening. We can only try to make it less bad for our children’s children.
“Either we throw up our hands and resign ourselves to a future of flooding, forest fires, and mass migration as parts of the world become uninhabitable” This is not our future, this is right now, this week.
Fusion might be the holy grail these days but it would be great if there was a green and efficient way of breaking the Carbon Oxygen bond in CO and CO2 leaving just free oxygen and particulate carbon. Unfortunately to break that bond would require a lot of unicorns and magic that is needed to make fusion work… 😁
Fusion powering generation local electroreduction makes sense, and the single step oxygen production’s lower than having intermediary CO. Nice research keeps being produced, not least the decoking step where carbon and carbonate are motivated to go off together in particulates whose condensation you encourage. That leaves the 🦄 to Fusion Energy for the most part…
That’s kinda the fundamental problem with carbon capture. If you want to turn it back into oxygen or some kind of fuel, you need to put energy in to make that happen.
The above process that makes methane probably gets a decent portion of this energy from the natural gas reformed to make input hydrogen. Then it’s really just the conversion of one kind of fuel (“dirty” hydrogen) to another (methane).
That leaves processes that sequester the CO2 without transforming it much. Would be nice if there was a magic concrete that absorbs CO2 instead of oxygen, for example.
I do think that artificial photosynthesis will someday supplement harnessing natural processes to allow us to create natural gas: CO₂ + 2H₂O + energy -> CH₄ + 2O₂
I think a potential carbon neutral natural gas based economy is a more likely way forward than having everything be electrical.
Thank you for that. Agreed.
I consider myself very environmentally conscious. But as an engineer I live and work in the real world which, in addition to dealing with physics, also has to respect economics.
As time goes on, electric car zealots sound more and more to me like priests in a “saving Gaia” cult, as opposed to people offering practical solutions to legitimate problems.
Creer du methane avec du CO2 est une heresie!!! Le methane est classé 100x plus toxique que le CO2 pour la couche d’ozone et le rechauffement climatique…
Moi asks a similar question I was going to, isn’t methane slip a key greenhouse gas contributor?
Exactly, if even only a small fraction of this methane leaks from the production, distribution on consuming system, then the process as a whole will easily become carbon positive.
Methane à cracking producee plus l’graphene, l’MXene, l’élastique compostage, intermédiaire a produit sérieux.
L’idée est d’utiliser le méthane comme source d’énergie.. pour le bruler en CO2. On crée ainsi un cycle en absorbant le CO2 que l’on peut rejeter dans l’air pour le reformer en méthane avec une source d’énergie propre (il y a toute polémique là dessus aussi si ça existe véritablement..).
En somme il n’y aura pas plus de CO2 dans l’air qu’actuellement (voir même avoir la possibilité de le réduire si on en pompe plus)
Mighty shame we can’t find a way to use a converter to reform CO2 into carbon and oxygen in the car’s exhaust….
https://www.science.org/content/article/liquid-metal-catalyst-turns-carbon-dioxide-coal
or even into Carbon Monoxide…
https://newatlas.com/materials/five-metal-alloy-2d-catalyst-carbon-dioxide/
No, the real shame is people are still driving polluting cars.
No way you get any trucking done on battery powered electrocity. Going to cost a pretty penny to refurbish the railroads to handle long hauls and use EV for short haul goods transportation. Maintaining this living standard will mean a lot of people driving with the cheapest cars and fuels that gets them from point A to B. Only way to cure that, presently, would be by inducing massive taxes on fuels, which in turn hits the poorest automobilers the most. That leaves you with a lot of people unemployed and relying on social security. The true shame is that most people breed like rabbits with no regards to the limited resources on this planet.
Look, I don’t care what the process is; there is no such thing as energy break-even UNLESS an outside source of non-carbon generating energy is used! Everything, solar, wind, hydro, nuclear, etc. takes ENERGY to create and that energy, at it’s base is CARBON based.
Whether it’s diesel to run the construction equipment, natural gas to run a power plant to make electricity to run the construction equipment and operate the plant, carbon energy to mine the materials to make those photovoltaic cells or what not!
Stop with the panacea nonsense.
Show me the EVIDENCE that this or ANY similar process uses LESS energy to create the fuel than producing it by conventional methods: i.e. drilling.
If you want to talk carbon reduction, FINE, but don’t even pretend it’s carbon neutral, there is no such thing in existence yet.
And I for one am done with the happy-happy joy-joy crowed that can’t look beyond what is right in front of them to see the REAL carbon footprint of these types of technologies.
Where the heck do you think that the chemicals used come from? Thin air? Please…. start addressing the REAL cost of carbon and quit wasting our time.
Yeah, you would source the energy from renewables. Pre-grid or microgrid. What a whiner supposing that agriculture and sources from Neu would be left out; strictly speaking perhaps we should look at subzero hanging fruit like making people truly howl with misery at property pricing, not being able to reach phone assistance at the IRS etc. then charging to get out of the way of making it worse in the same vein as fossil fuel industry does, but that is fraud of course. Don’t be satisfied with that looming in your Jevon’s Window of normalcy. 🥞
Nobody ever said anything about magicing up energy from nowhere, what this is is an improved carbon capture tech, that leads to carbon negative/neutral ways to use gas, far more environmentally friendly than drilling up crude half a world away, all that shipping and then the refining steps to get to the useful lighter fuel products. Not to mention many sites just burn the methane off as its too costly/inconvenient to store and ship compared to crude.
Yes it needs outside energy but the point is such systems can be used to create long term energy reserves, near/ already connected to the demand from the excesses of renewable energy when it spikes. And not all energy is carbon based at all – much of it is Hydrogen fusion based, via various levels of directness…
Some renewables already generate more energy in their lifetime than energy input taken to manufacture install and maintain them.
We are already there with hydro (long ago), and offshore wind power. Solar PV is just about there.
As said above, then use that unreliable energy to ‘lock up’ the energy in manufactured hydrocarbons, that are easy to store, and then use it at your convenience rather than when it’s windy, rainy or sunny.
OFFSHORE WIND POWER? That will never offset the carbon dioxide needed to construct and maintain it. Every time you need to do maintenance you need to take a boat to the unit, that has to have local power, that boat is manned by a crew that needs to drive to the location to load up the boat with size appropriate spare parts…etc. Then if it needs a crane crew to do maintenance the carbon footprint is off the charts.
Wind power is good for small projects, but unlike Hydro (where it’s really gravity we’re exploiting, using water as the working medium) it relies on a resource we can’t easily predict, and that the very tapping of that resource can change the climate in a more damaging way that anything we’re doing with a carbon footprint. If we en masse do wind power on this planet, and begin disrupting the weather patterns as a result, it’s a really bad option.
Nothing is without consequence. Battery technology with sufficient power mass is a chemical hodge-podge that will either burn without an easy extinguishing source to tame it, or poison the planet when the chemical bonds that we exploit are no longer useful, and that battery needs to be disposed of.
What’s the carbon footprint of the disposal process of the high density lithium batteries? If that substance poisons the soil and we have nothing to eat, I’m not going to have a care about the new green economy.
I’d like to see more nuclear with Gen IV reactors, using hydro power for peaks and storage. Then when we can generate electricity with clean energy like this, then we can discuss what we can do to capture CO2 from the atmosphere and bind it so it remains solid and not a gas. Since any process that will do this requires heat or electricity being able to generate these without adding to the problem is vital. Being able to do it when the world is dark and there’s no wind to speak of is important. Nuclear and on site hydro storage for peak use is the best way forward. Solar and wind have their place, but as a replacement for what we utilize on a day to day basis they cannot be seriously considered.
Another advantage of using hydro as a peak storage medium is we can discuss the mitigation of floods by tapping that rising water and moving it to place in the world where there are droughts. There’s plenty of water, we just need to move it around to where it’s needed. Seems simple, and it is. We just need to have the force of will to do such a project.
Follow the money. If we’re serious about solving this money won’t be the driving factor. But right now it’s a bunch of researchers and corporations looking to cash in on a research project that will yield an invention that makes them rich, not the concerned citizens of planet earth looking to do things cleaner and fix the problem. And all the world’s governments are happy to dole out money to research the issue and not actually FIX it.
Damn shame that.
Yes, offshore wind power, currently all-in it’s 11 grams of CO2 emission per kilowatt hour of electricity generated. That will come down as more materials can be produced by the energy generated, to be true zero carbon (actually negative carbon if you include the carbon-fibre blades and then bury them after their life is up).
You are nit-picking sir, I agree currently the maintenance by boat is diesel-driven, but in the future we’ll have electric boats and cranes, obvs powered by the very same wind farm.
Most weather is 1000ft up (or more). The turbines aren’t that high – yet.
Maya, this may not just be applicable on Mars.
Here on Earth, in a remote place maybe, about half the FG-to-SNG process with the IC3M technology flowsheet falls away. Let me explain.
I am looking at biogases recovery from a lake in Africa where is about 12.5 tcf of gas available, 20% methane and 80% CO2. I am planning to build plants to extract 80-90% methane stream and a 99% CO2 stream. We recover 87% of the methane and can recover about 60-70% of the CO2 on a continuous basis over 50 years. The biomethane is for power generation and pipeline gas to displace charcoal use as the principal fuel (saving the forests).
The CO2 has a number of potential uses, but this one is intriguing. There will be ~50ppm H2S, to be verified for the CO2 stream. The cost of the CO2 production is virtually zero. Some cost is incurred on piping it to shore. We estimate that one million tons per annum of CO2 is available per year, but this can be increased by 50-100%, possibly with more H2S.
So, with this process able to deliver cool (25C) gas at better concentration (~98%) with about 1% CH4, how does that look as a feed to a CO2-to-SNG process with IC3M technology? There will be a directly linked power plant and a gas distribution pipeline to take all the production. As a bonus there will be up to 400 MW (maybe more with the extra methane) power plant waste heat to provide the heating needs at temperatures above 250C.
The remaining item is a hydrogen source, possibly from steam reforming of methane. The question is, how does this methane-hydrogen-CO2-methane work in terms of mass and energy balance?
As a (once-was) chemical engineer, this looks to be potentially more viable, simpler and far cheaper than the flowsheet described above.
That’ s pretty great, and a good excuse to keep a sidebar in H2 back in energy production, plus of course a path to longer chain fixed products like soil amendment (without extra biochar steps maybe;) but getting large sheet carbonate MXene or graphene or also DWCNT would be good to yield also.
CCS / CCU is never going to work at the scale required to actually reduce PPM in atmosphere in a meaningful way.
https://youtu.be/MSZgoFyuHC8
Despite pointless overuse of the “F” word, and odd overdubbing, this video is hilarious.
You could just grow a field of beans…
When eaten, that would produce too much methane.
Beanz meanz fartz.
I agree with this sentiment, without fusion reactors we really don’t have a long term sustainable energy surplus and are just playing accounting games with where the energy actually came from and how much we are consuming with each aditional step in the supply chain being subject to the losses imposed by nature as defined by the laws of physics.
Hmm, suggest take a serious NPV in that work out land area across both Northern and Southern hemispheres for total global electricity demand at a reasonable solar panel efficiency – it’s much less than desert regions even without input from ocean or land based wind !
I read about 25 years by iirc Don Lancaster that all USA electricity demand could be supplied by 15% efficient (overall) solar panel source of land area of 50Kms by 50Kms of course spread efficiently over the country that wasn’t including wind.
As for storage, look up liquid metal batteries, cheap to make and intrinsically safe, can even ship by air or sea when cold as have No voltage so no fire risk. Gates is putting in a few million, I think MIT prof has co startup. Look up their 10ft shipping container get unit, modelling and great simplicity :-)
Although I’m in favour of fail safe thorium type fission reactors and fusion overall for high power density, it seems key analysis of land area in conjunction with existing solar power, wind and storage is realistically going to be more than enough for our local and likely global needs overall if pursued over even as little as ten years :-o)
As Saudi Arabia and couple of other regions around there have shown, cost of solar down to USD $ 0.04 per KWHr at wholesale as of 5 years ago, nothing to suggest it can’t go further down – other than propaganda from fossil fuel lobby – of course :-/
CO2 isn’t evil… All living things exist, because of carbon molecules. Plants are the only source of carbon in our diets, since they are the only living thing with the capacity to take carbon from the environment. In the form of CO2… We strip a lot of acreage of vegetation, to plant our solar and wind farms, in addition to the condos, parking lots, and strip-malls.
Plants do extremely well at higher CO2 levels. Most all indoor farming, augment CO2, to about 1200 ppm…
And it’s absolutely true that planting more surface of the earth would be a great way to stop global warming. But there are all these _humans_ around, at least for now.
Coexistance of farming and solar is an interesting problem at the moment, though. There are some pilot projects here in Germany playing around with dual-use. I’m no farmer, but apparently it’s trickier than you’d think.
CO2 isn’t evil but is deliberately wiping out the vast majority mammalian life on this planet something you would consider evil? How about just killing billions of people? Is that evil? Is murdering people that inconvenience you evil?
Harvey, you’re either a sociopath or an idiot.
Evil is a human concept. Has nothing to do with nature. Evolution dont give a rat´s arse about notions of good/evil.
Fusion power has been five years away for half a century now.
Interesting. Where I work is developing a similar process. Our aim is for better hydrogen transport. The idea is like this: 1. capture CO2 from the air [here]. 2. Use it with Green Hydrogen to make Methane [here]. Liquefy Methane for transport. 4. Use Methane where required – CO2 returned to atmosphere [there].
Liquifying Hydrogen takes a lot of energy – liquifying Methane should use less total energy even counting the energy needed for capturing atmospheric CO2.
What’s the point? We need more CO2 in the atmosphere, not less.
That’s absolute uneducated rubbish C check your basic physics & biochem !
More CO2 causes heaps of problems eg more IR retention, worse psychrometry re water vapour, protein plants produce cyanogenic glycosides, lignin grows faster less dense more wild fires, some crayfish change mat g patterns, thinner shells etc etc etc
Nothing useful re more. CO2 unless you only eat carbs without proteins ie worse nutrition !
Far better reduce to approach pre industrial levels – education so very important, look up spectroscopy, food chemistry, radiative transfer, statistical mechanics etc
Maybe they are Venusian – its the only way I can see more CO2 could possibly be considered a good thing…
If your plan is to cause a fullblown mass extinction event then yes, you are right.
If C’s going to play that card, I might not have to play my “moon crashes back into Earth” card…
Mass extinction due to slightly elevated CO2 levels? That’s a far-left conspiracy theory. Besides, CO2 capture often increases CO2 output.
“C” raises an interesting point, “we” and by that I mean the planet, has been here before….
CO2 levels in the past have been as high as 4000ppm (0.4%)
O2 levels have been as high as 30%
Currently we are living at a time that is “comfortable” for humans.
It seems likely now that we’ll engineer the biosphere to retain the status quo, more or less. So the planet can say goodbye to any further natural (and excessive) swings. Is this a good thing or a bad thing? Debate…
“Is deliberately causing a mass extinction event a good or a bad thing?
Debate…”
How are people like you this stupid?
Indeed. The notion that slightly raising the CO2 levels in the atmosphere will cause mass extinction while live on earth thrived when it was much, much higher is really dumb. What is the optimal level of CO2? No one will answer that question. Or they will change the topic and say that the rate of change is a bigger problem.
But that is the thing the rate of change is the problem – change slow enough for naturally slow processes of evolution and species migration to keep up isn’t the end of the world… But changes as fast are we pushing on the planet really could become such a mass extinction event easily – only so many times we can put a finger in the leaking dyke trying to keep the ecosystem functional before it (the dyke) collapses around us…
So the optimal level of CO2 in the atmosphere as far as the planet is concerned right now is something like the pre-industrial level, as that is around the level it should still be at had we not buggered with it so dramatically over such a short a time – its the level all the flora and fauna expect, keeps temperatures globally in the region that would be expected so doesn’t drive insane weather events that the ecosystems of the area can’t survive. Being somewhat higher isn’t a big deal, but being an ever increasing and very large amount higher over such a short time frame is way more than most species can adapt to – Oak trees for instance don’t reproduce at all untll they are around 40 years old, so the genetics that favour their survival in the changing climate can’t possibly be trended towards naturally in such a short time – that would be like asking you to breath 80% Carbon Monoxide because your grandparents had to breath 4%….
> The notion that slightly raising the CO2 levels in the atmosphere will cause mass extinction while live on earth thrived when it was much, much higher is really dumb
“Life” will survive. The question is what life is compatible with the new situation. Our food chain very likely will not be compatible since it will not have time to adapt, and that’s the problem people are actually worried about.
Gaseous hydrocarbons will not be used for sequestration, it’s too inefficient volume wise. We need solid or liquid hydrocarbons for sequestration. Also we can’t afford very much loss of materials to the air and bad recycling (and we are terrible at recycling). It doesn’t take a lot of loss to make things short term unsustainable once applied at trillion dollar scale. Suddenly “plentiful” materials can become limiting.
Methane will be burned, or temporarily “sequestered” to push out oil and be burned later.
Upfront I would like to excuse for the fact that I’m not a native English speaker.
It’s an interesting article.
I am an involved in these kind of projects in the role of inspection and assessment of the pressure equipment and assemblies. This in the role of inspector of an European Conformity Assessment Body as well as an national Assessment Body. And some research councils out of personal interests. (E.g. Supervision/inspection) regarding CE marking and periodic inspection of non-/critical equipment). This involves small scale to infrastructures..
There are several interesting developments and Inventions. Unfortunately some are not being exploited because the investor is to enxious to explain what they are doing.. and sometimes the smart professor/inventor is not capable to stand up to the inferior competitor who has a trained sales person or marketing team. (Technician vs marketeers)
Discussing the benefits and cons of a certain invention on a platform as this seems rather pointless since there is not a simple equilibrium which meets everyone’s interests. Therefore unfortunately it is/seems to involve marketeers/influencers even if a tech wants to sell tech to a tech….
It all depends upon at which stage in the fossil-fuel path you intervene. Take all the carbon out BEFORE it’s burned as fuel, and pollutes the atmosphere, then we have little problem. That would have been incredibly easy…but now?!?!
-=swoosh. This obsession with ‘necessary’, and very energy hungry, atmospheric carbon remission!?!?
Did you know, it’s child’s play to remove all carbon from natural gas, as a stable, solid carbon dust, yielding clean-burning hydrogen gas?
The energy requirement is less than 15% of that used in the most efficient water-splitting processes.
In terms of the energy required to remove carbon from the air, once ‘dirty’ gas has been burned as fuel, well, that energy investment is absolutely miniscule. It could easily be supplied by renewable means…no batteries required!!!
Yep, the petro companies have been sinking the concept, almost from the start.
https://en.wikipedia.org/wiki/Pyrolysis
Despair.
The only effective way to sequester carbon is to leave it in the ground in the first place; burning it only achieves oxygen sequestration when put back underground. There are only a very few pilot CO2 genie-back-in-the-bottle pilot plants in the world, subsidised exhorbitantly by taxpayer dollars, and _none_ of them demonstrate viability. The failed concept won’t
matter once the fossil burners are all closed down. c.f.
https://www.abc.net.au/news/2020-12-17/bluewaters-coal-fired-power-station-written-off-books/12990532
There won’t be a mass produced ICE car in 2030, and coal power plants will all be bankrupt, but CO2 -> cH4 ->
plastic or other durables would be good for minimising the looming climate hammer. The disruption will achieve verticality in this decade, and there will only be wreckage of ICE based businesses by the end of it.
To me this seems a remarkably over engineered solution when an alternative already exists (that avoids the caveats involving high temperatures and pressures) utilizing biology.
The cycle involved is rather simple: use plants to capture the carbon via photosynthesis (at which they are very efficient) and microbes to break down organic material to produce natural gas. Both of these are based on evolutionary processes with a long history and are very good at what they do.
The facility that breaks down organic matter into natural gas is called a digester, for the simple reason that it utilizes the same microbes we all carry inside. (Where do you think farts come from?) The feed in a digester can be just about any organic matter. It makes little sense to grow crops specifically for natural gas production as the feed could just as well be organic waste. A few examples that come to mind are:
* bales of hay gone bad work fine (although they are slow to break down), anyone with connections to horse farms or dairy farms will have seen that these are on ample supply.
* potato peels are excellent feed which will give quite a boost to the process, speeding digestion and thus gas production. Even water from washing potatoes can be run through a digester for similar effect.
* fish waste (the heads, bones and intestines left over after fish steaks have been separated out) is an excellent booster for natural gas production, just as potatoes mentioned above
* water purification plants produce a significant amount of organic matter that is separated out when clean water is produced. Many of these plants already produce natural gas from their organic waste.
* with the examples mentioned above and a little imagination, it’s not hard to see that organic waste byproducts are in ample supply from a whole host of industries
* wood, however, is antiseptic and thus harmful to the microbes involved and should not be used
The produced output is roughly 60% methane, about 40% CO2 and small traces of sulphurous oxides (the amounts of which depend on the feed in the digester). We have filters to separate the CO2 to be utilized in industry, and keep in mind that the methane and carbon dioxide produced here are captured from the atmosphere making the cycle fully carbon neutral. The sulphurous oxides can be damaging to internal combustion engines, which is why they are also important to filter out.
And none of this is new technology. I drive a bi-fuel car that can switch between methane or gasoline on the fly, and such vehicles have been common in Asia for decades. It’s great to see interest picking up on environmental concerns, but this article leaves an elephant in the room for technology that already solves the problem discussed here. I’d love to see natural gas adoption grow globally, and frankly I’m not convinced that jumping directly to electric is necessarily beneficial over the long run when considering the full life cycle of the car (including batteries and electronics) over compressed natural gas and ICE cars.
Just my two cents.
– Lupu
I should have used the word “biogas” instead of “natural gas”. The carbon neutral cycle is only valid for biogas produced from organic matter and does not apply to gas obtained from oil wells or from fracking.
The question with using nature to your own ends is one of scalability – only so much land area to go around, and only so much waste. I don’t disagree but its not an easy solution to use, especially in the tightly packed urban nations – its criminal we often are not making better use of some of the useful byproduct as you mention, but there is still only so much to go around.
If you can manage to create efficient vertical farms to grow useful product AND the waste organics end up being used usefully etc it starts to become more plausable to get enough. But so far Vertical farms are just not that good, and I’m not sure they ever would be for it – as you shrink land area covered for the same plant content you must have to add lighting, usually being indoors there is then the need for constant active irrigation – its good stuff, no doubt will be useful somewhere, but can’t see it being a good producer of waste organics – and if it is the other job its supposed to be doing – providing food/fibres it can’t be doing well..
I’m hoping for what I would consider a “seasonal battery” that can be scaled down to sizes and prices that work for single-family homes. What is a seasonal battery, you ask?
Something like the Tesla PowerWall would be a daily battery. When your solar array produces more than you consume during the day, it goes into the PowerWall. If sized correctly, that battery has enough storage to keep your home powered through the night until the sun is up and can charge the battery up for the next day. As long as the days are sunny enough, you don’t need to consume electricity from the grid at all.
A seasonal battery is the same, but at a yearly scale. If your solar array is generating more than what you consume most days during the summer months, the excess is stored in your seasonal battery. During the winter when the sun is out less and you’re using more than you produce each day, you draw from the seasonal battery. Size everything right, and you produce enough energy during the summer to power your home all winter.
One form of this would be generating hydrogen through electrolysis during the summer, and burning hydrogen for power during the winter. However, storing enough hydrogen to power a home for several months has its own set of problems. LNG would be easier, but still has many of the same issues.
Ammonia may be the best contender, being it can be stored at atmospheric pressure, has decently high energy density, and can be handled and consumed much like existing liquid fuels. It has the added benefit that it has no C at all; just N and H. We just need a way to produce it cleanly at small scale and decent efficiency with common input materials like the N in air, the H in water, and excess solar to power the conversion process.
https://www.euractiv.com/section/energy-environment/opinion/ammonia-a-fuel-of-the-future/
Depending on where you live another good contender is simply heating mass – ideally well insulated mass, as then your heating all winter is pre-stored (and much of the hot water all year) – so you lower daily generation in the winter only has to power the lights and cooking type daily loads, which if you have the right ballpark of solar generation capacity it should be able to do while still recharging you ‘daily’ battery.
Also don’t rule out simple liquefied/compressed air – yes its less energy dense that a compressed combustible fuel, but its also really simple to make, durable, can be efficient at basically any scale, the pitfalls seem on paper to be almost entirely extracting too fast for the size of your system ruins the efficiency and that it takes a rather large volume for the energy stored – that volume will be the killer for many places wanting really long term so very high total capacity – but its not impossible – especially if you are not trying to store more than needed – if you scale up your solar production capacity just a tiny bit so your average winter day is break even/slightly positive energy wise you only need to store in your rainy, stormy month energy fund a fraction of what it would have to be if nearly every winter day is a big drain – if you can fit that much solar is a whole other matter, on our roof we are probably able to go off grid electric, but it would be very tight and definitely needs more ‘battery’ – but being a smallish European home its got bugger all roof space compared to many American homes – and with similar levels of insulation etc the energy consumption difference between American and European lifestyles doesn’t seem vast…
As with all these longer term local energy stores there are limits, and to really get the best out of them you should have designed the building around them, retrofit into the 100+ year old brick, or 50 odd year old concrete house is tricker – wooden houses are somewhat simpler – as the upper floors etc don’t weigh half as much so you can remove load bearing timber around to create the right shaped space relatively easily.
Those are interesting suggestions, @Foldi; thank you. However, the mass and temperature needed to store a season’s worth of heat energy is beyond enormous, and insulation technologies are just not sufficient to keep it up to temp for months at a time.
Compressed air is easy and available, and can be stored for longer times without degradation, but would also require an enormous volume for seasonal storage. Plus, air is vulnerable to explosive decompression; one tank splitting open in the middle of a farm of tanks is likely to rupture others as well, with no time to respond or mitigate. Fuels burn, but don’t generally explode, and fire suppression is well understood. Neither should be stored within living quarters, IMO.
Those are my reasons behind searching for a chemical solution for seasonal energy storage. Plus, with ammonia as the product, excess can be sold or used as fertilizer, or if you are short one year, just buy some from a commercial supplier to tide you over until the sun shines again.
The mass isn’t actually a big problem – you can use the ground – all the mass you can get. Some folks have even built houses that without any active heating use the ground as a giant heat regulator (one build like that did cover the thermal store in expanded insulation foams and a layer of turf) – stay cool in summer and hot in winter passively or close to it having active water flow for heat transfer – rather clever, but it needs well insulated structure with the right exposure to the sun for energy input. The idea however does require the right location – if you are in the centre of a continent so more prone to massive temperature swings winter to summer it gets much harder to make work.
No denying the volume problem with compressed air, but if it does suit your needs its a good choice, and worth mentioning as it scales quite well, and can be ‘abused’ for stupendous energy output at terrible efficiency safely. Also ‘explosive decompression’ happening in compressed air system really means you treated it terribly or built in tissue paper with no safety measures – take something like a fire extinguisher it can take high pressures, much much higher than the working pressure to the point it almost doesn’t need the safety valve as in realistic terms if its got that overpressure you have bigger problems, and tanks like that are damn tough on the physical abuse front too – I’m not suggesting building a system with quite that level of over specification, as at home power scales that just doesn’t make sense, but give it enough safety margin in the design from the working pressure you aim for and add a safety valve or two set under your design limit and getting it to go wrong in anything but an ‘oh bother, we wasted some energy’ kind of way is going to be rather tricky unless you actively try to make it happen.
The question you have to ask yourself is how much long term storage do I need – you can go off grid just on solar with nothing but a few days worth of battery power – if you go off grid with the right level oversupply on an average winter day for your energy requirements, which is quite possible, not even unreasonable in cost or footprint if your house is efficent, then your battery supply is just covering for those longer bad spells on top of the normal day night cycle – and it doesn’t have to do all the power you need even then, on even a really foul day a rooftop solar array produces something, the battery is just making up the difference so having perhaps the EV car playing battery pack (or similar size house battery) and an efficient house you have days maybe even more than a week of power in the battery alone, but that first day of foul weather doesn’t take a full days worth out of it – the panels did something remember, so on that first day of foul weather you perhaps are down 12 perhaps 18 hours worth, so you still have days left, your ‘week’ of battery power will actually go maybe two weeks – and really foul weather that shuts you down that fully isn’t likely to last nearly that long – and if it looks like it might bugger you have to be a bit more frugal and careful than normal.
If you really insist you must store up an entire winters worth of energy consumption supplied only from this energy store its always going to be really really large physically or your house has to be stupendously efficent – as even if you were magicking up something energy dense like petrol that is a huge volume of fuel required for the average home – remember the amount of energy you actually get out burning that fuel isn’t even close to its chemical potential…
Its not a practical goal at the personal scale – you are far better off just having on average more oversupply if you have the space reducing the time you need supplementary power – and you would need the space for that huge ‘seasonal battery’, these days panels are really quite cheap, wind is an option too and other than the places that only get one sunrise a year, deep in a sheltered valley they will both work somewhat reliably year round.
From what I’ve seen documentation or demos of I don’t think methane or ammonia are really there yet – not efficient for small scale, hydrogen is harder to store but if you have the supply of water probably the best choice as it stands – easy to generate and you can ‘burn’ it in a fuel cell, which for hydrogen seem to be pretty well developed, dependable and very efficient so if you are all set up around electrical consumption – my assumption based on your mentioning of solar – it will end up working best for you – you loose a great deal of the potential energy in combustible fuels, and the smaller the engine the more that is true so the round trip efficiency creating and burning it more conventionally is very low – which means you need a bigger store, using a fuel cell you get most of the potential out usefully – so need a much smaller fuel tank.
what people don’t seem to get about “carbon capture” is that the reason there is carbon that NEEDS capture is that producing it is how one gets energy. if you then CAPTURE it, that requires energy, thermodynamically, this has to be on the order of the energy that produced the carbon to begin with. the idea of carbon sequestration is just another glorious idea for a perpetual motion machine, albeit one cloaked in the hand-waving of chemists
Except its not at all perpetual motion – the expectation is to fuel the capture with excess renewable energy – Its artificial photosynthesis of a sort – using the power of the sun via reasonably direct means to turn atmospheric gaseous carbon and oxygen compounds into useful or storable compounds.
You could say this is using a cleaner energy source to undo some of the harm caused by the dirty one – its not at all burn more carbon based stuff, to then turn the waste gasses into less carbon based stuff directly…
A thought occurred to me on my drive today (I drive a Prius and regularly get 47 plug MPG).
The answer my friends comes to us from a line in The Graduate – Plastics!
Assume for a moment that we can get energy to use to mitigate the carbon dioxide buildup in the atmosphere. From renewable or Nuclear sources that emit much less carbon than burning fossil based fuels.
We extract the CO2 and use existing chemical engineering to reform that CO2 with added chemicals into a plastic.
We can store plastic pretty easy. How many carbon atoms can we store in plastic? How can we effectively store plastic so microplastics are not a threat to the environment? Embed them in concrete? Use them in composite structures so we use less wood? We know how to use plastic. That’s easy.
The thing I think we need to discuss isn’t the elimination of carbon based fuels…it’s the industrial processing of atmospheric gasses to prevent the use of carbon based fuels from altering the environment. It’s going to take us decades at least to fix this, but if we go down the rabbit hole of NO CARBON BASED FUEL USE, we’re fooling ourselves. It’s a very stable and effective source of energy. We can still use it, we just need to clean up after ourselves.
Using renewables and green energy where it makes sense is a STEP in the right direction. The mistake that many people make is that EVERYTHING can be run on renewables effectively. They can’t. I own an RV. 35′, and it weighs 22,000 lbs. They don’t make batteries that will run it. I’m not alone, there’s a lot of them out there. They run on gas or diesel. I’m not going to abandon it because some insist that the only way to solve this is to abandon carbon based fuels.
If we approach the problem logically, you’ll see there is no one absolute answer. There can’t be. The other thing we should do is accept as a species that we should learn how to do these things….carbon capture and solid storage, renewable energy, lowering the amount of CO2 in the atmosphere, etc….not because they will fix the problem, but to say “We can do this! It is possible and we can turn it on and off as we need to do it.”
Stop the blame game. Accusing people of supporting a specific ideology doesn’t fix the issue. Looking for ways to do things just might.
I think for now we really do have to push for cutting out all carbon based fuels going forward, as we have made one heck of a mess and are not cleaning it up fast enough (or at all currently – we are just making things messier slightly slower as it stands) to keep rolling on as we have been.
I do actually overall agree hydrocarbon fuels are a perfectly valid form of energy storage and consumption, that could have a place in the future – but at least for now we need to cut them as hard as we can for the renewable as nobody has perfected the cleaning up after ourselves effectively phase yet – and we have far too much to clean to add to it all the time.
I would also suggest that long term Hydrocarbon probably doesn’t make sense as burning them has more than just a CO/CO2 negative impact, where hydrogen especially when you don’t burn it freely but use it via fuel cell doesn’t have those same drawbacks.