Hydrogen peroxide – the same stuff you can pick up from a drug store or beauty supply store – is one of those very interesting chemicals that belongs on every maker’s cabinet. At concentrations of about 30%, it’s perfect for etching PCB boards, and at even higher concentrations – about 70% – it can be used as rocket fuel. Unfortunately for the home hacker, it’s very difficult and expensive to obtain peroxide in concentrations above 3% or so. That’s alright with [Charlie], though, because he’s come up with a way to concentrate peroxide and measure the concentration once he’s done.
There are a few YouTube videos of kitchen chemists concentrating peroxide by heating it on a stove to just under 100°C. Because hydrogen peroxide boils at 150°C, they’re simply boiling off the water and increasing the concentration of peroxide. This is a qualitative method, and you’ll never know what concentration you’re getting. [Charlie] rigged up a small-scale with a pipette to measure the weight of his concentrated peroxide per unit of volume, giving him the density of his concoction and thus the concentration.
We have to note that concentrated peroxide is dangerous stuff, but the results of [Charlie]’s lab work aren’t much more dangerous than what hair stylists work with every day. If you’re going for high-test peroxide, good job, that’s awesome, but do be aware of the risks.

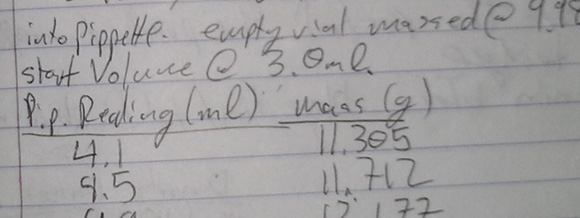













Hydroponics stores and sometimes pool stores carry hydrogen peroxide at 30%.
bruh the article said above 3% is expensive
And buying and distilling 10 times the amount of the diluted stuff is free?
1 gallon of 3% ~= 12 oz of 30% ~= $12
1 gallon of 30% (which is about the smallest you can get) ~= $45
So if you want a gallon of 30% your choice is clear. If you don’t, well, maybe a bit less so.
When your done distilling that gallon of 3% how much concentrate are you left with?
When you done distilling 30% you will have alot more concentrate than starting with a gallon of 3%, not to mention the time you save. In the end, to get the same amount of concentrate you will need to spend alot more than $45 at Walmart buying all that 3%
They sell H2O2 at dollar stores lol… so $4 for 12oz. Of 30%
Oh, you Americans and your old fashioned “galuns”
The guy is saying he can get 12 oz of 30% at Dollar General might need to get some glasses that is 3% Dollar General or any of those stores will not get anything over 3%
So making it yourself is more expensive (12oz = $12)? @ $1 an ounce it would be $128 a gallon.
You got that wrong Thad. He’s giving you the price to make 12oz of 30% from 4 bottles ($4 vs $12) of 3% hydrogen peroxide. You misinterpreted his statement and then blamed him for you own mistake. Lol.
In general I’m all for hacks, but are there any other more readily available substances that will give you the same results in your application as H2O2 you want . If there isn’t well, carry on. I’m just trying to clean a bottle I found in and old outhouse. must be from 1850 or so. Was actually looking for gold following up an urban legend of the CA gold country.
Im in Ontario, Canada and i buy 50% H2O2 no problem from a major chemical distributor. They have >1000 reagents including 30% and 50% H2O2, 95% sulphuric acid, 68% nitric acid…
The only thing is that 30% and 50% H2O2 are expensive becuase you cant pick it up in person, you have to pay for a shipping company with a license to transport hazardous matterials. What i find odd is that the local hydroponics store will let me pick up 28% H2O2 in person. Im not sure if that’s legal… But the do it.
The only thing that was a pain in the ass to get was pottasium permaganate. I had to fill in a form submitted to them stating why i needed it and where (the adress) it would be stored at.
The only problem i ever had is when a neighbour called the local bomb squad on me. They took a sample of each on my dozzens of chemicals to test that it was labled correctly and I was interviewed by them. After, they told the police that i DO have the chemicals to make many types of explosives, but I have other reasons for having them and I dont have huge quantities to even level a house.
In the UK trying this sort of thing may get the attention of the Anti-Terrorism Squad, given the recent past history of terrorists trying to use peroxide derived explosives. Just something to be aware of.
You can buy the 20% stuff in Lloyds pharmacy, if they ask what its for just say its for getting acrylic off a leather couch.
This should have been answered to another comment.
Here in Vienna we buy the stuff (30%) regularly in the 10l canister to fight algae in the swimming pool.
It warms my heart that he propagated his error and used an actual paper for his density-concentration curve instead of just using wikipedia.
Sure. Because every paper is already right while this pesky Wikipedia can’t be trusted upon, even if they (as they nowadays work hard on) make citations.
hydrogen peroxide also good for removing yellowish stain on computer plastic parts
thats smoke
some of it is, some of it is bromine from the fire retardant in old plastics. UV degrades the compound, leaving a bromide yellow. See http://retr0bright.wikispaces.com as they covered the stuff.
Nope. It’s not smoke. It’s a mix of the nature of plastics decaying when exposed to light and heat and chemical fire retardants used in older plastics.
It could also be smoke. When I was a smoker I had to literally wash every damn white plastic surface with strong kitchen soap twice a year because they became yellowish over time. Switched to normal cleaning when I quit smoking.
@qwerty
Surface is not the only thing that gets affected by smoke. I once buyed a CPU from eBay. They claimed it to be barely used. When took it out I noticed the dust on the heatsink and then that smell – it smelled like an ashtray.
Getting 12% isn’t hard, it’s called #40 activator from the hair salon supply store.
I was wondering if activator was ok (for cupric acid for instance), isn’t there other chemicals in there too?
As a user of Retr0bright, I recently needed to find higher than average concentrations of hydrogen peroxide as well. What I discovered was 5 gallon drums of 30% for sale from a local industrial cleaning supply place. It’s sold under the brand “Max lift” and, according to the MSDS sheet, is nothing more than hydrogen peroxide and water. I think they expect dry cleaners to buy it.
IIRC it was about $100 for a bucket of the stuff. That may sound like a lot, but it’s much cheaper than buying 50 gallons of 3% and distilling it to get the same 5 gallons of 30%
Hopefully this’ll prove beneficial to someone.
Huh, this must be about the first time I’ve seen that somebody in the US says it’s hard to get a chemical, while here in NZ our domestic ebay type site has several sellers of 35% h202 in 1, 2 and 5L quantities. A Litre of 35% would cost me about $20 US equivalent, including shipping.
Just looked on ebay.com and quite interesting, 35% listings in the UK, but none in the US, looks like you guys don’t sell over about 17%. Is there some law preventing the sale of higher concentrations, or is there maybe just no market for it.
One of the reasons is that 30%+ H2O2 mixed with acetone gives you triacetonetriperoxide and/or a big explosion. It is this reaction that caused the banning of liquids on carry on luggage in planes.
I thought you needed sulfuric acid too to make TATP
the acid is just a catalyst, only small amounts are required. That’s what car batteries are for ;)
You don’t need 30% peroxide to make an explosive, 3% is more than enough.
But seriously anybody making such a dangerous mixture should be considered insane. There are stronger explosives that are just a bit more complicated to make and have the advantage of not exploding by its own weight, exploding due to crystallization (which can increase inner strain) or explode due to light (!) pressure changes static electricity or the color of ones clothes. Ok, I made the last thing up on the spot but wouldn’t be surprised if TATP reacted with some coloring agents.
BTW here I can order 55% H2O2 without any problem or buy 20% locally.
Worst part about TATP is that it sublimates – leave some in a jar and it will sublimate and recrystalize in the threads. Say goodbye to your hands when you want to open it. But I agree, making nicer explosives is very easy, RDX is just hexamine (fuel tablets) with nitric acid – all you need then are some plasticizers and you have C4 or Semtex.
It isn’t THAT sensitive unless you totally screw up when making it. As long as you can keep it cold while reacting and crystallizing it is fairly stable. I’ve experimented with small amounts, a few grams at a time. There is nothing crazy about that as it isn’t particularly powerful. If you screw up and the reaction temps get too high you end up with some really unpredictable stuff that may just explode for seemingly no reason at all.
its been over a decade, but go off…
We have an abundance of people in the US that are just too stupid for their own good. For example mixing bleach, drano, and water creates a visible gas that almost instantly gives you an intense headache, stay exposed long enough and the headache goes away while euphoria sets in, even longer and your either in a coma or dead. The average reaction to this is not to vent the area, it’s to sit in the gas filled home while complaining about how much it makes their nose sting.
It seems like common sense, if it makes your nose sting, you should not be breathing it, but never underestimate stupidity.
Look at the “note to self” safety message he left on the peg board
“Wear yer apron and ya might not have to get nekkid during a spill” [sic]
=)
Be careful with peroxide, I work regularly with high concentration HP from 35 to 75%, and it can be dangerous. Keep away from organic materials if you don’t want to burn your house down !
This statement is incorrect “Because hydrogen peroxide boils at 150°C, they’re simply boiling off the water and increasing the concentration of peroxide”
H2O2 does decomposes into water and oxygen slowly a room temperature if you try to distill it, by boiling it you increase the rate of decomposition. This is why you can never
get rocket fuel from low volume H2O2. There is a much better method of peroxide
determination by iodometric titration, here’s a good reference its method 2
http://www.ch.ic.ac.uk/spivey/documents/iodometric.pdf
That leads to an error page
terrorist protection.
cant buy iodide crystal, or over 2% anymore either…
http://farmwars.info/?p=9963
How the hell is a terrorist going to successfully make a sufficient amount of nitrogen-tri-iodide in-situ (moving it is pretty much impossible) to do any real damage? WTH?
Make it wet and let it dry on target? Besides NI3 is better for scaring people. My chemistry teacher painted some spots on the lab floor with this stuff and laughed as people walked over it.
I am not 100% sure but I think the iodine restrictions are also because iodine is used in the making of illicit drugs (in addition to its uses in explosives)
you know you can place the stuff in the freezer and separate the water from it
http://en.wikipedia.org/wiki/Applejack_(beverage)
applejack used to be made that way they would would leave the raw liquor mash in the cold and skim off the ice/slush
i think you can do the same thing with soda leave a bottle of soda in the freezer too long and you get ice and slush and remove the ice slush and you should be left with the soda syrup.
No, that doesn’t work. You just get frozen soda.
Works with grapefruit juice…
Water freeze leaving you with the bitter sirup.
Freeze distillation, for the record, should work fine for hydrogen peroxide solutions. And I reckon it’s likely to be both safer and more effective in terms of yield than conventional distillation. H202 lowers the freezing point and raises the boiling point of an aqueous solution; this simultaneously increases the expected efficacy of freeze distillation and decreases that of conventional distillation.
Depends on how it’s frozen.
Had it happen sometimes to me, if its frozen at a temperature just below the freezing point of water.
In The Netherlands it’s very easy to buy 30% H2O2 (euro 8,32/l); most on-line pharmacies have it in stock. The same goes for 30% HCl (euro 8,8/l), sulphur (euro 5,51/800g) and many other fun chemicals.
(a) hydrogen peroxide (CAS RN 7722-84-1) in concentrations higher than the limit value set out in Annex I, but no higher than 35 % w/w;
(b) nitromethane (CAS RN 75-52-5) in concentrations higher than the limit value set out in Annex I, but no higher than 40 % w/w;
(c) nitric acid (CAS RN 7697-37-2) in concentrations higher than the limit value set out in Annex I, but no higher than 10 % w/w.
(This is from the document you posted).
That nitric acid concentration would be terrible, luckily one can purify by use of distillation, fractional would be ideal.
Why not here when making pure nitric acid is so easy. Mix salt peter and sodium bisulphate (stump remover, and dry acid from pool store.) And hear over a flame in a glass flask running into a glass Condenser. The residue is highly water-soluble potash to put on your lawn.
How about using a closed system and a simple vaccum pump and then make the destillation at room temperature? If the vaccum pump does not get damaged by the rather agressive hydrogen peroxide it should work.
Why distill? Hydrogen peroxide has a higher boiling point than water, so boiling off is enough. Distilling the vapor would give you water.
Because higher temperatures promote its decomposition
At 1 psi, pure water will boil at near room temperature. The idea here is to boil it with vacuum pressure then use a desiccant stored in the same system to remove the water vapor from the container. Could work readily to produce the small amounts of high conc hper that are kind of safe to work with.
Why do you need the desiccant, if you are drawing a vacuum on the container? Just leave the pump running.
i had to do this a few weeks ago for ap chem. we had to find the concentration of sugar in 7up assuming it was just a solution of sugar water. we had to do something like find the difference in the density of water and sugar and then do some other calculations to get the right answer by comparing the masses and densities. the special part we did with the difference in density gave us accurate results, while everyone was off by a good 6%
too dangerous, don’t keep it at your home
that’s what sank the Kursk submarine, it’s a highly unstable stuff and will burn your house down or just explode in your face
in a word: don’t
I thought ruskis sank the Kursk coz they refused assistance from available help
Whatever sank the Kursk and whatever subsequently killed the surviving crew, neither one was refusing anyone’s assistance. I think the final blow to the crew was the explosion of a superoxide chemical in an oxygen candle device.
Friend of mine had a chemical explosion in high school chemistry. 10 years and 3 eye transplants later finally had a successful transplant and regained vision in second eye. Know what you are doing before doing stupid stuff.
eye transplant? More probably cornea transplants. I don’t think you could transplant eyes.
it is very dangerus
What percentage did germans used in the h2o2 subs…also how did they work..i understand it was enought to burn underwater and provide water
and some o2.
It is used above 70% up to high 90% in the torpedoes. It is passed through a mesh of platinum which acts as a catalyst, changing the hydrogen peroxide back to water in the form of steam at over 600 degrees Celsius which propels it.
Your correct
Folks one point seems to be missing from the H2O2 discussions. That is if you buy Clear developer at Beauty supply store it is fairly cheap and no records are kept of the sales or other headaches. However it is sold as 20 volume strength, 30 Volume strength and other concentrations up to about 40 or 45 volume strength, VOLUME STRENGTH IS NOT SAME AS PERCENT CONCENTRATION ! IE 20 Volume strength is NOT 20% H2O2 !! it is much less than that. I don’t recall the exact percentages to volume strength conversions however even 20 Vol. St. will turn your fingers white and cause mild chemical burns (not real problem, mostly pulls oils and water from a few layers of skin, hand lotion and moisturizer and a bit of time cures these ‘wounds’, gloves is smarter choice!). Rocket fuel strengths of H2O2 that can power a jet belt (old jet pack by BELL seen in James Bond movie uses 80% H2O2 and higher as its fuel which is pushed by N2 gas into a reaction chamber filled with activated silver or Nickel gauze which decomposes it violently into very high pressure live steam, which is what is responsible for its thrust!) 80% is very different from any available H202 with out special purchase and handling requirements from very few supply houses.
Hi. I would like to ask. How can I dilute 10% H202 to 3% H202. I probably bought 10% H202 at the pharmacy just this afternoon. I am wondering if How can I possibly use this peroxide if it is 10% solution. I would like to try it in laundry, kitchen utensils, seed sprouting, vegetable cleaner as well as dishwashing cleaner. Can anyone help me please, It gives me a headache whether I can use it directly or not. The vegetable stuff and as a cleaning spray to my son’s toys. Pretty please. Help me with this
Its simply add cold water according to the below the equation: (M1*V1)/M2=V2)
where m1= initial concentration
where V1= initial volume
where m2= initial concentration
where V2= initial volume
That’s giberish. 2 terms for the same value? You must mean m2 is FINAL/desired concentration and V2 is final volume.
Probably the easiest way to accomplish what you want is to dilute your ten percent peroxide with twice as much plain water. Soa glass of 10% peroxide plus two more of straight water gives you three times the original amount, now at 3.3%. That’s plenty close enough for the uses you mentioned without the extra steps to get exactly 3% concentration. If you absolutely must have exactly 3%, your better off buying it at the dollar store!
During distilling off water from a mixture of 30/ 70 % Hydrogen Peroxide, it is quite possible that peroxide will be lost?
How much?, It is a matter of finding experimentally.
http://www.tecaeromex.com/ingles/destilai.htm
If you have the time and money you can contact this lunatic. He’s been doing scary stuff with hydrogen peroxide for a while now.
I was just wanting to make higher concentration to whiten teeth?
I hope you’re joking. Please use the special toothpaste, or you can use regular 3% H2O2 as an oral rinse. I strongly recommend rinsing your mouth with water after though, from personal experience.
Carbamide peroxide is used in teeth-whitening gels. They are absurdly expensive and very easy to make. Or used to be. Just add urea to peroxide (30% p. is used but you’ll get away with lower) and chill down. Collect the crystals. You can find the ratio it on wikipedia. It doesnt decompose like the liquid H2O2. When mixed with water it dissociates back to urea and hydrogen peroxide in desired concentration. Urea is good for teeth btw. Maybe not so much for funny stuff, who knows… A solid H2O2 still sounds like magic to me.
If you decide to use it dont smoke or drink coffee for some time. I did and the yellow color came back even worse. You must fill the micro cracks with some minerals. I suggest zinc oxide. Phosphorus would make sense maybe. Calcium (hydroxide) turns yellow easily. Enamel is made of both.
I used to use it while sunbathing my teeth with a C-shaped retractor. UV light from the Sun works on its own. Just not in the cracks and very fast. Both can be hard on your gums. It’s complicated
The developer at the beauty supply has additional chemicals in it. too many to list
What is even more stupid: thanks to *one idiot* trying to make a boom boom box out of of defunct laptop batteries, you now can’t get 18650’s locally. I mean I ask you, you can go to any corner shop and buy a “phone charger” pack with 18650s but they have been replaced with gelpacks now because of IATA restrictions.
Can I dilute 50 % down to 3% and use for general household cleaning
Yes
Damn i just wanna clean some old yellow star wars figures
Can any one please help. I have people who use food grade 30% H2O2 (one minute cure ) for their cancer and it seems to be working however in UK they no longer allow 30% conc.They only allow 12%. Can someone please tell me how I can make 30% from 12%.
could be as simple as if I am adding 3 drops a day to little water i ll add 7 drops?
or is there a way to calculate?
Hydrogen Peroxide (H2O2) is measured in w/v, which means number of grams in 100ml.
30% is 30g in 100ml
12% is 12g in 100ml
If all that mattered was amount of H2O2 consumed then;
30/12 = 2.5 – This is your dilution factor
I.E. 3(drops at 30%) * 2.5 = 7.5 (drops at 12%)
So what is consensus on
using 20% peroxide for whiter teeth.
new dentist procedure utilizes UV light to heat the applied medical grade peroxide the heat activates the peroxide thus whitening your teeth
instantly but cost as much as $500.00
UV light doesn’t heat things well, sure they didn’t use IR light?
Well, I really don’t know about the point of using peroxide in that process – unless gingivitis or periodontitis is being treated alongside tooth whitening? I state that because for the past fifteen years, a light for DIY tooth whitening has been sold from a myriad of places online. The light worked alone. I’m kickjng myself for not buying one from an over stocked wholesalers when I had the chance, for about £10!
the idea in todays world is to find an easy (or not) way to make h202 frm h20 or whatever. the more you can create from nature on a personel scale the better for us and nature.
Buying more than 12% concentration has been banned since 2020 (?) is now banned for non professionnals in the EU. Considered biocide explosive etc… Can no longer find it in swimming pools appliances.
Befriend a hairdresser?