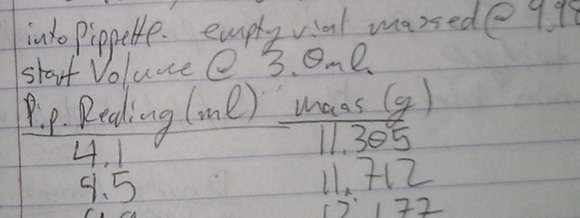Multiple people have recently shared this exciting demonstration (nitter) with us – visualizing airflow using a smartphone, called ‘background-oriented schlieren’. On a hot summer day, you might see waves in the air – caused by air changing density as it warms up, and therefore refracting the light differently. Schlieren photography is an general set of techniques for visualizing fluid flow, but of course, it can also be applied to airflow. In this case, using some clever optical recognition tricks, this schlieren method lets you visualize flow of air using only your Android smartphone’s high resolution camera and a known-pattern printed background! Continue reading “Observe Airflow Using Smartphone And Background-Oriented Schlieren”
density5 Articles
“Hey, You Left The Peanut Out Of My Peanut M&Ms!”
Candy-sorting robots are in plentiful supplies on these pages, and with good reason — they’re a great test of the complete suite of hacker tools, from electronics to machine vision to mechatronics. So we see lots of sorters for Skittles, jelly beans, and occasionally even Reese’s Pieces, but it always seems that the M&M sorters are the most popular.
This M&M sorter has a twist, though — it finds the elusive and coveted peanutless candies lurking in most bags of Peanut M&Ms. To be honest, we’d never run into this manufacturing defect before; being chiefly devoted to the plain old original M&Ms, perhaps our sample size has just been too small. Regardless, [Harrison McIntyre] knows they’re there and wants them all to himself, hence his impressive build.
To detect the squib confections, he built a tiny 3D-scanner from a line laser, a turntable, and a Raspberry Pi camera. After scanning the surface to yields its volume, a servo sweeps the candy onto a scale, allowing the density to be calculated. Peanut-free candies will be somewhat denser than their leguminous counterparts, allowing another servo to move the candy to the proper exit chute. The video below shows you all the details, and more than you ever wanted to know about the population statistics of Peanut M&Ms.
We think this is pretty slick, and a nice departure from the sorters that primarily rely on color to sort candies. Of course, we still love those too — take your pick of quick and easy, compact and sleek, or a model of industrial design.
Continue reading ““Hey, You Left The Peanut Out Of My Peanut M&Ms!””
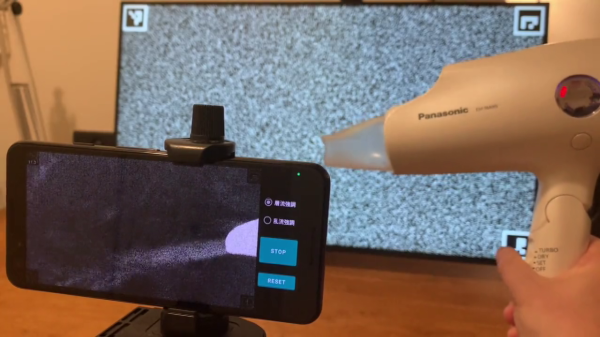
Schlieren On A Stick
Schlieren imaging is a technique for viewing the density of transparent fluids using a camera and some clever optical setups. Density of a fluid like air might change based on the composition of the air itself with various gasses, or it may vary as a result of a sound or pressure wave. It might sound like you would need a complicated and/or expensive setup in order to view such things, but with a few common things you can have your own Schlieren setup as [elad] demonstrates.
His setup relies on a cell phone, attached to a selfie stick, with a spherical mirror at the other end. The selfie stick makes adjusting the distance from the camera to the mirror easy, as a specific distance from the camera is required as a function of focal length. For cell phone cameras, it’s best to find this distance through experimentation using a small LED as the point source. Once it’s calibrated and working, a circular field of view is displayed on the phone which allows the viewer to see any change in density in front of the mirror.
The only downside of this build that [elad] notes is that the selfie stick isn’t stiff enough to prevent the image from shaking around a little bit, but all things considered this is an excellent project that shows a neat and useful trick in the photography/instrumentation world that could be useful for a lot of other projects. We’ve only seen Schlieren imaging once before and it used a slightly different method of viewing the changing densities.
Sounding A Sour Note Can Save People From A Sour Stomach (Or Worse)
We’ve covered construction of novel music instruments on these pages, and we’ve covered many people tearing down scientific instruments. But today we’ve got something that managed to cross over from one world of “instrument” into another: a music instrument modified to measure a liquid’s density by listening to changes in its pitch.
This exploration started with a mbira, a mechanically simple music instrument. Its row of rigid metal tines was replaced with a single small diameter hollow metal tube. Filling the tube with different liquids would result in different sounds. Those sounds are captured by a cell phone and processed by an algorithm to calculate the difference in relative density of those liquids. Once the procedure was worked out, the concept was verified to work on a super simple instrument built out of everyday parts: a tube mounted on a piece of wood.
At this point we have something that would be a great science class demonstration, but the authors went a step further and described how this cheap sensor can be used to solve an actual problem: detecting counterfeit pharmaceuticals. Changing composition of a drug would also change its density, so a cheap way to compare densities between a questionable sample against a known good reference could be a valuable tool in parts of the world where chemistry labs are scarce.
For future development, this team invites the world to join them applying the same basic idea in other ways, making precise measurements for almost no cost. “Any physical, chemical, or biological phenomena that reproducibly alters the pitch-determining properties of a musical instrument could in principle be measured by the instrument.” We are the ideal demographic to devise new variations on this theme. Let us know what you come up with!
If you need to do quick tests before writing analysis software, audio frequency can be measured using the Google Science Journal app. We’ve seen several hacks turning a cell phone’s camera into instruments like a spectrometer or microscope, but hacks using a phone’s microphone is less common and ripe for exploration. And anyone who manages to make cool measurements while simultaneously making cool music will instantly become a serious contender in our Hackaday Prize music instrument challenge!
[via Science News]
Making High Concentrations Of Hydrogen Peroxide At Home
Hydrogen peroxide – the same stuff you can pick up from a drug store or beauty supply store – is one of those very interesting chemicals that belongs on every maker’s cabinet. At concentrations of about 30%, it’s perfect for etching PCB boards, and at even higher concentrations – about 70% – it can be used as rocket fuel. Unfortunately for the home hacker, it’s very difficult and expensive to obtain peroxide in concentrations above 3% or so. That’s alright with [Charlie], though, because he’s come up with a way to concentrate peroxide and measure the concentration once he’s done.
There are a few YouTube videos of kitchen chemists concentrating peroxide by heating it on a stove to just under 100°C. Because hydrogen peroxide boils at 150°C, they’re simply boiling off the water and increasing the concentration of peroxide. This is a qualitative method, and you’ll never know what concentration you’re getting. [Charlie] rigged up a small-scale with a pipette to measure the weight of his concentrated peroxide per unit of volume, giving him the density of his concoction and thus the concentration.
We have to note that concentrated peroxide is dangerous stuff, but the results of [Charlie]’s lab work aren’t much more dangerous than what hair stylists work with every day. If you’re going for high-test peroxide, good job, that’s awesome, but do be aware of the risks.