Walt and Molly Weber had just finished several long weeks of work. He was an FBI agent on an important case. She had a management job at Houghton Mifflin. On a sunny Friday evening in February of 1995, the two embarked on a much needed weekend skiing getaway. They drove five hours to the Sierra Mountains in California’s Mammoth Lakes ski area. This was a last-minute trip, so most of the nicer hotels were booked. The tired couple checked in at a lower cost motel at around 11:30pm on Friday night. They quickly settled in and went to bed, planning for an early start with a 7am wakeup call Saturday morning.
When the front desk called on Saturday, no one answered the phone. The desk manager figured they had gotten an early start and were already on the slopes. Sunday was the same. It wasn’t until a maid went to check on the room that the couple were found to be still in bed, unresponsive.
A botched repair on the room’s gas-fired heating unit caused it to vent exhaust into the small room. Carbon monoxide quickly built up, poisoning the athletic couple. Walt was dead, and Molly barely alive. She was rushed to Loma Linda University Medical Center. She presented as clinically brain dead. Her athletic physique made her a great organ donation candidate. [Dr. Takkin Lo] refused to declare her though, instead going with his gut and trying out an unproven treatment to save her life. She was placed in a Hyperbaric Oxygen (HBO) chamber, and given an aggressive schedule of treatments.
Her family rallied around her, coming from all corners of the country to be with their loved one. Against all odds, Molly awoke from her coma nine days later. She wasn’t out of the woods though. The ordeal left her with a severe brain injury. She needed to re-learn how to walk, swallow, and perform everyday tasks. Most distressing to Molly’s family was the fact that her whole personality changed. The person who came out of the coma wasn’t the same one who checked into that motel.
 Sadly, Molly’s story isn’t fiction. It’s a fact that she and her family have lived with for over 20 years. Molly’s sister Lyrysa Smith has documented the story in an excellent book called A Normal Life: A Sister’s Odyssey Through Brain Injury. The book which describes the human side of dealing with severe brain injuries. She manages to put into words how difficult it is for caregivers of people with brain injuries. Psychologists say that when a loved one receives a brain injury, the whole family receives a brain injury.
Sadly, Molly’s story isn’t fiction. It’s a fact that she and her family have lived with for over 20 years. Molly’s sister Lyrysa Smith has documented the story in an excellent book called A Normal Life: A Sister’s Odyssey Through Brain Injury. The book which describes the human side of dealing with severe brain injuries. She manages to put into words how difficult it is for caregivers of people with brain injuries. Psychologists say that when a loved one receives a brain injury, the whole family receives a brain injury.
This tragedy could have been avoided had a carbon monoxide detector been installed in Walt and Molly’s hotel room. Many motels and hotels still don’t have carbon monoxide detectors, which is why Lyrysa recommends always traveling with a battery-powered detector.
The Silent Killer

Carbon monoxide is an odorless, tasteless, colorless gas with nearly the same molar mass as air (CO is 3% lighter on average). This means that CO doesn’t rise or fall, but disperses evenly into the air of an enclosed space. That’s why detectors can be placed low on a wall at an outlet, or high up on a ceiling. The gas is toxic to humans at concentrations above 35 Parts Per Millions (PPM). Because of this, carbon monoxide has been dubbed the silent killer. We’ve all heard not to use a kitchen stove as a heating appliance, or not to run a generator inside the house. The reason is carbon monoxide.
The American Center for Disease Control has stated that unintentional carbon monoxide poisoning accounts for over 20,000 emergency room visits each year, including over 400 deaths. Carbon monoxide poisoning starts with a headache. It progresses to dizziness, nausea, and general flu-like symptoms. Most people think they’re just coming down with the flu, and head to bed. This is often a fatal mistake.
Biology
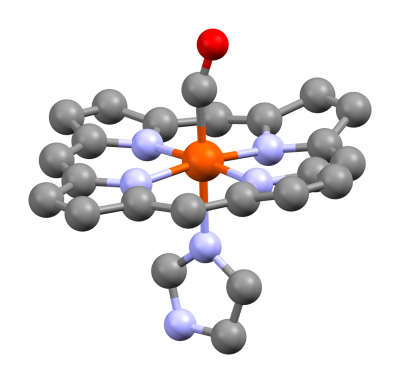
Carbon monoxide can always be found in small amounts in the human body. The molecule is known to have some therapeutic anti-inflammatory effects in humans. At higher concentrations though, CO becomes incredibly toxic. The most frightening part about carbon monoxide poisoning is the method in which it operates. Hemoglobin is the molecule in red blood cells which carries oxygen. Hemoglobin loads up with oxygen in the lungs, becoming oxyhemoglobin. The circulatory system then carries this oxyhemoglobin throughout the body, where it delivers its payload to muscles and organs. Carbon monoxide also bonds to hemoglobin, creating carboxyhemoglobin. In fact, the bond is over 200 times stronger than oxygen. This means carboxyhemoglobin doesn’t separate so easily. The carboxyhemoglobin essentially becomes an inert molecule riding through the circulatory system, starving the organs of oxygen.
This is where things get nasty.
Everyone knows that the treatment for carbon monoxide poisoning is to get to fresh air. However, it won’t immediately remove carboxyhemoglobin from the blood. That takes time. Carboxyhemoglobin has a half-life of 4 to 6 hours. There is a way to speed things up though. Administering pure oxygen to a victim can reduce the half-life down to less than an hour.
In extreme cases, hyperbaric oxygen treatments are used. The victim is placed into a pure oxygen chamber pressurized to three atmospheres. This forces oxygen to diffuse into the blood plasma, where it is carried to starved tissues.
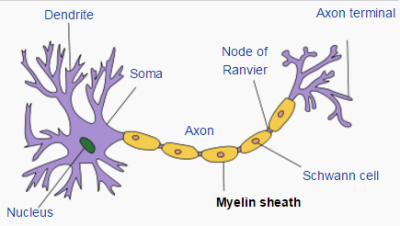
For acute poisoning patients, surviving the initial episode doesn’t mean the worst is over. Many patients begin to make a recovery, but between 2 and 40 days later, things change. The patients rapidly show signs of further brain damage. Balance, memory, and cognitive functions all affected. This phenomenon is called delayed neuropsychologic sequelae, and it was devastating for Molly Weber. The mechanism of neuropsychologic sequelae is still not completely understood. Research has shown that carbon monoxide damages Myelin Basic Protein (MBP), the material which surrounds nerve cells. The damaged MBP triggers the body’s immune system. White blood cells called leukocytes attack and remove the damaged MBP. The leukocytes don’t stop there though. They begin to attack healthy MBP, destroying healthy brain tissue. The result of this biological one-two punch leaves permanent brain injuries that can take years to recover – if recovery is possible at all.
In researching this article I was reminded how little we know about the brain, how it can be injured and how it recovers from those injuries. If there is one place where bio-hackers can really make a huge difference, it’s in studying and trying to understand how all this works.
Environment
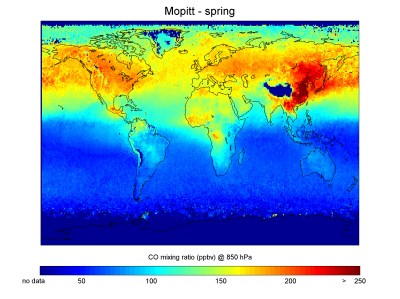
Carbon monoxide is created by several different methods. Volcanoes and other geological sources release carbon monoxide into the atmosphere, as do forest fires. The major contributor though is man. Satellites such as NASA’s Terra spacecraft keep an eye on carbon monoxide in the atmosphere. Streaks are often found over cities and where crop residue and forests are being burned.
The chief way CO is created is through incomplete combustion of carbon-based fuels. If there isn’t enough oxygen present to oxidize a fuel to CO2, CO is the result. Internal combustion engines produce huge amounts of carbon monoxide. A properly tuned gasoline engine can produce as much as 30,000 ppm CO. In the United States, gasoline and diesel vehicles produced after 1975 have catalytic converters which greatly reduce CO emissions. However, not all vehicles are well maintained. Every year deaths are reported from people sitting in idling cars with faulty exhaust systems.
Small engines such as generators and power washers don’t tend to have catalytic converters, yet they still produce large amounts of carbon monoxide. Generators running inside homes kill families every year. Even running a power washer in a semi-enclosed space such as a parking garage is enough for the gas to build up to dangerous levels.
In the home, most carbon monoxide poisoning events happen due to problems with gas-fired appliances. A properly tuned water heater, boiler, or furnace will create some CO. If the air band isn’t correctly adjusted, CO levels rise. If the exhaust becomes blocked or compromised, the CO will find its way into the living spaces. Just in the last week, a home in Oklahoma filled with CO when roofers blocked the water heater exhaust stack. Thankfully, the family had a carbon monoxide detector in their home, and nobody was injured.
Sensing Carbon Monoxide
As the above Oklahoma family learned, the best defense against carbon monoxide poisoning is a good detector. This requires a method to sense the gas, which is no easy task. To be reliable as safety devices, detectors and the sensors within them need to be accurate and repeatable. There are a few different ways to measure CO. Semiconductor sensors use tin dioxide heated to 400 °C. The resistance of the sensor changes when exposed to CO. A great example of this type of sensor is the MQ-7, which is popular in the maker crowds. Unfortunately the power requirements of such a sensor means it isn’t very well suited for battery-powered devices.
The vast majority of carbon monoxide sensors use electrochemical cells, very similar to fuel cells we often see here on Hackaday. The typical carbon monoxide sensor is a two or three terminal device with two or three electrodes: a working electrode, counter electrode, and sometimes a reference electrode. All the terminals contact an electrolyte fluid. In carbon monoxide cells, the electrolyte is often sulfuric acid.

The working electrode is where the action happens. Typically made of platinum, the working electrode sits just behind a watertight but gas permeable membrane. Air enters the cell and contacts the electrode. The platinum oxidizes any carbon monoxide, causing an imbalance in the cell. The counter electrode is a place where electrons are added or removed to “counter” the reactions occurring at the working electrode. If present, the reference electrode is isolated from the carbon monoxide, and provides a reference to the potential of the electrolyte. It is often omitted from modern detector designs. Nowadays, the counter electrode does double duty – acting as a measurement point and balance input to the cell. Every cell is different, which is why most manufacturers test each cell, even printing the mA per PPM value right on the cell case.
Managing and reading carbon monoxide sensor takes a fair bit of electronics. In the classic analog system, an op-amp circuit called a potentiostat monitors the reference electrode, and changes the voltage at the counter electrode to keep the cell in balance. The overall output of the cell is measured as current traveling into or out of the working electrode. This current is in nanoamps, so a transimpedance amplifier is used to convert current to voltage, as well as boost the signal to a level which can be easily measured by an analog to digital converter.
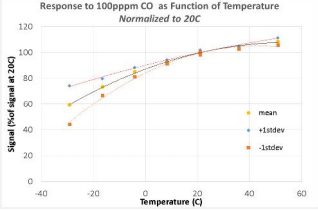 For a practical view on working with carbon monoxide cells, I reached out to [Victor Aprea] of Wicked Device. [Victor] created the Air Quality Egg, an open source air quality measurement device. Victor found that getting accurate carbon monoxide readings requires more than just reading the sensor. Sensor response curves change based upon environmental conditions. Temperature is the big one – CO readings can be off as much as 15% in extreme temperature situations. Atmospheric pressure is another factor, as are the influence of other gasses. [Victor] spent quite a bit of time working out the curves and modifying the firmware of the Air Quality Egg to compensate. He wrote several informative blogs on his work: here, here, and here. [Victor] also has found cross-sensitivity with other gasses is an issue. Thankfully for the particular CO sensors used in the Air Quality Egg it isn’t quite as big a problem as the environmental issues.
For a practical view on working with carbon monoxide cells, I reached out to [Victor Aprea] of Wicked Device. [Victor] created the Air Quality Egg, an open source air quality measurement device. Victor found that getting accurate carbon monoxide readings requires more than just reading the sensor. Sensor response curves change based upon environmental conditions. Temperature is the big one – CO readings can be off as much as 15% in extreme temperature situations. Atmospheric pressure is another factor, as are the influence of other gasses. [Victor] spent quite a bit of time working out the curves and modifying the firmware of the Air Quality Egg to compensate. He wrote several informative blogs on his work: here, here, and here. [Victor] also has found cross-sensitivity with other gasses is an issue. Thankfully for the particular CO sensors used in the Air Quality Egg it isn’t quite as big a problem as the environmental issues.
I should note that there is more to a detector than just the sensing element. Most countries have safety standards for designing carbon monoxide detectors. In the USA, the commonly accepted standard is UL 2034. The 2034 standard is based upon a 10% carboxyhemoglobin saturation in humans. The idea is that a detector alarms when concentrations are high enough to be dangerous, but only after a certain amount of time has passed. An example would be at 70 PPM, the alarm should sound between 60 and 240 minutes. It sounds a bit counterintuitive, as humans begin suffering from CO poisoning at around 35 PPM. The time delays are to reduce false alarms. A carbon monoxide detector is no good if it false alarms so much that someone pulls the batteries out due to false alarms.
As part of my research for this article, I ran a few tests on commercial carbon monoxide alarms. You can find my the results of my tests and see the tear down over on my Hackaday.io project page.
So, Hack?
So why is all this on Hackaday? Simple, because CO detectors aren’t prevalent enough. In the US, there are laws regarding the use of carbon monoxide detectors. However, they are still incredibly soft.
According to the National Conference of State Legislatures (NCSL):
- 5 states require CO detectors in schools buildings.
- 12 states require CO detectors in hotels and motels.
- 37 states require CO detectors in homes.
Just about everyone is carrying a smartphone these days. Why don’t we have carbon monoxide detectors inside them? The same goes for smartwatches. The much hyped sensor suite on the Apple Watch is capable of measuring oxygen saturation in the wearer’s blood. Adapting this sensor to measure carbon monoxide saturation is a matter of adding a couple of LEDs and software.
There have been a couple of efforts to mix smartphones and sensors. The CELL-ALL project was a 2010 effort on the part of DHS to add sensors to cell phones. Unfortunately it hasn’t really gone anywhere.

SensorDrone is a sense-everything device which did well on Kickstarter back in 2013. SensorDrone did get delivered to backers, yet it never got the community support Sensorcon was hoping for. Today, it seems to have disappeared from Sensorcon’s website. Atomtube is a similar project which recently finished up on Indiegogo. It hasn’t delivered yet, so I can’t say much on how well it will perform in the real world.
Standalone wearable carbon monoxide sensors do exist, but they have some caveats. Companies like BW and Sensorcon sell these sensors for firefighters, HVAC techs and other professionals. Priced at $100 and up, the sensors are still relatively expensive. They also require periodic calibration and bump testing, which means either investing in an expensive calibration gas system, or sending the sensor back to the manufacturer and paying a fee. Much of this is required because electrochemical cells don’t age well. Their response decreases over time. The cells can also be damaged if exposed to chemicals and certain solvents. Still, wearable carbon monoxide detectors are proven to save lives. Two EMTs on Long Island were eating breakfast at a diner when their CO detectors went into alarm. The EMTs were able to evacuate diners before anyone was sickened. Just one year earlier, one man died and 26 were hospitalized after a CO leak at another Long Island restaurant. Working out new ways to detect carbon monoxide – spectroscopy, different silicon sensors, or technologies which I haven’t even thought of – might be better ways to handle the problem. Any advancement in Carbon Monoxide sensing and detection would make an excellent entry in the Hackaday Prize. If everyone works together on projects like this, we can work toward ensuring that stories like Molly’s never happen again.
Author’s note: I’d like to thank Lyrysa Smith and Victor Aprea for their help in researching this article.
















This is an issue that shouldn’t exist, because we shouldn’t be burning gas for heat and hot water in homes. Domestic and business space heating, along with cooking on gas, is responsible for roughly a third of all our CO2 output.
With direct solar water heaters, storage boilers, underground seasonal heat storage in the district level and heat pumps it’s possible to entirely eliminate the need for gas and heating oil in most locations in the US and Europe, and this is both cheaper and more effective in reducing the use of fossil fuels than trying to produce renewable electricity to the grid at great cost and complexity.
The only difficulty is the extremely low cost of gas at the moment, because it’s hard to beat 1-2 cents a kWh, so people install instant gas boilers instead of storage boilers that could run on solar or wind energy.
In some places, electric heat is not financially possible. Here in New Hampshire, for example, our power costs are among the highest in the nation, so most of us heat with oil, gas, or kerosene. The choice is heat with these fuels or literally freeze to death. Electric heat is only feasible for the very richest among us, and they usually heat with oil or gas also because it’s many times cheaper.
That’s why I said heat pumps and direct solar collectors with storage boilers.
There’s a whole community in Canada which heats itself solely with solar heat that is collected throughout the year into a series of 30 meter deep boreholes in the middle of the town, and then pumped back into the homes in the winter similiar to European district heating.
If the weather outside is not too much below freezing, even an air-air heatpump can deliver 5 times the heat to the input electricity, and solar collector systems still work in the winter every time they get even a little bit of sun, which also slashes the electric bill.
http://inhabitat.com/a-unique-solar-powered-community-in-canada/
“There are 800 solar panels located throughout the community on garage roofs, and they produce 1.5 mega-watts of thermal power during a summer day and supply heat to the district heating system. The whole system meets 90% of the annual heating and hot water needs of the homes.”
52 home community heated on solar power, in Alberta.
https://en.wikipedia.org/wiki/Drake_Landing_Solar_Community
Homeowners are receiving an average of $60 per month solar utility bill for heating.
You seem to have side-stepped the issue though.
It’s great this technology is being used, but many people can’t afford the initial installation/retro-fit costs.
Management in many cities don’t see in a progressive light so encouraging them to make the city ‘better’ when it works already is hard to do unless you can share with them a method in which they can recoup the costs during their term.
“It’s great this technology is being used, but many people can’t afford the initial installation/retro-fit costs.”
Of course it only fully applies to new homes, but even with older homes it’s not that expensive to install air-to-air heatpumps and better storage boilers with vacuum tube collectors on the roof. The market is simply in its infancy at this stage so there’s not much supply and offering yet.
As a rule of thumb, heating with an A2A heatpump is 40% cheaper than heating directly with electricity. I’m reading reports from Scandinavia where there’s millions of homes installing them.
New Hampshire residential gas price seems to hover around 15-25 dollars per 1000 cubic feet, 28 cubic meters, which is about 6 cents a kWh on average. Electricity is around 16.07 c/kWh. If you get at least 40% off the electric price using heat pumps, and reduce 40% off the total energy demand using solar collectors, you’re going cheaper than gas and actually save a bit. The pumps and collectors themselves cost a couple thousand dollars.
Of course, the warmer the climate, the more you save with a heat pump, because the coefficient of power improves as the year-round difference between indoor and outdoor temperatures gets narrower. If in Sweden you get a 40% saving, in New Hampshire you may get 60% or better – who knows.
I can’t reply to your other comment about A2A heatpumps as it’s too deep, but here is an example that we are having installed in our apartment in Eastern Europe:
http://www.mitsubishipro.com/en/professional/products/heat-pump-systems/m-series-multi-zone/m-series-heat-pumps—outdoor-units-(multi-zone)/mxz-b
These split-system units are fairly popular in Europe for cooling – ducted AC is only really found in offices – and they can both cool and heat your home. The system can operate down to 5°F (-15°C) and output a maximum of 7.5kW (25500 BTU) of heat, with an input of 2.6kW of electricity. In terms of costs it’ll be around €4000 including installation and taxes, but there are cheaper (noisier, not as efficient) units for a lot less.
There’s a monitoring page set up for checking the status of the system for the Canadian community. http://dlsc.ca/
Electricity can be a an affordable option for heating if you happened to one live somewhere with mild winters and two be near a nuclear or hydro electric plant.
Still electric stoves are horrid to cook on.
In Toronto we have fairly mild winters, live near the Niagara Falls Hydro Dams and the Pickering and Darlington Nuclear Plants and we still pay some of the highest hydro (electricity) rates in the country. On yea, electric stoves do suck to cook on.
Induction is nice. Instant heat and continuously variable.
+1 for Induction cooktops. That said I still prefer gas, if it was unavailable I would be satisfied with induction.
Never understood the mentality of glass-ceramic cooktops. “Oh you know what would make this heater better? Covering it in awesome insulator!”
Induction works a treat. A roaring naked fire in my home? No thanks. We live in a seismically active area – gas piping in the house and in the neighbourhood is not appealing.
Designing and building to Passivhaus standards means very little heating is needed. Nuclear (or other) power station close by not needed.
That is the problem with your vision. The great “we”. That and thinking CO2 is a problem. Plus once you cook on gas you never go back, though I suppose a hydrogen gas range is OK if you can deliver on it.
The great Emile Zola died of CO poisoning. Some said he insisted on closing the window while using the little coal or charcoal heater in his room in the winter. Others say his chimney was blocked and it was murder. He made many enemies during the Dreyfus affair.
Would you need a detector in a house with no gas?
-“The great ‘we’. That and thinking CO2 is a problem.”
That’s almost straight from the objectivist phrasebook. Ayn Rand never did manage to solve the issue of the tragedy of the commons in any practical way, so her followers typically resort to denying there is any tragedy to avoid dealing with it – to avoid such ridiculousness as “air should be privately owned”.
When the sum of individual actions of self-serving individuals leads to adverse collective consequences that cannot be effectively solved by everyone acting more selfishly… why, that is impossible! There is no such thing, and if there is then it must be a good thing, or something you simply cannot avoid.
It’s a surprisingly common sentiment, whether it’s about gun rights or climate change. People are getting murdered in the streets because of the unchecked proliferation of guns – “that’s the price of freedom”. Sea levels rising and climates slowly turning hostile – “that’s just a leftist conspiracy”.
I can only comment on Massachusetts, but yes. we have an all electric house (with a later addition of a pellet stove), and I still need a CO detector on every living floor (even without the pellet stove). This is only checked at the sale of the house though.
“Once you cook on gas you never go back”. Disagree – I wouldn’t touch gas again with the proverbial barge pole, not since trying out induction.
Have you ever tried to stir-fry in a wok on an electric element? It just doesn’t work.
Yes it does. It just depends on how much power you got. Some wheezy 1200 Watt unit isn’t going to cut it, but they come in up to 7 kW
I’ve used gas and electric. I will never go back to electric. I had to run a gas line when we moved last year as my wife refused to go electric.
Quote: “This is an issue that shouldn’t exist, because we shouldn’t be burning gas for heat and hot water in homes. Domestic and business space heating, along with cooking on gas, is responsible for roughly a third of all our CO2 output. … ”
Maybe so. But maybe you missed the point that until we have reached the abstract goal of the “Kindom Come of the CO2 Religion”, there will be accidents with CO. It’s about real people’s health and lives *today*, right here amongst us. If it makes you feel better: in a neighboring town we had an accident where some dumbass parent used a charcoal (CO2 neutral heating source) BBQ as a space heater – 5 kids of a birthday party had to be airlifted to hospitals with CO poisoning … This article is about the prevention of those tragedies, hijacking it to vent your personal ecophobia is understandable (as all phobias lead to loss of perspective) but also distracts from the key topic of the article – which is unfair to those whose health we would like to protect.
A solution to a problem is to remove the problem.
It’s not an argument against all the other solutions.
Your ideas doesn’t eliminate CO and they would transfer the modern world to the stone age (but without fire).
Besides you should be aware that there are plenty of places that doesn’t use gas for heating/processing but still being at a risk for CO poisoning…
Eliminating the need to burn stuff in a house – gas or otherwise – does eliminate the possibility of CO poisoning in that house. It would be pretty difficult to get CO in there otherwise.
In places where there’s risk for CO poisoning still remains, install CO meters.
As for the stone-age comment – solar and wind energy are cheap if utilized correctly. Cheaper than fossil fuels. Even if you don’t believe in the climate change and CO2 link – great – using renewable energy to heat homes just means you get an over-abundance of cheap gas to use elsewhere. You could drive your car on it for example.
Where did you get your 1/3 CO2 output data from? The US EPA estimates put it around 10%. https://www3.epa.gov/climatechange/ghgemissions/gases/co2.html
I agree that there are better ways but I also live in New Hampshire like the other commenter. A centralized system only works if the town\city is centralized. I don’t have town water, gas, or sewage. Hard to imagine them pumping heat to my house. I would prefer a decentralized system.
That’s a slightly different metric, since it separates the emissions by sector, and not by what goes into heating and hot water or cooking. For example, much of the energy in industry goes into keeping factories and warehouses warm and ventilated.
http://www.documents.dgs.ca.gov/dgs/pio/facts/LA%20workshop/climate.pdf
“Buildings Account for 39% of CO2 emissions in the United States
The commercial and residential building sector accounts for 39% of carbon dioxide (CO2) emissions in the United States per year, more than any other sector.”
Actually using gas for heating is efficient much more so then electricity even when using a heat pump as heat pumps loose efficiency below 5C or 41F and are pretty much resistance electrical heating units at -10F or -23C.
That’s hardly an issue, since most of the year the temperature is not anywhere near -10F. Most of the time the pumps work very efficiently.
The use of heat pumps and storage boilers would enable the use of more renewable energy in the grid by providing a place to put it.
The major reason why things like wind energy are so difficult to utilize is because they push other powerplants offline – the power has to be used when it comes, or it’s simply thrown away, so everyone else has to yield. Wind power causes drops in the wholesale price of electricity due to oversupply when it’s forced into the grid, and then a corresponding increase in prices due to undersupply when the turbines go offline again and the other producers return, with an overall effect that increases the average electricity prices because it makes the other powerplants more costly to operate.
The base cost of on-shore wind for example is about 5-8 cents a kWh which is competitive with conventional power. It’s the transmission that is costly because of the see-saw effect with all the other powerplants. If there were a large number of heating systems ready to accept the power whenever available, the transmission cost would drop, and the heat pumps would multiply the effective amount of energy you get at least 2-3x which would result in a cost that is competetive with heating on gas – with no CO2 output.
Plus the fact that wind turbines tend to produce about 30% more energy in the winter due to denser air, which corresponds well with heating demands.
Hot water is a cheap energy storage medium. At 4.19 kJ per kg per Kelvin, a cubic meter of water going from 25 to 90 C has a storage capacity of 75 kWh. The same amount of lithium batteries costs you the price of a new car – the water just cents.
At constant pressure, liquid water is second only to ammonia in terms of the highest specific heat capacity [4.1814 kJ/(kg*C) at 100 kPa (~sea level), constant pressure and 25 deg. C]. Fortunately, storing the energy in heated water is fairly simple and efficient [e.g. a vacuum insulated container]. Unfortunately, rapid and efficient conversion of the stored energy in heated water is so not easy, this limits the applications.
“That’s hardly an issue, since most of the year the temperature is not anywhere near -10F.”
That’s like saying CO alarms aren’t needed because most of the time there are no CO leaks! Trust me, when it’s -10 F outside, and electricity is expensive where you live, a gas furnace is really nice!
And money is very much an important factor with renewable energy. If it wasn’t, just send those solar farms up to space. A renewable solution does no good if you can’t pay for it.
Per the Wisconsin State Climatology Office website, the average winter temperature for several areas in Wisconsin was between 1971 and 2000 was 4 °F. In the same time period, over half the state had an average of more than 30 days below 0 °F. per year, and some areas had an average of over 50 days below 0 °F. per year. Note that these numbers are averages, so some years have even more cold days. While heating with electricity works for some, the cost (for additional power generation plants, new transmission infrastructure for remote locations, and heating unit replacement) is prohibitive when considering upgrades. In new construction, perhaps, but it will take decades before a majority is reached. Additionally, you have to consider the uptime requirements. If the power goes out, people can die if everybody is heating with electricity. The electrical grid is too vulnerable to accidents and attacks to be relied on for safety-critical applications like heating in cold climates.
The heat pump still works at -10F it simply uses more electricity by running more, since the power coeffficient drops with temperature. That said, there are plenty of modern air-to-air heating units that work down to -10 F and still put out twice as much heat as the electric power in.
In other words, while a gas furnace could be used in the coldest days to save a bit of money, it just isn’t necessary.
” If the power goes out, people can die if everybody is heating with electricity. ”
Hence why the insulated storage boilers. A cubic meter of near-boiling water stores enough heat for the average house for a day, and that can be stretched by turning the thermostat down.
When assessing the affordability of electric heating with a heat pump, it’s the average cost that matters and not what it costs on the few cold days when the pump isn’t very efficient. Sure, on those days you pay more to heat, but most of the time you pay very little to heat, and in sum total you pay significantly less.
” If the power goes out, people can die if everybody is heating with electricity. ”
Besides, it’s the same problem – if the gas main gets cut in the winter, what do people do? Burn candles?
A portable generator can easily run a heat pump and heat a home for the same reason why the pump saves on energy – it pumps more heat than the electrical input, so you don’t need so much of it.
That’s an interesting thought. Just how does one objectively assess the relative efficiencies of heating when comparing such very different methods of heating? I suspect that Energy In / Heat Out is too simple a measure – maybe. As you point out, heat pumps (the air source types) don’t do well at very low temperatures – ground /water source heat pumps do a lot better but need access to the earth or a body of water, which is often not possible. The cost of heating (or cooling) is inversely proportional to energy efficiency of the building. Make the cooling/heating load small enough and the financial cost of different modes of heating/cooling becomes a small part of the discussion around the technical, societal and environmental factors.
the second law
We have a CO detector operating from 115VAC, plugged into an outlet on our main
floor hallway. Is there a practical way to test a device like this? Something short of
a small fire in the vicinity? Our smoke detector in the same hallway, always gets our
attention from extra dark toast, But other than the CO detector giving a beep or two
after a power outage and restoration, it has been quite quiet over the years. And,
how long is such a device expected to do the job? Do they have a ‘useful life’?
Thanks,
Ralph
The ones I’ve seen have all been labelled as having a 10-year service life.
I think ours has a 7 year service life…
There was a place on the back label to write the date it was put into service.
At least here in the USA, modern CO2 Detector versions (as with permanent-battery smoke alarms) have a life counter and will go into a failure/alarm mode at the end of their useful life.
Yes, you can get cans of CO test gas. Every safety device (smoke, CO, etc.) should have a date on it and manufacturers recommend replacement at least every 10 years (some as often as 5).
Every time I install something in my home I label it. Water filters, compact fluorescent bulbs, batteries on the water heater leak detector. A quick sharpie with install date makes it really easy to check when it was installed and when you want to replace it.
“Every time I install something in my home I label it. Water filters, compact fluorescent bulbs, batteries on the water heater leak detector. A quick sharpie with install date makes it really easy to check when it was installed and when you want to replace it.”
This is also a great way to help identify shoddy compact fluorescent bulb manufacturers.
Dang it, I’ve neglected to label the cat. No telling how long he’s been here… or how long he’s got left.
On one of my CO detectors the instructions said the smoke from the end of a lit cigarette could be used to test it. No good to me, as I wouldn’t want to light a cigarette indoors, but of some use to some people.
Ralph Irish, I test my CO meter by sticking it near my car exhaust. I don’t know if this damage the sensors and it’s easy because I have a battery unit, but I’m sure you can figure a way to do something similar with yours if it’s plugged into AC with a regular cord (if not, I don’t know what to do.)
Don’t put your CO detector near a car exhaust. That’s the worst thing you can do. There are chemicals in car gasoline which can damage the detector.
I’ve had CO detectors in my home for at least the last 15 years. A few years back a detector saved the lived of my wife’s parents when CO built up in their home after they had spent the better part of the day cooking on it. I’m a believer.
As an interesting side note: Being an electronics nut, I did a tear down on an out-of-date detector and was pleasantly surprised to find an off-the-shelf PIC micro running the show.
Oh, and I forgot to say: Excellent article about a topic which needs more public awareness. Thank you!!!
Thanks! Hopefully I get some people thinking about it.
Indeed, many thanks for this!
Yeah, the one I tore down also had a PIC
Microchip has a few dedicated parts for smoke detectors, like the RE46C129. They’ve always been a good choice for low power “runs for years on a couple of batteries” devices.
It’s annoying how most carbon monoxide alarms artificially show 0 ppm when the level measured is below 30 ppm. So one cannot easily detect small, continuous leaks.
FWIW an Air Quality Egg doesn’t truncate information in that way, and reports data to the internet several times a minute, so you can download and look at actual time series measurements.
I have one beside my gas stove, and occasionally it will read 3-5ppm, so they do exist, mine is a first alert brand iirc. I know it shouldnt be right next to the stove, but its the only outlet available and it doesnt nuisance trip so it wont hurt.
In Ontario we must have one detector on each level that has bedrooms. However I out of an abundance of caution have combination smoke/co detectors, as well as CO detectors in garage (ev only), laundry room (gas dryer), kitchen and furnace room.
So far I’ve never had one go off, but some family friends nearly died from CO poisoning and only escaped when an alarm clock went off arbitrarily and they noticed they felt strange and called 911. Dont mess with this stuff, for 50$ it could save your life, or your familes.
My wife is more sensitive then our CO detectors. At our old house we had oil heat. The front cover over the burner was ever so slightly loose from thermal cycling. She got headaches and we noticed they went away if we left the house or the heat was off. I tightened the cover and all was well. The CO alarm never alerted. About 2 years later the same cover was loose again she got headaches immediately.
When you say not easy to detect small leaks your absolutely correct. It was by luck we coralated the headaches to the heat. Everyone else in the house always felt fine.
Indeed… I’ve wondered about this with both of ours. A true zero measurement 100% of the time is quite unlikely, and yet it’s never move upward of zero. Hmmm…
also never burn charcoal in the house as a heating device either
My hat to you Sir, a very good article!
Thank you.
Adam, really well done! You’ve successfully scared the crap out of me.Time to test our CO detectors, and to remember to make sure we set my daughter up with one when she gets her first apartment.
Glad to hear it – I kept scaring myself while reading up on this stuff.
They’re a fantastic housewarming gift (though including a bottle of wine would probably make the gift slightly more socially acceptable in most cases). Either way it’s a gift that’s sure to be remembered, and likely to be appreciated as well!
So I read this carefully. Now I see some parallels between the late effects of CO ans multiple sclerosis. How many MS patients do not have MS but had too much CO?
There’s a similiar argument with Alzheimer’s as well in elderly people, because the offical salt intake recommendations are based on the idea that one should take as little salt as possible, basically teetering on the brink of hyponatremia.
So many of the elderly in care homes are fed diets that have too little salt, get chronic hyponatremia, and as a result develop symptoms resembling Alzheimer’s disease: memory loss, mood swings, disorientation, gait problems, balance problems… etc.
Bullshit! The level of salt that is required for a healthy living is extremely small compared what most people consume and even lower than the recommended amount. Science and research – ever heard of it?
Malnutrition is a problem for elders though. Salt deficiency? Nope.
http://www.aafp.org/afp/2000/0615/p3623.html
“Cross-sectional studies suggest that hyponatremia may be present in 15 to 18 percent of patients in chronic care facilities.2 A 12-month longitudinal study showed that more than 50 percent of nursing home residents had at least one episode of hyponatremia.”
Here’s the controversy:
http://www.medicaldaily.com/cdc-salt-intake-guidelines-were-wrong-most-people-no-great-benefit-dramatically-reducing-dietary
“A study commissioned by the Centers for Disease Control and Prevention (CDC) — performed by the Institute of Medicine of the National Academies — has found that there is no benefit of reducing salt intake to below 2,300 mg per day.”
“The national dietary guidelines previously suggested to stay around 1,500 milligrams of sodium a day — a little more than half a teaspoon of salt”
” In a 2011 study, 28,800 subjects with high blood pressure ages 55 and older were analyzed for almost five years. Their risk of heart attacks, strokes, congestive heart failure, and death from heart disease increased significantly when they consumed more than 7,000 milligrams of sodium a day or consumed less than 3,000 milligrams of sodium a day.”
Too low sodium intakes actually increased the risk of health complications.
0%? Your suggestion is utterly ridiculous and would (if it wasn’t) imply that science couldn’t work.
Beware of some cheap CO2 detectors on ebay. Often with no brand name they have been shown not to meet European safety levels. EN 50291 should be listed on the unit for European users. See here for a list of failed units:
http://www.carbonmonoxide.ie/htm/co_alarms.htm
Also note if you have a gas heater or cooker there is also the risk of a gas leak killing you. Gas detectors are a different device. A leak inside a gas appliance may not trigger a detector mounted outside, but may pose an explosion risk. Normally for mains gas you need to mount the gas leak detector high while for bottled gas mount the detector low.
Wow, both of my CO detectors (two different models) are on that list. And bought locally, not from eBay. Time to return..
Dan – you’re right! Check out my .io project, where I test a no name against a name brand:
https://hackaday.io/project/11555-testing-carbon-monoxide-detectors
As it turned out, in my test the no-name brand went into alarm faster than the name brand. But looking at the sensor and overall build quality of the no-name unit, I really don’t trust it over the long haul.
Another often overlooked place a CO monitor should be installed (flammable gas too) is in recreational vehicles!
If it has an engine or any combustion going on inside, plop down a 12V powered CO/CNG detector (the combo is a nice tidy package). I threw one in my RV because it’s old and was pleasantly surprised to find CO was not building up inside when driving down the road. I know this was a legitimate issue with leaky school bus back emergency exit doors and exhaust gases looping back into the cabin.
Folks with aircooled VW’s are also familiar with heater box leaks (they use exhaust pipes with heat exchangers to heat the cabin)…get a pinhole in that and life is not so carefree.
In a RV the silent killer is the multi powered fridge or blown air gas heating systems + fires. I fitted a unknown name brand 3-in-1 fridge to replace my original 3-in-1 ammonia electrolux fridge in the camper van and it sets off the CO2 alarm we have installed because we’re responsible parents when we left it running on gas unattended as a test. If I had of just screwed it in and gone out camping like a lot of people I know would do, I & entire family may have been dead by the am.
I tried to return it, but the company selling them stated that it noted in the small print that it was only certified for use on 220v and 12v power sources as the UL markings didnt cover the gas side, therefore I could obtain no refund for a unit that could kill.
I have to take it out, & replace it with a genuine electrolux unit at my cost, because I have a expectation a real name firm will actually care about dead buyers. I’ll still test it with the CO2 alarm in a dry run first though.
I dont need a smartphone co2 monitor. The battery powered one on the celing of the camper is just fine thanks. We have a second battery powered one we can take out with us if we’re staying in a motel or similar.
Same here I’d rather have a stand alone device too many things have to be smart phone enabled these days and it usually causes more problems then it solves.
@Adam When you wrote:
“The much hyped sensor suite on the Apple Watch is capable of measuring oxygen saturation in the wearer’s blood. Adapting this sensor to measure carbon monoxide saturation is a matter of adding a couple of LEDs and software.”
you seem to insinuate a technology not based on chemical cells, but one similar to oxygen saturation measurement of the bloodstream. I know how to use a search engine, but I was wondering if perhaps you found detailed or just interesting sites/papers/articles on how that works for measuring CO in the bloodstream? It seems like a more direct way (less false positives) if the bloodstream is the source.
And about:
“They also require periodic calibration and bump testing, which means either investing in an expensive calibration gas system, or sending the sensor back to the manufacturer and paying a fee. Much of this is required because electrochemical cells don’t age well. Their response decreases over time. The cells can also be damaged if exposed to chemicals and certain solvents.”
It would seem more reliable to use https://en.wikipedia.org/wiki/Photoacoustic_spectroscopy than chemical cells. Although I wonder to what extent it can be miniaturized, made power efficient (but what would be more expensive: the periodic calibration price, or “price of energy” X “difference in energy consumed during a calibration interval”?)
I’m talking about CO-oximeter devices like this one: https://en.wikipedia.org/wiki/CO-oximeter
You can also measure CO in exhaled breath: https://en.wikipedia.org/wiki/Breath_carbon_monoxide.
The one issue with this as an early detection device is that a person is already in the process of being poisoned if their levels are rising.
Also, I remember hearing that CO dissolved in ice, in a bucket could be used as a murder weapon in a closed room, what remains is a bucket of water…
Implausible, since CO or CO2 dissolved in water lowers its freezing point dramatically, and you need tremendous pressures to dissolve more than about 2 times the volume of the water in there. A 10 liter bucket of CO in a sealed room with no air leaks, say a volume of around 30 m^3 would be about 333 PPM which is harmful but not yet deadly. Any air leaks would put the concetration further down as a bucket of ice would take many hours to melt.
Perhaps if you put the person in a broom closet with the bucket and duck-taped the door to seal it. Even then the person would probably get pissed off about the rising CO2 levels and kick the door out before the bucket would melt.
Thanks for pointing out, again, I didn’t claim the veracity of what I heard. Looking up alternatives … just kidding…
CO2 concetrates on the floor, when cool and doesnt dissolve with air. It takes a while until it dissolves. So the concentration is not equal at any level. When someone lies on the floor the co2 concentration could be lethal.
Living in South Florida home CO poisoning is not a real issue except when people use grills inside during a hurricane or run a generator in a garage.
It seem to me that for cars the solution or at least a strategy would be to add a CO alarm to the car that would open the windows when it detected a high level of CO.
For the home having the alarm activate a ventilation system as well as sounding an alarm might be a good idea as well as shutting down all CO producing devices.
I’d rather have a detector with it’s own display then one that depends on some stupid phone app to tell me what’s going on.
Ohh I saw that one! If the power goes out and the generator starts to gas the house for some reason our lives would depend on wifi working correctly!
if the amount of CO that can cause damage is so low, wouldn’t it make sense to deploy it in living areas or include it as a module in a standard smoke detector? While combustion is the most likely source of the gas, can you tell me that every open area in a home is CO free if it has no combustion sources?
That hotels globally do not have mandates to have these sensors deployed is insane. You’d think from an insurance standpoint they would have had them years ago.
35 PPM isn’t actually deadly. 800 PPM starts to become acutely dangerous. 1,600 PPM kills you within two hours.
Our CO / Natural Gas detector (dual sensor) instructions (IIRC) that CO (or was it the NG?) detectors work better closer to the floor.
Petroleum gases are all heavier than air. They’ll collect in the lowest parts of a room. The instructions were probably for the NG side of the sensor.
That’s for the natural gas. CO is close enough in density to air that location of the sensor isn’t important – it mixes freely with air.
As I mentioned above gas leak detectors for mains gas need to be mounted high, for bottled gas mounted low. CO does not matter
I remember from college biology class reading how CO affects the body.
The cell membrane has a “chemical staircase” which transfers Oxygen through the cell wall to the interior of the cell.
At one of those “steps” CO breaks the process.
I always thought it was the binding of hemoglobin to CO instead of oxygen whenever CO is available… so there are bottlenecks for oxygen when CO is present,hmmm
CO2/CO and gas detectors are only needed if your home or office/workplace uses any kind of combustion for heating, cooking or industrial processes. If it’s all electric or uses other non-fire sources for things that need heat – no need for those detectors.
But of course there are clueless idiots in government that have passed laws in some states that require the detectors *everywhere*. Want a detector? Call your local fire department. Some of them have low cost or free ones.
Same goes for stupidity on heat pump technology. In Idaho there’s no difference for installing an air to air or open loop water to air heat pump. But if you want to install a closed loop water to air heat pump, that gets into permits and other folderol. Open loop runs the water through and dumps it, on the ground, perhaps down a storm drain or into the sewer – where it puts more load on the sewer treatment system. What about the water source? City water costs money (most places). If you have a well then that’s running the water pump more.
Closed loop gets filled once and re-runs it forever. Why does the non-wasteful type get saddled with more rules and regulations?
Many homes that don’t use carbon base fuel for cooking, space/water heating still contain carbon based fuels that can be the source of slow burning CO producing fire ignited by wiring fault. A CO detector would be a back up to smoke detector. In the event your close loop system is using the same water that the source of my potable water; yea I want you have to obtain permits and meet good construction standards, just like I’d have to obtain permits and meet standards to tap that potable water source. Ground water doesn’t abide by surface boundaries, so I don’t get bent out of shape about fair regulation, but I expect everyone using a common resource to be subject to the same regulation. Sorry idiots exist outside of government as well. My bad Hackaday, but I couldn’t resist pointing out the obvious.
Would a pulse oximeter detect CO poisoning? If so, a very convenient wearable could be made or added to a watch.
I’ve read that the answer is no for the cheaper ones. The medical staff quality ones read multiple wavelengths so yes. Problem is that diagnosis of CO poisoning means you’re already in trouble. Good idea though; some detectors read through chemicals that change color. Look up ‘CO detector patches’ I would link but can’t for SR. They have a very short shelf life; jogging past a lawnmower would probably set it off.
Great article. People need more reminders about the dangers.
I don’t think that would be a very reliable solution. Often nurses have to futz with them to get them positioned to get a reading. I wouldn’t trust one to stay in position overnight.
Thank you so much, Adam, for helping to raise awareness about CO poisoning, a very serious and under-reported, underestimated public health problem. (FYI: For the few states that require CO detectors in hospitals, nursing homes, motels and hotels, most do not require a CO detector in each bedrooml, as federal laws require a smoke detector in each bedroom. Usually hospitals, hotels, etc. may put one CO detector in each hallway, which is ridiculously inadequate. Pack and use your own battery-operated CO detectors, people.)
CO poisoning causes deaths, illness, and, as in my family with my sister, very severe brain injuries. All of our lives were changed forever.
Please scrupulously inspect and maintain ALL fuel burning appliances and vehicles (including electric ones due to smoldering fires caused by faulty wiring, which have also caused CO poisoning) and please use multiple CO detectors in your home and take a battery-operated CO detector with you every time you sleep or spend a night away from home. See this primer on CO poisoning for more sources of CO poisoning that you may not be aware of and how to protect yourself and your loved ones: http://www.lyrysasmith.com/can-prevent-carbon-monoxide-poisoning/
CO poisoning is the brain injury and silent invisible killer that we can absolutely avoid. Thanks for caring, everyone.
Take care.