Before the SARS-CoV-2 pandemic took hold, few people were aware of the existence of mRNA vaccines. Yet after months of vaccinations from Moderna and BioNTech and clear indications of robust protection to millions of people, it now seems hard to imagine a world without mRNA vaccine technology, especially as more traditional vaccines seem to falter against the new COVID-19 variants and the ravages of so-called ‘Long COVID’ become more apparent.
Yet, it wasn’t that long ago that Moderna and BioNTech were merely a bunch of start-ups, trying to develop profitable therapies for a variety of diseases, using the brand-new and largely unproven field of RNA therapeutics. Although the use of mRNA in particular for treatments has been investigated since 1989, even as recently as 2017 there were still many questions about safe and effective ways to deliver mRNA into cells, as per Khalid A. Hajj et al.
Clearly those issues have been resolved now in 2021, which makes one wonder about the other exciting possibilities that mRNA delivery offers, from vaccines for malaria, cancer, HIV, as well as curing autoimmune diseases. How did the field of mRNA vaccines develop so quickly, and what can we expect to see the coming years?
Vaccines in a Nutshell
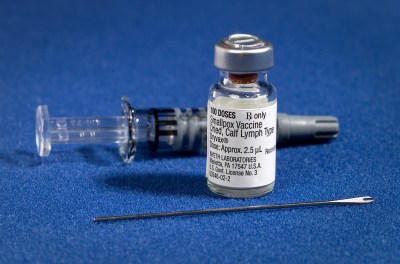
Before there were vaccines, the practice of inoculating was quite common, especially in China, where references dating back to at least the 15th century have been found. Focusing mostly on smallpox, this so-called variolation (smallpox inoculation, derived from the name of smallpox, Variola) involved taking live pathogen from a smallpox patient and introducing it into someone without pre-existing immunity to the disease.
When Edward Jenner popularized the practice of using cowpox instead of smallpox in the 18th century to induce immunity against the latter, this was the beginning of vaccination (Latin vaccinus, ‘of or from the cow’). Unlike inoculation, vaccination seeks to use a heterotypical pathogen (like cowpox), inactivated or weakened pathogens, or specific elements (units) of a pathogen which can induce immunity against that pathogens.
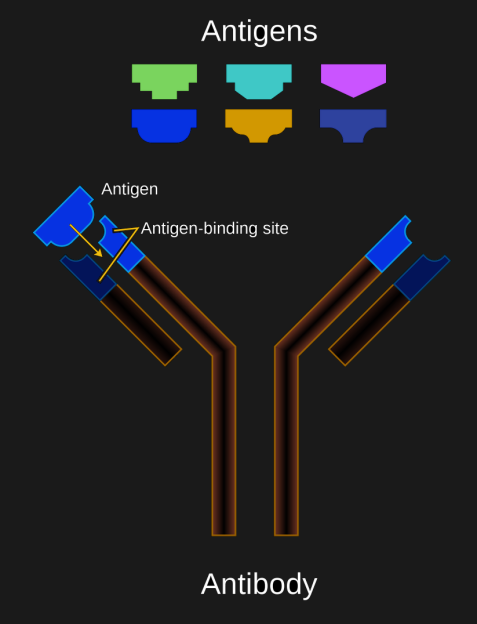
At the core of the human immune system lies the concept of antigens, which are specific proteins or other elements of a pathogen or toxin which the immune system can recognize using antibodies. These antibodies are Y-shaped proteins that can bind to specific antigens. They can be either attached to B cells (B cell receptors, BCRs), or free-floating in extracellular fluids (e.g. blood plasma).
With natural immunity from infection by a pathogen, the body’s adaptive immune system usually learns to recognize the foreign antigens and deal with the threat. The use of a vaccine is to present the immune system with these antigens (e.g. the SARS-CoV-2‘s distinctive spike protein) so that the immune system (specifically B cells) gets a chance to memorize it so that it can respond much sooner to an infection.
Vaccines Without the Complexity
As the main point of a vaccine is to present the immune system with the most effective antigens, this has led to a wide range of vaccine types over the years that seek to optimize the immune response. Here the rapid development of SARS-CoV-2 vaccines has been highly instrumental in illustrating the state of the art. Essentially, just two types of vaccines were able to be developed in the minimal amount of time available: viral vector and mRNA vaccines.
Both of these are new types of vaccines and neither have been deployed on this scale. Where they differ from other types of vaccines is that they do not require that the pathogen is grown outside of the body, but instead they focus on delivering genetic material (mRNA) into cells where these can be translated by the cell’s ribosomes into the proteins that form the target antigen.
In theory, this would skip a number of complexities, as it only requires that enough of the RNA strings are produced and inserted into the body’s cells, rather than needing to synthesize the target antigens (proteins) and inject those. In the case of traditional vaccines which use whole pathogens, the use of RNA skips the risk of accidental outbreaks caused by these live pathogens, including attenuated strains as seen commonly with polio vaccines (vaccine-induced polio).
For all their similarities, however, mRNA vaccines have a major advantage over viral vectors: the use of a lipid nanoparticle-based (LNP) delivery system which stabilizes the fragile mRNA string. This was first used with Patisiran, an RNA therapeutics medication that uses siRNA (gene-silencing RNA) to treat polyneuropathy. Through careful design of these LNPs, they are effectively as unremarkable to the immune system as the (lipid-based) membranes of the body’s own cells.
Since viral vectors by definition use viruses for breaking-and-entering into cells, these viruses are highly likely to be detected by the immune system, and some may have antigens which are already known to the body. An mRNA vaccine’s LNP coating makes it comparatively stealthy. Another major issue with viral vectors is that they require modification of the viral RNA, inserting the RNA string that needs to be carried as payload, while ensuring that these genetic modifications were done correctly.
Spiking Those Cells
Once an mRNA vaccine is injected into the body, the vast majority of the LNP-coated particles are absorbed by dendritic cells, which roam the extracellular space and can be regarded as the sentinels of the immune system. Within the dendritic cell the mRNA string is released, where it is ultimately processed by the ribosomes as any other mRNA string that originates from the cell’s nucleus. This results in the production of e.g. the SARS-CoV-2 spike protein, which the dendritic cell will then expose on its membrane to B and T cells.
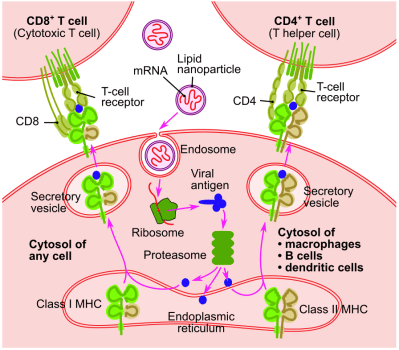
Important to note here is that these are not the exact same mRNA strings that would originate from the nucleus, as the cell’s defensive systems monitor the intrusion of foreign RNA. This is circumvented through nucleoside-modified mRNA (modRNA), in which nucleosides are swapped for synthetic analogs. This does not affect the handling by the ribosomes. In the case of CureVac‘s SARS-CoV-2 mRNA vaccine, the use of unmodified mRNA strings is likely the reason why in trials it showed very low efficacy that currently prevent it from being a viable vaccine.
Based on the data, especially from the Moderna and BioNTech-Pfizer SARS-CoV-2 mRNA vaccines, we can deduce that the combination of the LNP coating and modRNA is what makes it as effective as it is. Another interesting recent finding by Jackson S. Turner et al. was that these mRNA vaccines also trigger B cells into forming germinal centers, which are effectively training grounds for B cells that promote a long-term memorization and thus immunity to a specific antigen.
Addressing Autoimmune Conditions
When it comes to disadvantages of an adaptive immune system, one can definitely point to autoimmune conditions. These involve T and B cells produced by the body which react to self-proteins, meaning the proteins that normally make up one’s own body. Autoimmune conditions range from type 1 diabetes to lupus, each involving a specific type or types of self-proteins that are attacked by our own immune systems.
Normally, after T and B cells are formed in the bone marrow, their cell receptors are randomly arranged through V(D)J recombination to give them the widest possible range. Central tolerance — negative selection of T and B cells — exists to tests each of these new lymphocytes to see whether they self-react and thus are likely to be involved in autoimmunity. Any T or B lymphocyte which responds to the presented self-proteins are terminated via apoptosis (programmed cell death) or are reprogrammed to not self-react any longer.
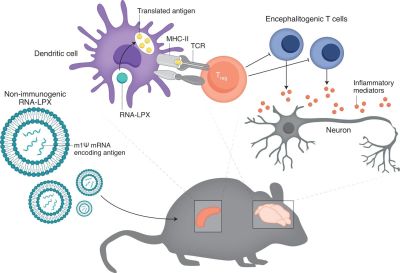
Although central tolerance, along with peripheral tolerance exist to prevent the possibility of autoimmune disorders while giving the best possible protection against new pathogens, there are many factors which can derail this, including a mutation in the autoimmune regulator gene (AIRE) and in the case of type 1 diabetes a mutation in the INS (insulin) gene.
Christine M. Wardell et al. describe a study by Christina Krienke et al. of a treatment for multiple sclerosis using a tolerizing vaccination approach. In mice, this treatment essentially cured an MS-like condition in the mice that were injected with this vaccine. Through a process of what could be called exposure therapy, the autoimmune response is downregulated and might conceivably be reset.
Related to this research is that of reducing or eliminating the immune response to harmless allergens such as pollen. As detailed in e.g. a literature review by Sandra Scheiblhofer et al. from 2018, which references both human and animal studies. These studies show that mRNA vaccines could hold the promise of a life-long reprieve from allergies as well.
Vaccine-To-Go
The good news around mRNA-based vaccines and RNA therapeutics is very hard to ignore at this point. The effectiveness of being able to essentially print strings of modRNA for a desired protein is hard to underestimate, along with the implications this has for e.g. cancer therapy. Both BioNTech and Moderna are involved in clinical trials that seek to assess whether customized mRNA vaccines that target the specific neoepitope antigens of a patient’s cancer can provide an effective therapy if not a cure for various types of cancers.
A recent paper by Katherine L. Mallory et al. covers a potential mRNA-based malaria vaccine. Encoding the PfCSP coat protein of the malaria parasite, initial findings show a sterilizing (i.e. no potential for transmission to other hosts) protection against the disease. If an effective vaccine could be developed based on this, it could save over 405,000 deaths yearly and countless millions of malaria cases.
In another paper that was published this year by Zekun Mu et al., the progress and future paths of an HIV mRNA vaccine are explored. Such vaccines would provide a layered response, initially eliminating as many of the HIV pathogens as possible, followed by a secondary, cytotoxic response that seeks to eliminate any cells which have become reservoirs for HIV. While still early days, there is a lot of promise here too.
The Holy Grail of Immunotherapy
Probably the most attractive aspect of RNA therapeutics is that it forms essentially the best form of immunotherapy, by working directly with the immune system. By reinforcing or silencing gene transcription, certain deficiencies and excesses can be directly addressed, while mRNA vaccines seem to hold the promise of both up- and downregulating the immune response.
In this way it works in harmony with the body’s natural systems, while having the power to provide corrections and information as necessary. Simultaneously, considering the ease and relatively low cost of customized mRNA vaccines, this might mean that even orphan diseases might finally get some much-needed attention. This might make RNA therapeutics and mRNA vaccines probably one of the biggest medical revolutions since the discovery of antibiotics.
Although there were concerns last year about the ultra-cold storage requirements for mRNA vaccines, as we learn more about mRNA vaccines and their stability over time, we now know that we can forget about them in the back of a regular refrigerator for a month without any worries. This should help with making mRNA vaccines and RNA therapeutics the norm over the coming years.
(Heading image: Schematic working of mRNA translation by a ribosome with help from tRNA)
















I very much appreciate such a concise summation of mRNA delivery of synthetic spike protein therapy, and in an ideal world it is a technology that would work as advertised. Unfortunately there are potential problems with it, i.e. the proteins not staying stuck to the cells and entering the blood stream, which they are not supposed to do, serious potential problems with the reproductive system and child birth, etc. VAERS reporting is dubious and there are too many gate keepers to problems being documented properly. For the MSM to call it “safe” is misleading as there simply isn’t enough of a track record to make such a claim. Until some time has gone by I’m simply not willing to be test subject #n.
But again, thank you. I’ve printed this out and it’s in my big file ‘o covid stuff!
It’s literally the safest vaccine humans have ever made. Seriously, every vaccine has side effects which is why we have programs to help those who are injured by vaccines. Additionally, in all prior vaccines, the long-term side effects have shown within two months. You are a fool for thinking you would be a “test subject” because it’s been proven to be safe as it’s been used on hundreds of millions without issue.
I think an interesting thing to keep in mind is that the covid vaccines have shown a much lower risk from side effects than the risks of contracting the virus if unvaccinated. There’s a concise and informative discussion of this available from Johns Hopkins:
https://www.hopkinsmedicine.org/health/conditions-and-diseases/coronavirus/is-the-covid19-vaccine-safe
By your logic, you would never have deployed the smallpox vaccine, because by itself it did not eliminate the pathogen.
But, with a suitable percentage of the population vaccinated, smallpox was eventually eradicated.
You are arguing for an unachievable minimum for any therapeutic intervention, which, in practice, means no intervention.
You cannot know that; literally or figuratively or in any other way. Perhaps you have hear the phrase that “Nine woman cannot make a baby in one month”? But using this logic 24 million woman could make a baby in 1 second. For example it is know that this spike protein tends to concentrate in the ovaries. It was not supposed to do that, but there it is. Knowing this, please tell me what percent of, say, 12 year old girls (who would otherwise have approximately zero problems with COVID-19 itself) will have problems related to having had the vaccine in the next 30 years? I literally don’t know; and neither does anyone else.
So far there’s no evidence for or against side-effects years from now.
I am more concerned of the long-term effects of contracting COVID-19. The severity of the illness varies widely from person to person, and we know the more recent variants have been making young people sicker than the original strain. I consider this a bigger risk than the possible side effects of vaccines.
I wish it were possible to know the future, but without that foreknowledge we need to use the tools science has provided to make the best choices possible.
>Would you stick a USB stick into your computer that you found on the ground?
Funny thing is that the mRNA works like putting up the plans for fake virus spikes on USB sticks and placing them in parking lot of a protein factory.
@tekkieneet I can’t tell if you’re trolling, but either way you’re certainly not helping.
You seem to be referring to part of a table from a pdf supposedly leaked from the European medicines regulator. The pdf relates to a pharmacokinetic study of the lipid nanoparticle in rodents.
The study uses doses of labelled lipid nanoparticles a few hundred times greater than used in the mRNA vaccine in humans (by body weight) and shows levels of the labelled lipid nanoparticle are greatest at the injection site, and are higher in a few other fatty tissues than the ovaries… but it seems people prefer to believe “the spike protein targets ovaries”
I mean statisticaly it would be safe to say for any 12 year old girl posting on this site obesity is more likely to have a negative effect on their reproductive system.
But I am genuinely amazed how many middle-aged over weight men are desperately concerned how this vaccine is going to affect their ability to become a mother.
Or a parent. Remember China has abandoned their One Child policy. Other nations are facing a similar problem.
Please don’t do this. Polio had 5%-10% fatality at 15 years with late effects and this is turning it on its head to spread fud. If we’re going here, you can’t know if you have it backwards and there’s a 10% death rate for people who caught C-19 15 years after the fact and you need to put a gold watch on the table if you’re going to continue to show you’re serious. I don’t have to put down anything because I’m not going to pull a card out to champion it on a repeated basis beyond diluting any notions of convoluted prescience.
VAERS keeps track of this sort of data in the US, and from Dec 14, 2020 to June 14, 2021 – 5,343 Americans died from the vaccine (0.0017% of doses administered caused death), so it is pretty “safe” considering the huge number of doses given.
https://vaers.hhs.gov/data.html
5,343 Americans died from the vaccine
“VAERS accepts reports of adverse events and reactions that occur following vaccination. Healthcare providers, vaccine manufacturers, and the public can submit reports to the system. While very important in monitoring vaccine safety, VAERS reports alone cannot be used to determine if a vaccine caused or contributed to an adverse event or illness. “
I worked for a drug testing lab. We were testing a generic equivalent of Procardia. Two weeks after the last dose one of our study participants got in a bike accident and broke his arm. Holy shit, we had to report this as an adverse event to the FDA like within the hour.
I’m pretty sure that the heart medicine didn’t make him crash his bike two weeks after it was out of his system.
Even if the number of deaths is 100% accurate in the attribution to the vaccine, I’d hazard a guess that the people were in greater peril just driving on the public roads to get to the place to take the vaccine. Remember, 809 people supposedly died in 2008 by becoming tangled in their bed sheets. Life is dangerous.
>the proteins not staying stuck to the cells and entering the blood stream, which they are not supposed to do,
You are gonna lose your MIND when you find out what a real viral infection does.
https://www.hopkinsmedicine.org/health/conditions-and-diseases/coronavirus/what-does-covid-do-to-your-blood
>In addition to the lungs, blood clots, including those associated with COVID-19, can also harm:
>The nervous system. Blood clots in the arteries leading to the brain can cause a stroke. Some previously young, healthy people who have developed COVID-19 have suffered strokes, possibly due to abnormal blood clotting.
>The kidneys. Clogging of blood vessels in the kidney with blood clots can lead to kidney failure. It can also complicate dialysis if the clots clog the filter of the machine designed to remove impurities in the blood.
>Peripheral blood vessels and “COVID toe.” Small blood clots can become lodged in tiny blood vessels. When this happens close to the skin, it can result in a rash. Some people who test positive for COVID-19 develop tiny blood clots that cause reddish or purple areas on the toes, which can itch or be painful. Sometimes called COVID toe, the rash resembles frostbite.
My own sister suffered a Covid related stroke. Now she can barely speak. Good luck explaining why you didn’t take the vaccine when you can only get 3 words out per minute.
And the COVID virus is safe?
Or the car you drive is safe?
Or that take away you are eating is safe?
Well, since contracting the virus, I’ve been having mild memory problems and I consistently smell ammonia instead of mint (which is somewhat annoying). Had a vaccine been available at the time, I wouldn’t have any of these symptoms now. You can fear about imaginary future effects of the vaccines all you want, but I’d be more worried about the very real, present, and long term effects of actually contracting the virus.
Thank you. Some people just seem to think this is “just the flu”. While in some, this is true. But others, like one of my wife’s family members, died, and another family member spent 40 days in ICU on a ventilator. Amazingly the latter recovered, but has issues with tiredness. Time will tell if he has other long-term effects.
I personally know people who have been hit by a car. It was not Holy Grail but BMW brand.
The header image hearkens back to another age. Nice touch.
What I like most about recent mRNA development is that we also came to know (https://www.pnas.org/content/118/21/e2105968118) to what extent RNA integration into the cell genome is possible: it practically does not happen, except for sporadic reverse transcription of subgenomic sequences.
In the beginning, a lot of time has been spent telling people that integration cannot happen due to virus and vaccine information being RNA-based, and I hope people will embrace the refined knowledge we now have to put that criticism to rest.
In terms of DNA-based gene therapy for humans, we were let down by the promises of CRISPR. Off-target mutations are an ongoing issue (https://www.pnas.org/content/118/22/e2004838117) and seem to prevent its use for the time being.
Given a way to test for genetic predispositions to develop type-1 diabetes, I like to think one might be able to come up with an mRNA vaccine for children that prevents its emergence later in life.
I’m looking forward to this future.
> prevent its use for the time being
use in humans, that is
viral RNA integration in the human genome is a documented fact, sit´s called endogenization.
Some estimations range from 8 to 10% of human DNA coming from viral RNA sequences.
Nope. It does not happen with your line of a mill RNA virii. You have no reverse tranacriptase anywhere in your genome, neither do they. Endogenization is driven by retrovirii (like HIV), and DNA virii which are very rare in humans.
CRISPR is a way to delete a sequence. RT + an integrase is a way to add a new sequence. Two different gene editing tools, with the latter being far more controllable.
I’ve no idea why “ModeRNA” rebranded themselves to “Moderna”. Maybe the original name was just too clever for American tastes.
“Hacking the software of life”
So we’ve been pronouncing it wrong for over a year!
This is the first I’ve heard of using mRNA vaccines to eliminate autoimmune conditions. That’s huge.
I wonder if you could temporarily tune the immune system. Have it clean up fats for a week. Or even target fats found in your arteries.
Most of your brain is fat, I would not like to see a random mutation on that one.
Is that were one gets the expression, “fathead”?
I have no idea if that is where that expression comes from. But about 60% of a human brain is composed of fatty acids, and 84% of that is omega-3 (or ~50% overall by weight).
Just wondering why this has comments turned on while the article about Hubble had them turned off? Like of the two I would think this would be the article that has comments disabled.
I think the hubble comments being closed was just a mistaken click prior to publication. They were turned on after we realized the problem. https://hackaday.com/2021/07/02/the-trouble-with-hubble-payload-computer-glitch-stops-science-at-the-space-observatory/
OK, I will google. Most google hits seem to decribe a successful polio eradication campaign, which was set back by inaccessibility of some regions due to armed conflict. There are some New Age sites claiming without evidence that the vaccine itself spreads polio–a patent nonsense.
Your disapproval of Bill Gates and Anthony Fauci is unconvincing. They are complex characters, but they both mean well and accomplished a lot of good. You are welcome.
One imoortant thing, I gather, is that mRNA is safer because it’s a fake version of the virus, rather than a sample of the virus.
So it may not actually be effective for the immunocompromised, but it won’t make things worse.
I’ve read, in the past year, that there is hope the process can be used for cancer, and HIV. But no, I’ve not seen mention of autoimmune diseases. They know a lot more, but still don’t know how they happen. Something about genetics, but not hereditary (though relatives maybe inclined to get one, but not the same disease). Why did I get to 59 before it kicked in? But I saw stories last year about two teenagers with the same rare disease.
I’m no expert, but know infinitely more than I did two years ago, but I’m trying to visualize what you would innoculate against? Autoimmune diseases means the immune system is overly active, and thus the immune system does damage. It’s within my lifetime that my disease has gone from terminal, to chronic, Dr. Fauci won an award for his work on my disease.
There’s no cure, but now it can be kept in check. An expensive drug every six moths that suppresses my immune system, so it can’t be overly active. Ironically, that makes me vulnerable to other things. And means vaccines have little or no effect.
I can’t speculate as to what your immune system is doing, but mine is itching for a fight with anything. Lacking a worthy opportunity, it is attacking me. Hopefully mRna research can solve my issue within my lifetime, since i am pretty sure i don’t get do-overs
>You would rather be locked yourself up for the next 5-10 years until it has been tested?
Why would he need to be locked up?
I’m just guessing based on what I remember about the context, but I think they meant as in a self imposed locked in (as in afraid to go out and live until it’s been tested fully and all side effects well documented). I could have misunderstood what they meant entirely though idk. And the original comment disappeared so no way recheck lol.
Maybe in a Typhoid Mary sense?
That’s what *I* meant… Not sure why my post was deleted by the mods here.
For a person who is so risk averted to not want mRNA – best vaccine, the only safe way is to lock himself/herself up 5-10 years until everything is proven to be safe.
The alternative is the traditional, but not so effective inactivated virus used in Sinovac.
Or the person is just making anti-vaccine excuses…
Or you just go trough the disease, you know like flu…
Or you could get your flu shot every year, providing a pretty good shot at immunity against that year’s big flu strains, and pretty good protection against minor mutations.
For children, Covid is probably less dangerous than the flu, for adults, there’s definitely a higher risk of death, hospitalisation, or long term illness.
Mind you, flue rates over the last year have been *tiny*. Enforcing mask use during flu season seems like a good idea.
Covid-19 isn’t like chickenpox, you’re not immune for life once you’ve had it, we can’t start giving children covid parties and think we’re doing them a favour.
A bit of a side note on one other tweak that is found in all of the successful vaccines – CureVac also used it but it alone wasn’t enough:
https://cen.acs.org/pharmaceuticals/vaccines/tiny-tweak-behind-COVID-19/98/i38 – the double proline substitution was discovered during SARS/MERS research in 2017, and was a key component of all of the successful vaccines. CureVac shows that it is, however, not enough. (Alternative terminology for the PP substitution is “pre-fusion stabilized”, which is mentioned in the CureVac article)
One of the few widely deployed vaccines that lacks this tweak is SinoVac’s product (that’s a simple inactivated virus), and that probably explains why it doesn’t seem to be performing as well in the real world.
People comment that these vaccines were developed too quickly, but people also forget that scientists have been working on SARS for nearly 20 years now, and MERS nearly as long.
I understand that. But, oh boy, did that second Pfizer shot hit me hard. I hope I never have to repeat that! My mind tells me not to assume that the side effects are the result of it being mRNA specifically, but my “don’t touch the hot stove twice” instinct tells me otherwise.
(For the record, I’m not saying that I wouldn’t take such vaccines, just that I wouldn’t look forward to taking them.)
Something got munged here. I was replying to:
Gravis says:
July 26, 2021 at 8:50 am
It’s literally the safest vaccine humans have ever made.
I had a pretty rough experience with both shots but it was nothing compared to when I actually had covid about a year ago. It wasn’t like my life was in danger or anything that serious but it was literally the worst two weeks of my life. I don’t think I’ve ever felt that sick from any other type of illness.
There is a difference between unsafe and causing discomfort for a couple of days. And these mRNA vaccines seem to be quite safe. no, we do not have long term data, but there is nothing so far that suggest these vaccines cause any long term damage.
For covid we also do not have long term data. However we have found quite a few things suggesting that it can and does cause lasting damage, even after mild infections.
The reactions to the vaccine seem pretty random. I’ve had both doses of Pfizer, and apart from a sore arm I didn’t feel a thing.
The sore arm isbecause of the nee dle, and that it has to go deep. I didn’t like needles as a kid, because of the needle, not the vaccine.
I shivered a few hours after the first dose, but tyat may have been the cool rainy weather when I walked home.
The second dose nothing, even though that’s the dose I’m counting on.
Pfizer BioNTech. First jab nothing. Second jab strong chills and shivering for 10-12 hours. No fever or any other issues.
The ADDRESSING AUTOIMMUNE CONDITIONS section sounds quite interesting and made me think, if autoimmune conditions (body attacking self) can be down regulated then couldn’t a similar approach be applied to organ transplants? Using the DNA from the transplanted organ to make an mRNA “vaccine” to shut down the immune of the recipient to the transplant foreign protein, etc. If it worked, after, say six months of mRNA treatment the patient could be free from taking immune suppressing drugs, which would lead to better health and probably longer life for the patient.
Maybe. I’m aware that the expensive drug I get is also used for some types of leukemia, and specifically kidney tansplants.So I think it’s the same mechanism.
After I posted earlier, I did a search, and there’s been testing of mRNA on mice with autoimmune diseases. I don’t follow how it works, I onky glanced, but apparently it is eventually doable.
I know someone who got a transplant and they had to use steroids to knock down the immune response and it “rotted” away his hip, he needed a hip replacement, so anything that can improve anti-rejection (I’m sure you’ll agree) is good. As for how mRNA helps (mice with) autoimmune diseases I have no idea either but, thankfully for everyone, I’m not involved in the research, smart people are.
Correlation either does or does not imply causality, depending on the point of view of whoever pays most.
I gave an example on a girl longer time ago in 2017 who got sick as I did in 2018 .
Somehow It didnt get into the text .
Long story short it was a girl in europe who got sick and ended in hospital .
Final repport say lung flu .
BUTTTTTT … well it look similary too CoVid ( because it is a CoVid )
Medias is just all shut about those cases .
I forgot to say a mysterious Lung Flu started killig wapers and smokers In the US
and it started beack 4 years . Today there is NO doubt this was also THAT CoVid .
Wipe wipe under carpet , nobody notice if we doesnt speak.
Assuming you’re a US citizen your right to free speech is only protecting you from government censorship. You can still have comments removed or be banned from Internet platforms, especially when your words violate a rule of the platform.
I’m not sure what your point is, I think it’s that we should not vaccinate against any type of flu including covid as there are too many variants to cover. Except we do vaccinate against the flu, covering the most likely and deadly candidates each year as part of ‘flu season’ and ‘the flu jab’. There’s a fuss over this variant in particular and it’s easy to spread. You normally don’t get flu in summer but you can catch covid-19.
Why is easy to spread bad? We can only care for a limited number of people who get seriously ill from a flu. Let’s say it’s 1000 people. What if you’re the 1001st person to need hospital care? We can’t provide it and your chance of death goes up a lot. That’s why something with the potential to make millions of people need hospital care at the same time is a big deal and something we’re trying to vaccinate against. Not that that stops it completely, there’s still something like a 2-5% chance of needing hospitalisation even if you’re fully vaccinated. Having the disease also doesn’t stop re-infection so it’s 2-5% every time you get infected.
So you don’t want to get vaccinated. Technically that’s fine, but what if everyone decides to do the same? Each person opting out increases the chances of an overwhelming need for hospital care that can’t be met and the chances that you’re one of the people without care.
This is a very strange question. The “one in a million” stuff aims to confuse by making something look harmless.
But the essence of the question is “can people be forced to not kill other people” and the answer, in most every ethical / moral / religious system on this planet is “yes”. Yup. Sure. Absolutely. Without a doubt.
The part of the argument where the one group gets no personal benefit from not killing the other group isn’t even really relevant.
““can people be forced to not kill other people”…..Yup. Sure. Absolutely”
but if society forced several million people in group X to take a vaccine that will kill 2 in a million of them (more than the hypothetical disease would, which only kills 1 in a million), this would itself be people killing other people. The important factor in the question is that group X are more likely to die by taking the vaccine than they are by getting the hypothetical disease itself. Although by doing so this might save more people in other groups, and most people would probably agree with minimising the overall number of deaths, I don’t see this as such a straightforward ethical decision, especially if group X were much younger than the other groups.
The question is if one group can be forced to accept a treatment which does carry a cost/risk to them, to protect the other group.
Which means it’s essentially a rewording of the trolley problem.
A very clear cut case is if group X have professional obligations to some vulnerable group, i.e. group X consists of nurses or doctors or dentists, then I think it is entirely reasonable that they be mandated to undergo suitable risk reduction procedures to minimise harm for their client.
It’s not that different to expecting a health care worker to wash their hands before examining a patient.
I’d probably agree that health workers might be asked (or even required) to take a vaccine, but I don’t think this is the same as requiring that they wash their hands before examining a patient. Handwashing probably has a zero chance of causing harm to the health worker. Taking a vaccine can definitely have a non zero chance of causing harm (or even death) to the health worker. For instance, the AstraZeneca covid vaccine in the UK has been linked to several deaths caused from blood clots.
https://www.bbc.co.uk/news/health-56620646
Yes, having a vaccination is more dangerous than washing your hands, so a better comparison would be: side effects from the vaccine, vs the risk of the disease itself, and that’s no contest.
Actually, repeated hand washing (especially with alcohol and similar) does carry risks – it damages the skin, which can vary from cosmetic to painful to dangerous if it leads to cracks / cuts in the skin.
However, it’s a slow gradual risk, easy to spot, and can be mitigated with moisturiser.
While we are mentioning COVID vaccines I learned something quite interesting the other day. (apologies if you already knew this but it was news to me…)
The Oxford/AstraZeneca vaccine (which is a DNA vaccine not mRNA) has its start in a research project funded by the WHO pre-COVID19.
The idea was to be able to quickly target a new emergent disease by stuffing custom-generated ingredients into an adenovirus carrier.
Up til a few weeks ago I had no idea that the WHO was being so proactive with this stuff. Good on them!
Least someone is being proactive. The rest comes to money.
https://www.nytimes.com/2021/07/25/health/fauci-prototype-vaccines.html
Yeah, so proactive against a man made virus escaping a lab !
Interesting scenario. You could think of your hypothetical disease as the Human Papilloma Virus. It doesn’t kill men but causes cervical cancer in women. Here in Australia, we had vaccination campaign that targeted school girls with great success. Should boys get vaccinated too? Well, as the virus is generally passed via sexual transmission it would still be a good idea as this would be a second protection for girls that either can’t have the vaccine or had a dud dose.
Not much difference to the covid situation in nursing homes. It’s most likely to kill the residents due to their age and failing health so they are a priority group, however, the staff should have it too so they reduce the chance of transmission to a resident.
Finally something on vaccine research with a focus on the mRNA vaccine development, structure, mode of action, that goes into depth and answers many open questions that have been with me since being diagnosed with a chronic autoimmune disease. Why do I know more now than the specialist/doctor? Thank you for the transparency and the promising outlook regarding this achievement and its potential, which has eclipsed everything that was before– grateful that sb. shared the essay (here Tw),publicly accessible for everyone who wants to read throughoutly,to comprend and assimilate- very encouraging these days..