When you’re standing at the gas station filling up your car, watching those digits on the pump flip by can be a sobering experience. Fuel prices, especially the price of gasoline, have always been keenly watched, so it’s hard to imagine a time when gasoline was a low-value waste product. But kerosene, sold mainly for lighting, was once king of the petroleum industry, at least before the automobile came along, to the extent that the gasoline produced while refining kerosene was simply dumped into streams to get rid of it.
The modern mind perhaps shudders at the thought of an environmental crime of that magnitude, and we can’t imagine how anyone would think that was a good solution to the problem. And yet we now face much the same problem, as the increasing electrification of the world’s fleet of motor vehicles pushes down gasoline demand. To understand why this is a problem, we’ll start off by taking a look at how crude oil is formed, and how decreasing demand for gasoline may actually cause problems that we should think about before we get too far down the road.
Put a Little Plankton in Your Tank
When you fill the gas tank in your ICE or hybrid vehicle and start the engine, you’re closing a loop on chemical processes that started billions of years ago. The petrochemical fuel that powers most vehicles started as atmospheric carbon dioxide, greedily gobbled up by uncounted trillions of microscopic organisms like plankton, algae, and cyanobacteria via the process of photosynthesis, and locked into their biopolymers — lipids, carbohydrates, proteins, and nucleic acids.
These microorganisms multiplied fruitfully in the oceans and lakes of the primitive Earth, falling as sediment when they eventually died. The constant rain of death built thick layers of sediment, rich in organic molecules. Most of the carbon in these sediments decomposed via oxidation reactions, but in some areas, vast layers of the rich organic slime ended up covered by inorganic sediments thanks to geological processes. Locked away from the corrosive effects of oxygen and experiencing increasing heat and pressure thanks to the weight of the material on top of them, these partially decomposed sediments gradually transformed into kerogen, a deposit of organic material locked inside a sedimentary rock.

While kerogens are rich in large, complex hydrocarbons, and some are even rich enough to be used as fuel — coal is essentially a kerogen whose starting biomass was largely land-based plants — most kerogens don’t fully convert into petroleum. Kerogens like the shale oil fields in North America are said to be “thermally immature” — basically, they haven’t cooked enough. But other kerogens, ones that have deeper layers of sediment deposited on top of them, go through further geochemical reactions, breaking their complex hydrocarbons down into simpler and simpler compounds as time goes by, eventually forming vast pockets of liquid crude oil.
The most thermally mature products, the ones that have cooked the longest, are the short-chain gaseous hydrocarbons from the single-carbon methane to the four-carbon butane. On the other end of the thermal maturity spectrum, long-chain hydrocarbons, with perhaps 40 carbons or more in their chains, make up the thick, sticky asphalt compounds.
The hydrocarbons that are important in terms of motor fuels and lubricants tend to come from the middle of the thermal maturity range. Diesel, kerosene, and jet fuels tend to come from the C9 to C16 range, with longer carbon chains making up the heavy bunker oil used to fuel marine engines. On the shorter end, the C5 to C8 range hydrocarbons make up the bulk of gasoline, which is a complex blend of many different hydrocarbons, including straight-chain alkanes like hexane and octane, cycloalkanes, and any number of additives like ethers and alcohols, including ethanol.
Crude Estimates
It’s important to note that any crude oil deposit is going to contain a mix of hydrocarbons. Some will be richer in hydrocarbon chains of one length than another, but in general, each deposit is going to have at least some of every fraction, from methane to bitumen. What’s in there depends on the conditions the kerogen that formed the crude oil experienced, and how hydrocarbons from different sources mixed underground over time.
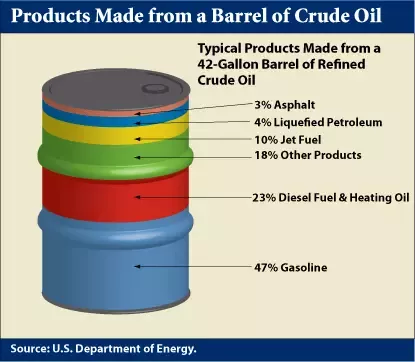 And that leads to the essential problem of petroleum extraction. While fuels like diesel and gasoline are highly engineered chemicals, they are not created from crude oil. Rather, they are refined from it. That might seem self-obvious, but it’s an important point. A typical barrel of crude oil contains about 40-50% gasoline (or more precisely, the C5 to C8 fractions that are blended into gasoline), and fully 84% of each barrel contains fuel-grade fractions, when you throw in diesel, kerosene, and bunker oil.
And that leads to the essential problem of petroleum extraction. While fuels like diesel and gasoline are highly engineered chemicals, they are not created from crude oil. Rather, they are refined from it. That might seem self-obvious, but it’s an important point. A typical barrel of crude oil contains about 40-50% gasoline (or more precisely, the C5 to C8 fractions that are blended into gasoline), and fully 84% of each barrel contains fuel-grade fractions, when you throw in diesel, kerosene, and bunker oil.
This is the root of the gasoline problem. Right now, electric vehicles make up a tiny fraction of the total fleet — perhaps 3% worldwide. But at some point, through a combination of better engineering, political pressure, improved battery technology, and climate awareness, demand for electric vehicles is going to take off in a serious way. Some estimates peg the percentage of EVs on the road in 2040 at 58%, at least for passenger vehicles. That’s a huge number of vehicles that won’t be stopping by the local gas station to fill up every couple of days, meaning demand for gasoline will necessarily plummet.
But, as we’ve seen, about half of every barrel of crude oil is gasoline. If we suddenly don’t need as much gasoline, the only way we can deal with the decreased demand is to not refine it from crude oil in the first place. That poses a problem if we still need any of the other fractions, which we likely will. Take diesel as an example. A 2019 report estimates that the medium and heavy truck fleet will only be about 4% electric by 2025, and that most of that will be trucks working local and regional delivery routes, mainly because of limitations in battery life. Long-haul trucks, though, will probably not be electrified for quite a while, meaning that crude oil will still have to be distilled to produce diesel to run them.
The same problem applies to airplanes — unless battery technology improves drastically, we’ll still need to get to the kerosene that’s in crude oil. Add in the asphalt needed for road building, the heavy bunker fuel needed to keep cargo ships running, the lubricants needed to run pretty much anything mechanical, and the petroleum distillates that go into fertilizers, pharmaceuticals, and plastics, and you’ve got a situation where everything in a barrel of oil is both useful and needed — except for gasoline, which happens to be the bulk of what’s in there.
Next Steps
And so that’s the problem: roughly half of every barrel of crude oil that we pump out of the ground is made of something that’s destined to be useless, at least in a world dominated by electric vehicles. And that wouldn’t be so bad if the other 50% of each barrel didn’t contain stuff that’s so darn useful; otherwise, we could just leave the oil in the ground and call the problem solved. That may be the eventual solution, once everything that currently burns fuel is converted to some other power source, but in the meantime, we’ll either have to figure out what to do with the waste gasoline, or find a way to get the hydrocarbons we need some other way.
Stick around for part two of this series, where we’ll dig into synthetic fuels, synthetic oils, and how perhaps we can engineer our way around the gasoline problem.
















Can the gasoline be used in some kind of thermal based powerplant, but with less pollution for the same amount of energy as in ICE automobiles?
Of course… The text is a little dramatic, oil is not designed with the specific proportions to meet our demand, on the contrary, the market adapts to the supply. If the price of gasoline falls, other uses that were previously unfeasible will emerge.
One of the big advances in oil refining was catalytic cracking and allowed them to get more gasoline and less of the heavier products. The oil industry can control (within limits) the ratios of the various products produced.
I assume the first response to reduced gasoline demand will be to adjust the process to produce less gasoline.
When the shipping industry started to phase out heavy bunker oil, the refining process was adjusted.
No. Amount of energy released in process of oxidation of gasoline depends only on gasoline composition and does not depend on the way it done. It is just an exothermic reaction and energy output is just a difference of energies of chemical bonds of initial components of reaction and final products. So, the way of burning does not change anything if reaction is the same.
Same is correct for, say popular airlines scam with offering to pay additional money for “lowering CO2 pollution” or “rising efficency” by using “more expensive” “bio” aviation kerosene. Aviation kerosene has to have strictly controlled composition regardless of its source. So kerosene made from plants, used frying oil or by usual way from fossils have absolutely same CO2 and energy output during burning. There just can’t be any “lowering CO2 pollution” or “rising efficiency” at all.
If the chemical composition of fuel is the same and reaction of burning is the same, energy and products outputs will be always the same regardless of the way you burn it or the source of fuel.
Same is with nuclear energy too – you can’t change energy output from some nuclear reaction.
Afraid you are entirely wrong with your “No.” statement. While being mostly right…
The more efficient extraction of the energy means you need to burn less of the source of it – so produce less pollution overall, and probably a different blend – burning more efficiently more NOx compounds by percentage than the burnt and unburnt carbon ones is likely. The reaction isn’t the same even though fuel is, or is similar – as you are changing the condition under which the combustion is happening – and that can have huge differences to the pollution – obvious case in point when some old car is smoking away making visibly sooty exhaust – its the same fuel that went in, but clearly large amounts of it went through that engine and did nothing…
Also adding ‘bio’ to your fuel – which often means alcohols of some sort, can in the right engine be more efficient, and efficient again means you needed less fuel for the energy you got out of it, the whole point is to change the fuel composition..
So while you are correct there is a fixed maximum energy that a fuel could deliver in all cases you have to account for how much of that energy you actually captured and the actual conditions of the reaction taking place – burning in a calorimeter with maximum oxygen to ensure full combustion (as the whole point is to measure the maximum energy potential in the fuel) is very different from burning in a piston where you only have however much of the reagents is inside and the temperature and pressure changes though the burn – and the heat is entirely waste all you wanted was the pressure differential of expanding gas.
You also have to account for how well you can clean up the exhaust – a car/aircraft has not got the volume or mass to spare to really do a good job and really clean the exhaust up, and doesn’t produce a volume of exhaust gas worth say shipping to a green house – where its suddenly a useful product – so the pollution of burning a fuel, even at the same paltry efficiency of ICE engines on the road in a big power station is going to be better – and guess what big power stations are actually vastly more efficient too! Largely for the same reason they have cleaner exhaust – they can have all the volume and mass needed to work more efficiently so get much much more of the maximum potential out of the fuel they burned!
Agreed.
It is actually more efficient to run a fuel thermal power plant to create electricity, to then charge an EV, than to use the same fuel to power an ICE car.
First, scale matters.
Second, ICE cars are incredible inefficient. An IC engine is at its peak efficiency for a very narrow window of rotation speed. That “sweetspot” is almost never met in any ICE car, because when driving (especially in a city), you are constantly speeding up and down, stopping, etc.
That’s why there are hybrid cars : the aim is to have an ICE that [almost] only and always runs at its best regime.
If your driving asks for more power, the electrical engine kicks in. If the ICE puts out too much power, this excess is stored in a battery. Once the battery is full, the ICE turns off and you drive without burning fuel.
Even with the multiple conversions ICE→Generator→battery→electrical motor, it is still much more efficient than having a direct connection between the ICE and the wheels. That’s how inefficient ICE truly are.
Not really, when you consider the embedded energy cost of the battery, the transmission losses, and the charging losses. That drops the total energy efficiency of the system quite close to your everyday car, and well below what it would be in some optimized systems like hybrids or FCEVs.
That doesn’t count the environmental cost of everything in the electric vehicle, either. Lithium ion batteries are not clean to make, especially depending on the environmental standards of their country of manufacture. Rare earth magnets, electronics, and copper certainly don’t grow on trees (or in farmers’ fields) either, RoundUp-ready or otherwise.
[Citation needed]
You got some numbers for this? Not because I’m doubting, but just b/c I’m interested in seeing how it all breaks down.
> Also adding ‘bio’ to your fuel – which often means alcohols of some sort, can in the right engine be more efficient, and efficient again means you needed less fuel for the energy you got out of it, the whole point is to change the fuel composition..
I’m afraid you could go to jail if you fill the plane with passengers with kerosene with “changed fuel composition”. Rules are really strict on that matter.
> so the pollution of burning a fuel, even at the same paltry efficiency of ICE engines on the road in a big power station is going to be better
ICE engines already burn fuel with maximum efficiency. That is why there is so-called lambda sensor and complex control electronics with complex software – to keep burning conditions as perfect as possible. I doubt that you could burn same fuel at big factory with really noticeable difference in efficiency. Even if you get some insignificant difference, it will be incompareable with losses on conversion and distribution of that energy.
Centralizing burning of fuel seems to make things worse. You concentrate pollution in single location, usually insipid with plants and do it in large quantities, instead of distributed burning in small amounts usually with plants around, add energy transportation losses and finally you get an expensive dead spot somewhere with net of wires/pipes/whatever around for miles.
If you are worrying about CO2 pollution, burning fuel is definitely not your choice. Use nuclear, hydro, tidal energy. I didn’t forget sun/wind, I just don’t count them since they are unstable and unpredictable and need an additional energy storage which is highly ineffective and expensive, especially if you want to use batteries.
ICE engines are nowhere near maximum efficiency – most of them are not even approaching half what the very best of them can do, and the best of ICE can in ideal conditions only get something like 50% of the energy out usefully – for comparison most roadgoing ICE engines are more like 20%… They have lots of fancy engine management systems now to get the best out of the poor system they have to work with, but it can’t turn a poor system into an actually good one, and you don’t pay top end race car levels of material, maintenance and productions costs for your family hatchback…
Where your big central power plant will get way way more energy out of the same fuel – as its not going to be using the horribly inefficient ICE method to get the energy out anyway – turbine are much more efficient, and even if it was using ICE engines they do change efficiency with scale and rev range, your bigger than normal road engine does have the ability to gain greater efficiency… The transmission of electricity is remarkably efficient, and conversion to anything else generally only happens at use time without the massive idling wastage of burning stuff – while there are losses in conversion and distribution of any form of energy when you get probably 4x (maybe even more) energy out of the same fuel in the big power station to burning it in all the little ICE engines you get a feel for how meaningless those losses are in comparison to the really poor initial energy gathering efficiency.
Also I did state adding ‘bio’ usually means alcohol of some sort – because it does, I made no mention of aircraft rules and regulations, because I don’t really care what they are/are not and they are liable to change over time anyway, though I was under the impression all that actually matters was water content being acceptably low and the octane rating and viscosity being correct…
> energy out usefully
What do you mean by that?
I’m talking about efficiency of burning fuel, not about efficiency of converting that energy into, say, moving of vehicle. May be you are not aware, but there are cold countries exists where heat energy of fuel burning in ICE engine is very useful too, because you will just freeze to death if your engine will convert all fuel energy to the motion. So even if accept your meaning of efficiency, ICE engines in cold countries have maximum possible efficiency – all energy of burning used completely. In the same countries, electric vehicles with batteries have the worst possible efficiency, even worse than diesel ICE.
> Where your big central power plant will get way way more energy out of the same fuel
But you can’t. It’s just a chemistry laws. You will get the same amount of energy if you fully burn same fuel, regardless where and how you do it. Is that energy useful highly depends on situation. If you need heating you will welcome high percentage of heat, if you need electricity it will depends on generator you use, TEG, MHD or alternator on turbine/engine shaft. And so on. It is highly variable and subjective thing.
> Also I did state adding ‘bio’ usually means alcohol of some sort – because it does
No. It could mean alcohol, but does not have to. You could use vegetable oil in diesel, or convert alcohol on catalyzer to gasoline . Or make more complex transformation on carbonhydrates sourced from plants to get anything you need.
Alcohol, really is worse than oil or gasoline due to lower energy density. Better convert it to gasoline-like mix of carbonhydrates on ceolites like ZSM-5 or similar before using as fuel.
No matter how you try to sell it an ICE engine is garbage – you get less useful work out of it than a big power station turbine, by a huge amount, and it never burns as effectively either – if it did you wouldn’t go round strapping stupendously expensive precious metal filled Cat converters to everything ICE!
Even in cold climates the waste heat of the engine is often lost – can’t go filling your car/home with exhaust gas if you want to live, heat exchangers are either rather bulky or pretty useless.. Most of these cold places burn fuel in specific cabin heaters to actually get heat out of it efficienctly or use electric heating powered by the engine…
The only thing ICE engines are is impressive feats of engineering, that we have spent a great deal of time and effort on making sound nice, be small, jump into life quickly etc – they are not and never can be actually efficient, there is far to much energy lost for each stroke – it is just a terrible way to convert to chemical potential to kinetic energy, and because so much of the energy is lost in making it work also a terrible way to turn it to heat.
> an ICE engine is garbage
Yes. Absolute garbage. But unfortunately it is the best thing we still have as whole system of creating kinetic energy. Easy to get, store and refill fuel with high energy density, high availability, scalability, power to mass ratio and all that stuff.
I will definitely prefer a system with brushless electric motor and ZPM as source of electricity, but I didn’t see any ZPMs on the market. Even fuel cells is still something unreliable, stupidly overpriced (some patent troll still own a patent on proton membrane?) and ineffective. But in no case I will choose a battery as a storage of energy.
> ICE engines already burn fuel with maximum efficiency.
Incredible! You just keep coming out with more and more discoveries, completely contrary to everything we know about mechanical engineering. If ICE engines are inherently maximum efficiency, this would suggest that a 1950’s automobile engine was as efficient as a modern high-MPG automobile’s. These MPG ratings are some sort of illusion.
And then there’s locomotives which have spent billions of dollars designing their engines to improve efficiency, and it turns out they were already at maximum efficiency! Incredible!
You, sir, are the greatest genius to ever live.
> You, sir, are the greatest genius to ever live.
Seems like that… At least I understand the difference between efficiency of burning fuel and efficiency of converting some type of energy we get from burning into some specific thing you need right now.
Nobody still mention any available portable power system with better efficiency yet. Efficiency of converting 1 gramm of fuel (or gramm of charged battery for electric engine with battery) into the energy. Nuclear ones could be better, but they are not available. Thermonuclear ones does not exist at all. Any other options?
Your battery EV efficiently takes x in from the grid and turns into really really damn close to x in motive effort… Your ICE really really inefficiently turns y potential in the fuel into barely 1/5th of y in motive effort and then needs a Cat converter to burn all the stuff that didn’t burn…
Battery EV are perfectly practical and useable, for most folks vastly more practical than ICE – cheaper per mile by truly astonishing amounts, even more when you compare them to similarly high spec cars with all the trimmings – full of Air conditioning, GPS, fancy stereo infotainment systems etc – you go comparing them to best of the tiny no creature comforts at all maxium economy ICE in cost per mile EV still win though, and are vastly more comfortable in the process…
Yeah the battery is somewhat heavy, but that doesn’t change the equation in use at all – massive efficiency vs massive inefficiency on the road is very much the story. And if you compare mass of fuel burned even if you assume the EV is powered by fossil fuels it still slaughters the ICE in on the road efficiency…
The only big downsides to EV are for the poorer folks they don’t exist yet, and those with no charger at home/work will loose out on the biggest benefit of Battery EV – the magically full ‘tank’ every morning or end of their shift – so will have to sit around at the chargers for a while. With the range and charge speed at a fast charger the right battery EV even has validity for those doing a huge amount of driving – you were going to take a break every few hundred miles/ half day of driving anyway… Even Battery powered Heavy goods vehicles out there now, won’t be practical enough for every type of trucker right now, but will be for many of them…
You want something that mass for mass does well, and plays like ICE in practice look at fuel cells, great tech, the only downside is filling stations are rather uncommon, as are available vehicles / fuel cells in general – Electric is already distributed everywhere, finding a charger isn’t hard, though fairly soon your standard petrol station is bound to start catering to fuel cells.
Gas-fired power stations emit about 500g of CO
2 per kWh of electricity generated. Please note, this is natural gas.
The average passenger vehicle produces 411g CO2 per mile and 8887g per gallon.
The average electric car consumes 0.346KWh of electricity per mile.
Gasoline has an energy density of 36.1KWh/gal
Without heat recovery gas turbine efficiency may be as low as 35-40%
The internal combustion engine averages 20% efficiency. Has no potential for heat recovery boosting efficiency outside of a hybrid application.
EV’s see parasitic load in extreme cold conditions to heat the batteries causing reduced range.
ICE’s see reduced fuel efficiency and produce more NOx gasses in extreme cold conditions. Up to 10x increase in emissions on cold start from a temperature variation of 30*c to – 7*c
Propane ICE would be best considered for chronically cold climates. Which also outputs cleaner emissions than gasoline.
You’re literally burning gasoline to deliver gasoline to the places you have to burn gasoline to drive to to buy gasoline.
Electricity shoots through copper sky pipelines, sometimes underground, directly to your home with relatively high efficiency.
More people die from fuel delivery than electricity delivery. You could pipeline gasoline to one power plant, not to 500 gas stations.
Internal Combustion Engines, piston or turbine, should be called hot nitrogen engines. What does the most of the work is the 70% of the air which is nitrogen, heated and expanded by the combustion of the fuel with the 20% of the air which is oxygen.
Nitrogen is the “working fluid” of combustion engines. It’s the reaction force to flinging huge amounts of hot nitrogen + combustion byproducts out the exhaust that makes jet engines move.
The most efficient fuel mix is one that meters precisely the amount of fuel to combust with 100% of the oxygen in air.
A problem with that is the fuel usually includes oxygen in its molecules. That perfect (stoichiometric) mix also burns quite hot. Why this is a problem is due to the nitrogen.
The higher heat of a stoichiometric fuel:air ratio makes it easier for any unused oxygen molecules (O2) to combine with nitrogen to form nitrogen oxides (NOx). NOx + water vapor, which is also a byproduct of combustion thanks to any hydrogen in the fuel, = nitric acid.
But a stoichiometric mix produces less particulates, volatile organic compounds and other pollutants because nearly all of them get burned inside the engine.
So to reduce the formation of NOx, engines are usually set up to run just a little too much fuel to ensure that as much as possible of the oxygen from the air and the fuel is used in combustion.
When the carburetor was king, AIR (Air Injection Reaction) pumps were used to force air into the engine exhaust to burn up that excess fuel. The downside of that was more nitrogen in the exhaust made more NOx. A carburetor was a finicky thing to adjust to run just the right amount rich, and when they were right they could only be right within a narrow range of elevation above sea level. Some carburetors with a greater range of compensation for ambient atmospheric pressure were tried but none of them worked very well and when they needed rebuilt they required special tools and an experienced technician.
Electronic fuel injection was the answer and by the mid 1990’s the carb was almost extinct. So was the AIR pump.
But most cars still have catalytic converters because the chemistry of gasoline combustion in air hasn’t changed. Run the “perfect” mix and there’s risk of burning up the engine and it makes more NOx which the catalytic converter has to break apart. So EFI has to constantly monitor the incoming air and the outgoing exhaust and precisely adjust the amount of fuel injected to make the exhaust mix within the ranges the catalyst is made to work with.
The point of “bio” aviation fuel isn’t to reduce CO2 emissions from the engine. The point is that the carbon in that fuel was taken out of the air as CO2 by some plant, so by putting it back into the air, we are carbon neutral. This contrasts with using fossil fuel, where we are carbon positive because we took carbon that was locked in the ground doing nothing and added it to the atmosphere.
Yes, but I never seen that reason in a proposals to pay for “bio” fuel. Usually just “to lower pollution” or something irrelevant too.
And all-in-all fossil fuel really was also taken out of the air, as “bio” one. Just with a longer timespan between capture and release.
Interesting that fossil fuels created by nature literally from “plastic garbage covered Earth in carboniferous period”. There was no any bacteria to destroy a natural composite plastic made from lignin and cellulose, usually named “wood”. So, tons of dead plants covered Earth surface for eons slowly sinking to ground. That layers of plastic garbage become coal and oil later. :)
The carbon in fossil fuels largely came out of the air when the Earth was a whole lot hotter. By burning all this fossil fuel, we are busy returning the composition of Earth’s atmosphere to that of the Cretaceous… when tropical rainforests reached nearly to the poles.
The terraforming project will be the work of generations, but if we keep at it, we can change the world for our grandkids!
/s
Amazing! Are you suggesting that fossil fuels are produced in the present, and weren’t deposited millennia ago as part of geologic processes, and are therefore comparable to fuel sources that pull carbon out of the atmosphere in the present?
Sir, your knowledge of the world is so astonishing, you should write a book! Your knowledge defies everything we thought we knew about science and engineering! I can only assume you’re already a multi-billionaire from employing these incredible insights.
Sorry – wrong. ICE engines are about 30% efficient (F1 is around 50% iirc) but the efficiency of boilers is around 90%. So burning fuel oil to produce electricity is cleaner than burning the fuel in a car engine.
A boiler is not a heat engine – it does not convert heat into mechanical work, so comparing the efficiency of one to the other makes no sense.
> So, the way of burning does not change anything if reaction is the same.
Fascinating. So I can power my home with a small coal-powered generator as efficiently as a grid-scale coal plant? I had no idea that efficiency of converting heat into electricity remains constant regardless of scale.
Have you published this discovery anywhere? You’re in for a Nobel prize for this, you’ve over-turned centuries of engineering!
Very nearly, because the greatest power demand with most homes is space heating and hot water, and for that it’s more efficient to just burn the coal in a furnace than transmit electricity. If you’re also cooking on coal, that might be 2/3rds of your energy demands right there. Plus, there’s adsorption heat pumps that are driven directly by heat, so you could run your AC off of it as well, or multiply the gain of heat in the winter.
What’s left over, some LED lights and a TV? Takes hardly any power. Most of the year you could make that with a miniature CHP unit like they sell in Japan.
That would depend on the scale of the home and electric demand as well, no way a small scale can generate as efficiently, and burning extra to your heating/cooking needs to make up the electric is back into wasteful.
It is a good point often overlooked though that things like wood stoves can be effectively 100% efficient, if you are using them for heating and cooking any heat that gets out of the oven to the house is still usefully heating it up, and tossing some electric generation – even those rather inefficient thermo-electric pads on the back (peltier running in reverse) doesn’t loose any efficiency – the heat that passes through them without making electric is still heating your home and you got some electric out of it in the process..
However for most modern lives I would suggest home generation of the required electric with the heat is not even close to possible to do as efficiently as shipping it in from the big generator – you can live a life where that is more efficient, but its not going to be possible for many folks – shipping fuels by road is vastly more inefficient than shipping electric so you have to be getting the fuel from your doorstep, which rules city dwellers out. You also need to not have a high demand for electric (as if you assume a well insulated house – required for efficiency anyway) you just can’t generate that much with the normal heating and cooking output – your Freezer/Fridge for instance being a pretty constant draw adds up over a day very meaningfully, and add in the computer, television and network gear (all often left on it seems) that is a pretty high baseline power draw to try and match this way….
Yes, as the cost of it comes down it will be more practical as a liquid fuel for gas turbine plants that run on natural gas (methane) now. The additional energy to charge all those cars has to come from somewhere after all but the combustion process could be more efficient and cleaner in an industrial setting (I think).
Indeed, there are far more efficient ways to burn gasoline when the powerplant doesn’t have be small and light and hands-off enough to be used in a vehicle, and it’s a lot easier to do interesting things with the emissions too. If waste gasoline is being produced and it can’t be pumped back into the ground, the next best option would be to run it through a highly efficient power plant (turbine with thermal regen wheels + Stirling generator + Seebeck effect generator?) with a lot of emissions control equipment including a carbon capture system.
Solution #1 the Totem co generator. Use it with methane to provide both electricity and heat. It uses a Fiat engine, that for less powerful ones is the same 1400 CC engine that was used used on Grande Punto CNG models.
First models used a carburettor Panda engine, and to make them run on petrol was trivial, on these newer engine It’s possible that they have an methane only ECU and are missing the petrol pump and injectors.
Having a petrol engine running at a constant speed make it slightly more efficient compared to the same engine put on a car.
http://www.totem.energy/en/home-mobile/
Not if the pollutant you’re trying to eliminate CO2. This thread is full of unintentional red herring arguments about thermal efficiency. That just doesn’t matter. All the carbon in the fuel comes out as CO2 plus a tiny amount of worse pollutants.
Efficiency in that one uses less fuel to get a given result. e.g. heating house, driving car, etc.
Forgot a different but crucial problem.
The electricity networks being woefully undersized for EV’s and plug-in hybrids.
For a worst case scenario, look at Texas during the blizzard where everyone cranked their electric heaters up to 11, now imagine that with vehicles.
And forget about telling people their car can only charge at a set time slot and also has to act as battery for the grid, because you’re basically telling the common populace that they should bear the grunt of utility companies not doing their job.
One problem is the use of electricity for electric heating, using more efficent heating like heat pumps or even better carbon-neutral fuels like wood for heating and better insulation will make some extra electricity available.
To make people charging their cars at a given time, use the same system used by The Phone Company, different hourly rates, maybe having more than two prices for the day.
Can you get a more efficient heater than an electric one? Surely everything is turned into heat? Or do you mean electrical generator to create the electricity in the first place? But then is efficiency even part of this problem? Surely a 1% efficient sustainable carbon free system is better than a 100% efficient carbon based system?
Heat pumps deliver more heating energy than they consume from the grid. No physics laws broken here, the extra energy comes from the heat “pumped” from the cold side. So YES: a heat pump is more “efficient” than an already 100% efficient restive load.
Heat pumps are typically about 300% efficient. It’s not free energy but, 1watt of electricity can deliver ~3watts of heat since the other 2 watts are brought in from outside.
It’s more like 550% with current heat pump standards.
> Surely a 1% efficient sustainable carbon free system is better than a 100% efficient carbon based system?
Surely if we’re talking about synthetic fuels and bio-fuels, that would be begging the question.
It’s not the carbon but the source of it.
Heat pumps are far more efficient than resistive heating, so yes, even though the heat pump also uses electricity. It’s all about how much you need for a given effect.
CHP schemes (where waste heat from power stations is used to heat homes) can help boost the efficiency of carbon based generators, which means you use less carbon overall.
A 1% efficient zero carbon solution would still be shedding heat and using other resources, so efficiency has to be a factor somewhere.
Heat pumps are 500% energy efficient, for a kWh used by the electric motor, they could deliver 5 kWh of heat, because the heat is extracted from the surroundings, so if instead of a simple electric heater one installs an heat pump, the electricity consumption in winter drops to 1/5. Basically an air conditioner it’s an heat pump working in reverse, so if in a house air conditioning is planned, putting an heat pump it’s a better solution.
On using the wood, I concur that in cities and condos it’s difficult to use, but on the countryside it could be a good idea because the wood can be sourced locally, planting trees has advantages on the land and terrains and make lumberjacks happy.
No it doesn’t. The CoP drops as the temperature goes down, so the gain multiplier in the dead of winter is close to 1, while in the summer it can be up to 10-15 because the temperature differential is negligible.
The year-round average CoP is typically about 2-3 depending on location, because most heating takes place in the winter when the performance is poor. The difference between the adverts and the real world is that the manufacturer assumes an even heating load throughout the year, which never actually happens.
To add to what people are saying about heat pumps – gas heating is also more efficient than resistive heating, IF the electricity is coming from a gas power plant in the first place. A gas heater is putting all of that combustion energy directly into your house, while an electrical system has generation and transmission losses.
Once we switch to cheap, renewable power, this will no longer be the case. Even when using resistive heating, a wind powered system still uses far fewer consumable resources than one which includes gas at any stage in the pipeline.
The newest mini-split heat pumps can move heat from outside a building to inside, down to external temperatures as low as -5 degrees F. Below that there has to be another heat source like electric resistance heating, or combustion of natural gas (methane), fuel oil, wood, coal…
You know what? gas network is way undersized for the current gas consumption.
If all cars refuel at the same time there will be not enough gas in station to fuel them.
So how do we proceed now that I have proved that gas based car cannot work?
Gasoline vehicles don’t need to top up every day, whereas reasonably prices electric cars do, and they take far longer to charge anyhow. This makes it much more likely that a significant fraction of all EVs are charging up simultaneously.
Erm, no again. Reasonably priced EVs have roughly 150 miles range TODAY. That’s only going to improve. Most people average 30 miles per day, so don’t need a complete recharge each night, just a top up charge.
But the consumer will plug in every night…. Just like we do with our cell phones when we don’t have too… to top them off. Only reason we don’t with our ‘normal’ vehicles is the inconvenience of driving to the gas station every day.
Can you image the ‘lines’ at a electric ‘gas station’. Waiting in line for you chance to spend an hour charging your vehicle? zzzzz. Not to mention the infer structure to support all that energy being used…. Not practical… yet.
I think we’ve reached the nested comment limit, but I’m replying to “rclark” here.
Some of these things might be true, but these arguments revolve around a very gasoline-based way of thinking. Why should there even be an “electric gas station”? Think about it – what fraction of every day do you spend driving? Unless you work as a driver of one type or other, it’s probably miniscule. The rest of the time, your car is sitting idle – and it can spend all of that time trickle-charging from our existing infrastructure. Your workplace doesn’t NEED to install a line of DC fast chargers, when eight hours of charging from a regular outlet will easily cover the energy spent commuting to work in the first place. All that’s needed for this is a cheap, glorified power strip. The only reason DC fast chargers are ever really needed is to increase maximum range for road trips and such, which make up a tiny portion of traffic.
I can’t say whether the existing infrastructure will be enough to support this, but it’s certainly a better outlook. With city commuters slowly charging while they work or sleep, energy use is spread out over a longer time period and no fancy DC chargers need to be built.
Sweeney, 30 miles per day is still a ton of energy. From my napkin math that’s about the same energy the average household uses a day, and most households have multiple cars, so an over doubling of power consumption for each home.
>Reasonably priced EVs have roughly 150 miles range TODAY.
Is that “reasonably priced” for a middle class person? I find any EV approaching reasonable prices for a car has less than 75 mi range.
A reasonably priced car comes in the £10-20k range, almost all the EVs start above that range, and the ones just barely within that price bracket are dogshit.
Let’s not forget also that whatever range the car is advertised for, you can halve it to get the real mileage, so 150 mi -> 75 mi and 75 -> 40-ish. This is especially when the car is approaching 10 years and its EOL.
With a typical 300 wh/mi consumption figures, to drive 30 miles a day requires 9 kWh and charging out of a 230 Volt 10 Amp socket (16 amps really, but not continuously) it takes four hours to fill up. You come home at 5 pm, plug it in, and it’s drawing power until 9 pm. Meanwhile, everyone in the neighborhood is doing the same thing, while cooking and washing dishes, laundry, television, game consoles, lights…
Basically, if you do not force the users to defer the load to a later time overnight, you’ll see the peak demand at evenings roughly double. If you do, then people will complain that they can’t charge up their cars whenever they need to and want to, and rightly so.
> Basically, if you do not force the users to defer the load to a later time overnight, you’ll see the peak demand at evenings roughly double.
Easy problem to solve: time-of-day pricing, simple timers, network controllable load sequencing… the last is already in use in some businesses and high-demand neighbourhoods to limit or spread out peak loads from air conditioning.
>time-of-day pricing
Like that wouldn’t piss people off, because when else are they going to do the cooking and cleaning and washing laundry? 3 am?
ICE cars don’t need to go to the gas station daily unless they are driven so often to need to. An ICE car can be “recharged” in 3-5min at the gas pump, so people will generally run them down to nearly empty before “recharging” Electric cars on the other hand, especially with at home charging require many hours to recharge, so what you will end up with is everyone at 6-7p after their daily work commute all plugging in at the same time to keep their battery topped up and bringing the grid to it’s knees. Sure there are high speed electric car chargers available, but what does constantly stopping at a high speed charger do to the longevity of the battery? We know what it does to other devices like phones with high speed charging. Even with current tech high speed charging a car can still be a half hour to an hour ordeal depending on how depleted the battery is. Also the battery capacity of an electric car that can do a good 400-500mi on a charge is equivalent to the amount of energy a typical household might use in an entire month, and you’re going to pull all that power out of the grid in an hour or less?
Wood is a hideous fuel for heating, despite fitting the “carbon neutral” definition by torturing the English language. Burning wood produces large quantities of a large variety of toxic gasses, and also produces ash that must be discarded.
Conifers are among the fastest growing trees, and burning them produces more pollutants than hardwoods.
I’ve seen some nice gasifying furnaces being sold in Germany. You load them up with wood and light it up, then close the lid, and they control the amount of air into the insulated firebox to distill the wood in to carbon monoxide, which goes to a separate burner. That makes it possible to burn the whole charge of wood gradually over the entire day, and it doesn’t result in smoke because it’s not burning with a draft up a chimney. The toxic gasses you’re talking about are either converted as well, or burn in the secondary firebox.
Erm, no. The electricity network people themselves say we have enough capacity today, never mind allowing for future network expansion. Remember that EVs can happily charge overnight, when demand is lowest, helping even things out. As a plus they can also feed power back when demand (and prices) are high.
It probably varies per place. The electricity company that supplies Amsterdam and its port, has quit hooking up new commercial users because there is not enough capacity.
Charging an electric car can easily double the electricity consumption of a household. The average power consumption in the Netherlands of one household is about 800w. Charging electric cars goes with 2000 to 3000w, for hours on end.
Now also add the power consumption of the switch from gas heating to electric heating, and electric cooking, and we’re gonna have a bad time.
This means that the transformers (the local ones, that feed 2 to 4 blocks of houses/apartments each) need to be upgraded.
It has to happen some day, and it will happen some day, but it is not a trivial ‘oh, it’ll be fiiiine’ thing.
Even where there is the capacity, the transmission lines aren’t built to support it, and the wiring at the premises can’t support everyone on the street buying EVs and plugging them in at 5 pm after work while also cooking their dinner and taking hot showers with their instant water boilers… etc.
The system has to be sized according to some estimate of a maximum load, for each locality individually, not as an average, so the capacity that is “there” doesn’t help you when the local distribution transformer gets overloaded.
Firstly everyone won’t plug in and start charging at 5pm. Chargers will be set to deliver power overnight while demand is low.
Secondly the EHT transmission network is already sized to deliver peak loads during the day that exceed the extra power that would be used by EVs.
Thirdly they are a great way of soaking up surplus power from things like Solar or Wind power that currently has to be thrown away when the grid is in surplus.
Rarely as I agree with Dude on this they are correct – your current electric grid has been sized to handle the peak load of when it was built with whatever margin they built in. When you go adding in one area more load it is quite possible to exceed the suppliers peak for your area.
However that is both a simple fix, that is done often anyway with any development works – add infrastructure to support the change in peak demand, and not as big a problem as it sounds, as even if every house had an EV plugged in its going to charge cheap, and at a trickle in general.
If every house in my area had an EV demanding fast charge, every house put the kettle on and the local UNI was firing up its wind tunnel etc it would be way way more than the local grid to provide as a peak, and perhaps more total than it could manage in several hours, but at sensible and naturally spread loads – even though they will be tied to human lifecycles so peak and dip through the day the peaks won’t be so excessive as to cause trouble, and over 24 hours all that electric can be shipped easily. Also not likely to cause blackouts either way – most EV are also effectively house UPS, often set up to be able to feed power the other way too, so even if a spike was greater than could actually be supplied for a few mins its regulated away and nobody noticed.
The Orkney Islands are great for wind turbines to make electricity. So just about all of the best sites have turbines installed. The problem is they can’t get most of that electricity *off the islands*. Nobody wants to pay for the massive capacity increase to the underwater link to the main part of the UK in order to get the electricity to where there’s a use for it. https://www.youtube.com/watch?v=8UmsfXWzvEA
Upgrading the power link should have been done first.
Capacity is one thing. Getting it over the wires that were not meant for it is quite another. Have the same problem with wind and solar. Power wasn’t meant to be fed onto distribution lines…. Yet we (as a utility) hear it all the time. They see ‘wires’ and assume all we have to do is run a line over and ‘hook up’ to their neato wind generator and start making money…. Doesn’t quite work that way.
Thanks to the Green party in Sweden, the infrastructure can’t cope today. The country is split into 4 distribution regions (makes sense given the geography). The southern region was perfectly happy with its nuclear plants supplying them clean electricity, combined with renewables when the sun shines and wind blows.
Nuclear, being so not-green, meant the green party, who received barely 4% of the vote, forced the Social Democrats to shut down the nuclear plants in the south. Now the south needs to buy in coal-powered energy from Germany else start up the oil fired plants to keep up with demand. Yes, the Green party prefers coal & oil to nuclear.
We could of course transfer energy from the north – they have good water power, but the north being a few thousand miles from the south means there’s only marginal capacity available to transfer to the south, not enough for the needs.
Here in Gothenburg the council owned monopoly supply company in the summer introduced ‘peak hour charge’ for domestic customers (not those living in flats) whereby you pay a bonus charge on the hour in the month where you used the most energy. After they introduced it, they sent out letters to customers informing them of this, which applied for the month before they knew about it. This is on top of the exisitng ‘max fuse’ charge whereby you get a master (err primary?) fuse at a certain rating say 25A and pay a monthly fee for that. If you want e.g. 32A you pay a higher monthly fee. So now you get charged both for having your cake and eating it!
Meanwhile the distribution fee everybody pays which is supposed to go to maintenance and upgrades has gone to who knows where since they are claiming the need to introduce the above fee as the distribution network couldn’t cope if everybody plugged in their electric car when they got home. Not like anybody could have predicted electric cars 5 years ago.
Sweden is about 975 miles tip to tip.
Standard “green” tactic. It’s not about being clean it’s about getting the most money from the most people and increasing government control over their lives.
Car charging by residential users is largely done at night. Using a relatively smart charger, (which utilities can pay for!) you have it set the charge speed and time so that the load helps fill in the demand curve.
If a blizzard comes through, most people aren’t going to be going anywhere, so the first night might have regular demand, but the following nights would not. And vehicle 2 grid proposals don’t actually send the power back to the rest of the grid, but rather to the house.
Umm no. The charger for a plug in electric car is inside the car itself. That box mounted inside your garage or outside isn’t a “charger” it’s barely smarter than a normal outlet. Pretty much the only thing inside them is a contactor to cut off the power to the end of the cable you plug into your car for safety reasons unless it is actually plugged into a car. You know so your little johnny doesn’t get curious, shove two screw drivers into the end of the cable and electrocute himself with a feed capable of delivering 60amps at 240v. You plug it in, your car sends a signal up the smaller conductors that signals to the wall box it is plugged into a car and it closes the contactor to pump the 240vac straight into the car, where the car’s onboard charger then deals with the conversion of the 240vac to the required DC voltages for charging. The only thing that comes close to an external charger are things like the Tesla high speed chargers, where inside those chargers it does convert the AC power into 400ish V DC and feeds it straight into the batteries. Having a charger onboard the car that could handle those kinds of currents would be prohibitively large and heavy.
Or just using it rationally, rather than actually rationed – if I don’t need it why take it, if I can sell/share with my neighbours what I happen to have extra why horde it etc – a degree of common sense and goodwill creates ‘demand management’ to some extent naturally.
Take the recent fuel shortage in the UK, media makes a panic so suddenly every fuel pump is shipping weeks worth of fuel in a day and obviously the infrastructure can’t come close to keeping up, but there never was a particularly real shortage, just a sudden and stupid surge in demand creating one.
The trick is that you don’t get to decide whether you need it, or when you need it. If the power company has no energy to give you, you don’t get it either way. That is what demand management implies, and why it is equivalent to rationing.
It’s not an issue of panic buying, but normal everyday situations where you have to defer normal things like washing the dishes or the laundry, or yourself, because the grid happens to be out of capacity and can’t supply the energy. Who decides whether you get power and your neighbor doesn’t, or vice versa?
All it is, is a milder version of rolling blackouts.
i think you meant “bear the brunt”….
In most areas there are huge differences between peak and off peak electricity demand. For an example of this the province of Ontario has spread of ~4000 Mw today.
This peak and trough that occurs every day means the grid needs a lot of dispatch-able generation to handle the change in load. That peak is made up with natural gas when other sources are not available or it can result in nuclear units being derated when the trough goes below the normal baseload.
Anything that flattens that peak and trough cycle helps reduce the usage of high carbon intensity generation.
Since the grid is more than capable of handling a hot summer day with a massive afternoon peak it can easily support overnight charging. You can even incentivise people to do this with time of use pricing.
Charging at night for me consists of plugging my car in when I get home and the charger starts when overnight rates begin. If I want to charge on peak I just push a button on my phone to temporarily over ride the time of use setting.
This is not a case of a utility company not doing it’s job. It is giving people an incentive to make use of the grid in a more efficient manner rather than simply passing the high costs on to them.
“The modern mind perhaps shudders at the thought of an environmental crime of that magnitude, and we can’t imagine how anyone would think that was a good solution to the problem.”
Refinery flares.
Really with all this talk of energy there’s still all the inefficiencies in the process from the generation/making to the transportation/losses in the process. 1/3rd gets to the user for a fuel power plant. Reducing those would go a long ways.
Refinery flares burn gasses, so mostly methane, ethane, propane, and butane. They’re made to burn fairly cleanly, producing CO2 and water. That’s minor compared to dumping gasoline into surface water.
Refinery flares in modern refineries are mostly responses to intermittent problems in the manufacturing process. The alternatives are releasing unburned gasses or an explosion.
I grew up in an era where we learned to turn off the lights, close the refrigerator door, don’t leave the water running and close the door so you don’t heat/cool the outside. All these high-tech solutions rely on selling people “stuff”, when you can’t even get them to turn off a d*mn light when they leave a room, or close a door, or put on a sweater when they’re cold. And these are the same people lecturing me about how I’m destroying the planet by not buying an electric car while they tailgate me (in the slow lane), like resource scarcity is some brand new idea no one ever thought of before. Cue Grandpa Simpson yelling….
I think you need to re-examine your assumptions here. People still understand the value of shutting off lights, not leaving water running, and closing the doors when the AC is running — those are common sense and haven’t been lost to new generations. As far as “stuff” that helps with this, that’s a natural progression. Highly-efficient light bulbs, low-flow water fixtures, well-insulated water heaters that don’t waste energy up an open chimney, and multi-pane windows are all technologies that save on resources and many times make daily life a bit better (my house is much brighter with LED bulbs than it was with incandescent, and we’re much warmer in the winter with double-pane windows than with single-pane).
The assumption I would encourage you to adopt is that nobody is leveling criticism directed at you in particular. We’re all in this together, we all are interested in having choices that help address resource scarcity. Some of those choices will make sense to you, some will not and that’s fine.
> If we suddenly don’t need as much gasoline …
… the price of gasoline drops until we find the need.
Also, let’s not forget that between 60-85% of fuel prices is tax. In the UK for example, in 2020 a liter of petrol cost €1.41 on average, of which 81c was various tariffs and 25c was VAT, resulting in a total tax burden of 72.6%. Basically, three quarters of the cost of fuel is simply the government, which means two things:
1) The price of fuel has almost nothing to do with supply and demand, but politics.
2) The governments won’t let gasoline disappear off the market because it’s a huge tax revenue stream that props up all sorts of projects and promises they can’t back out of.
+1 This is a problem with “engineer’s reveries”, when they discover something that isn’t in, or hasn’t yet reached, the “memesphere”(for lack of a better term). They start looking at the numbers that result from, in this case, a chemical analysis, and come up with extreme examples “roughly half of every barrel of crude oil that we pump out of the ground is made of something that’s destined to be useless” (Yeah, don’t hold your breath for that!)
They completely forget economics, culture, sociopolitical factors, etc. Then, of course, one can go down socioeconomic rabbit holes concerning the “petrodollar” and petrodollar recycling. And then the oil refineries losing money seems less consequential to: well, wars in Iraq, and the present war by the Saudi state. One might conclude that dropping oil prices might be a lot more disastrous for international peace than has even begun to be contemplated here, or that we are already in a situation presently that; well, to extend the introductory metaphor, we’ve already made the river a gasoline gutter, there is no waterway left to save! The next article proposes to “engineer our way around the gasoline problem”?!?!?!? I don’t think so!
Indeed. If you are making prophecies about electric cars replacing petrol engines or chemical combustion driven engines in general, you have to remember that the E in “EV” doesn’t really stand for “Electricity”, it stands for “Ersatz”.
Then they forget that we have the ability to crack hydrocarbons to lighter fractions, and reform them to heavier fractions at will. It’s just a question of economics: if you have big surplus of lighter fractions, you can simply burn them for energy to make more of the stuff you do need. So far it’s been the other way around and gasoline and diesel are being made out of heavier oils to meet demand.
Whoops, I accidentally mixed some numbers. It was 81 cents tariffs, 25 cents VAT for a total price of €1.46 per liter which makes the portion of tax at 72.6%
https://www.fuelseurope.eu/knowledge/refining-in-europe/economics-of-refining/fuel-price-breakdown/
The actual price of the actual product is only between 30-40 cents a liter, and it’s a similar story with diesel oil between 35-50 cents. If it weren’t for persistent taxing and meddling with fuel prices, we wouldn’t even be talking about electric cars today. This is the same as it was in the 70’s when people started driving cars modified to run on lamp oil and even turpentine to dodge the crazy fuel taxes and import restrictions.
When the governments realize they can’t keep turning the thumbscrew on transportation without destroying the economy and losing even more tax money by the loss of GDP, the fuel prices will back down again.
And so far as the climate change goes, private cars are responsible for about one fifth of one fifth of humanity’s CO2 output. There are far better targets if you want to change things in a hurry than mess with what people drive to work – which is what ultimately pays all those renewable energy mandates and subsidies.
Mess with fuel prices, you mess with logistics costs, labor mobility – fundamental economic efficiency which is needed to drive the innovation and pay for the solutions to the climate problem. Taxing gasoline and combustion engines away is shooting the horse for a want of shoes.
Well that is what California is doing.
https://www.newsobserver.com/news/business/article254897052.html
(California law to eventually ban gas-powered lawn equipment)
Small IC engines are an entirely separate problem. Even though they burn less fuel and don’t run as long and aren’t as numerous as car engines, they are responsible for a stupidly high percentage of non-CO2 air pollution, and that’s what California is trying to fix.
Underpriced energy is a crutch for some otherwise lackluster economies.
Currently, gas taxes are the only mechanism for collecting near enough to cover the full costs and impacts of burning that gas. Often, the taxes on gas aren’t enough to pay for all the highways and roads that it’s used on.
These folks have more arguments for a higher gas tax.
https://www.nber.org/digest/jul04/effect-gasoline-taxes-work-effort
I do agree that there will be other uses found for gasoline, particularly large-scale uses with greater efficiency and less exhaust. If there was a way to burn fossil fuels without emitting all that crap, we wouldn’t be arguing about their use, would we?
> to cover the full costs and impacts of burning that gas
The money is hardly used for such purposes, and the returns from cheap transport and mobility far outweigh the cost of the fuel – especially as it helps the transition to clean fuels by not constricting the economy.
>Often, the taxes on gas aren’t enough to pay for all the highways and roads that it’s used on.
It’s rather that the government refuses to spend the tax on the infrastructure, and it’s a self-defeating tax because the less fuel the cars use the less money the state gets. This includes EVs that get off free – which means the state has to start taxing electric cars the same should they ever become popular enough.
Investment in infrastructure is a good that everyone benefits from. It’s not sensible to tax drivers for using the roads when the use of the roads earns the money to pay tax everywhere else. A tax dollar earned off of taxing cars and gasoline may be ten dollars lost otherwise, but it’s easy money for the state because people just have to get around.
Example:
https://fee.org/articles/californias-soaring-gas-taxes-arent-even-going-to-the-roads/
>From 2007 to 2010, $1.3 billion in transportation funds was spent not to build or repair state roads but to finance other programs that were apparently more politically rewarding (i.e., generated more votes) than fixing bridges and filling potholes.
Plus, fuel taxes are highly regressive. They impact the working poor the worst, while the rich get to write their fuel use off as business expenses, or simply don’t drive their own cars.
Whether taxes are reasonably used is another conversation. But the main point remains: unreasonably cheap gas as a crutch, and as justification for not making forward-looking changes like better urban design and public transportation that works.
> as justification for not making forward-looking changes
It would be a justification if the counter argument wasn’t climate change which is about other things than the price of fuel. The point is, taxing gasoline doesn’t make gasoline go away, nor does the government actually want it to go away – it just makes it slower to develop alternatives because the economy becomes inefficient.
>what to do with the waste gasoline
Actually, a lot of oil is thermally cracked to shorten the hydrocarbon chains in order to make more gasoline and diesel, because the crude oil is typically too heavy. First, we’ll just do less of that.
Then we’ll use catalytic reforming to upgrade the fuel to heavier fractions. Or, we could crack the fuel down further to propane, butane and methane which are increasingly in demand since electricity is getting too expensive for heating even with heat pumps. The reforming step produces extra hydrogen, which is needed for the cracking process, so the two go hand-in-hand.
Both processes are already and widely employed in the petrochemical industry, because the amount of a perfectly suitable hydrocarbon in the crude oil is often very little and varies by a lot – so some of the other fractions are either broken down or reformed up to make more of a specific kind of hydrocarbon for some specific purpose.
I was going to say exactly this, nice to, for once completely agree with you Dude…
Worth pointing out there is of course a further energy cost in the shifting chain lengths, but it is a pittance compared to how much energy is wasted hauling oil around anyway, and even more vastly dwafed if you go throwing out half of every barrel instead of doing so.
It is rather impressive how easy it actually is to “cut down” or “glue” together carbohydrates.
It is fairly energy efficient all things considered and solves a ton of issues. So yes, “not using” a given fraction is a fairly minor problem.
To be fair, it is far better that the oil gets used in more useful materials rather then just getting burned as fuel.
In catalytic reforming and additional CO2 from the atmosphere be added to the process to use the heaver products as a type of carbon sink? (Imagine just pouring ashphalt down decommissioned mines as a carbon sink.)
If governments won’t try to solve this problem it will self-regulate. Cheaper gasoline would mean higher need and there will be no waste.
The article glosses over some some very important details. However, the refineries primary function is to use catalytic chemistry processes to covert most petroleum into simple fuels, and the thick distillate sludge left over is sold to the shipping industry. Additionally, the literal mountains of sulfur left over from bitumen extraction is larger than the pyramids, and visible from space.
A lithium based battery technology is mainly a pollution relocation technology. The primary sources of true renewable energy generation could dominate, as it is already “more economical than coal” ;-) . However, there are already mountains of thorium dust from rare-earth mineral extraction, and ramping up the production simply increases the rate of growth. Despite all the hype, thorium dust is currently worthless, and a liability the industry really needs solved. Keep in mind petroleum was also a worthless commodity at one time.
For people who think geoengineering our way out of this challenge is an option… Its not… you could tap every mine on the planet and it will at best only buy another 8 years, or plant a billion trees and the long-term carbon cycles are not going to be corrected for thousands of years. It may seem counter-intuitive, but currently living plants are short-term carbon cycle (<5 years) fuels that do not really impact the global trends.
The primary problem with fossil fuels is the rate of consumption, as in reasonable quantities the planet remains at a fixed solar energy retention of around 2.7%. However, we recently blew by the worst case forecasts for the models, and this means the quality of life our decedents can expect will be on a downward trend for awhile. There is consensus on climate change science, but fiscally motivated groups are fearful of being singled out as being responsible for our collective negligence. Carbon capture solutions are presented as a panacea, but if one already had cheap clean energy to run the process it is pointless outside green-washing PR.
I think the future is WiFi-enabled goat drawn carts.
;-)
Well there’s always the lithium-sulfur battery.
https://en.wikipedia.org/wiki/Lithium%E2%80%93sulfur_battery
The problem with crude oil isn’t as dramatic as posed.
Firstly.
Any unused “gasoline” can be used directly as a fuel in a power station. Where the overall efficiency is higher than in a car. Since a power station doesn’t have to care about energy density, and can also operate far closer to peak efficiency. But its waste product can also be used as district heating, or as pre heating for various industrial processes.
Secondly.
We can break down a chain of carbohydrates into shorter chains, ie, turing C8 into two C4. And the opposite can also be done, ie turning 2 C8 into a C16. This is however less cost effective than just refining the crude oil itself, but if faced with few other options it is rather viable and actually done on an industrial scale already.
Thirdly.
A lot of greases, plastics, and so forth can be derived from other products. Crude oil is far from the only source. There is alternative sources, usually in the form of various plant oils that one can then synthesize into other compounds.
So no, the problem of not using a fraction of the crude oil isn’t that dramatic.
The worst portion to do any fancy processing with is however the asphalt range, these really long carbohydrates tends to be too thick to really process in an economical fashion.
Electric vehicles will pay for themselves in real estate value alone.
Currently the land near highways is undervalued and underused due to air pollution. Nobody wants to live next door to the freeway.
This land is potentially extremely valuable precisely because it is next to the highway. Get rid of the nearby pollution source and the value goes up. The town wins too because property taxes go up.
As inspiration look at Boston’s big dig, the increased property values and corresponding taxes have more than paid for it.
Cover the land adjacent to the highways with solar panels to provide nonpolluting renewable electric power for electric car charging stations. Store the excess in efficient rechargeable batteries for nighttime recharging use.
Better, you can power the EVs wirelessly through the road. No stops for recharging, and you end the trip with the same charge as you started. This would be especially beneficial for long haul trucking.
People don’t want to live next to a heavily traveled highway because of noise and danger to children. Pollution is seldom a realistic concern.
Boston’s Big Dig is a classic case of cost overruns, shoddy construction, and corruption. Due to government obfuscation, no economic analysis of it should be trusted.
It’s a very realistic concern, especially if you have children. Air pollution damages childhood development, raises your blood pressure, and contributes to heart attacks and strokes. These effects measurably decreased during Covid19 lockdowns.
There’s more pollution from cars than their exhaust.
For example, the dust that consists of brake pads and particles from the rubber wheels, and the road.
Only about 20% of crude oil can be distilled into gasoline. Where are you getting your numbers from? You can get 50-60% gasoline out of crude by cracking etc., but straight run gasoline is a small amount. Why do you think the Burton process and all consequent processes to get more gasoline out of crude were invented?
The situation isn’t exactly as dire as implied in the article. Diesel traditionally competes with jet fuel — not gasoline — in the production chain due to a major shared component. Using existing equipment, the proportion of diesel can be increased quite a bit.
If that isn’t enough, producing a higher proportion of diesel is really a process upgrade question. Besides financing, that means it’s mostly a problem in locales that abhor upgrades or changes of any kind to their refineries (looking at you, California).
Thanks to the title of the article, I thought there would be something about synthetic fuel. Nope. Maybe save that part for when the article actually says something about it.
Just stop digging it out of the ground!
Did anyone notice that the % values in the barrel graphic adds up to 105% ?!? Really US Department of Energy?!? 🤣
Refiners have spent the last century and change developing a variety of cracking and reformation processes that, taken together, allow them to derive the most profitable mix of refined products from any given barrel of crude. That means that even if gasoline demand evaporated, they’d adjust their processes to produce whatever was in demand. That’s likely to be kerosene fuels like diesel, jet fuel, and the like.
If we can develop an energy source that harnesses the anger generated when reading online comments, we will have enough energy for all eternity.
What for when the sky clouds over and we can no longer use solar?
We could also have some machines to harvest the energy. And if plugged into a sophisticated virtual reality system would mean we won’t have to physically go anywhere, saving more energy. Hmmmm….
I would note that there is an alternative path to Diesel fuel not noted here. Natural Gas can produce the desired Diesel fuel and a facility in Qatar has already been involved in the production of Diesel without use of Crude Oil. Shell operates the plant and the technology is nowhere near new, being noted in CNN Money in May 2012.
If the left over Gasoline from Crude Oil is a concern, diversion of Natural Gas for Diesel use is a suitable alternative stream avoiding a Gasoline glut. It is also theoretically possible to derive Biodiesel fuel similar to other biofuels and this is yet another source which avoids excess Gasoline production potential. Of further note, Internal Combustion engines will take some time to go out of circulation unless there is some clear prohibition on their use, which is exceedingly doubtful. The argument may not be spurious, but the worry about Gasoline accumulation is at best premature and at worst a straw man argument .
There’s heavy trucks running on liquified gas and hydrogen so I’m sure if the prices / environmental rules make it worthwhile fleets would switch to gasoline from diesel – perhaps even as gasoline/electric hybrids.