Chemical engineers at MIT have pulled off something that was once thought impossible. By polymerizing material in two different directions at once, they have created a polymer that is very strong. You can read a pre-print version of the paper over on Arxiv.
Polymers owe many of their useful properties to the fact that they make long chains. Molecules known as monomers join together in strings held together by covalent bonds. Polymer chains may be cross-linked which changes its properties, but it has long been thought that material that had chains going through the X and Y axis would have desirable properties, but making these reliably is a challenge.
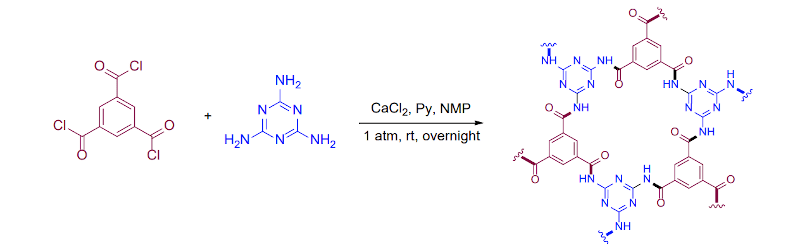
Part of the problem is that it is hard to line up molecules, even large monomers. If one monomer in the chain rotates a bit, it will create a defect in the 2D structure and that defect will grow rapidly as you add more monomers. The new technique is relatively easy to do and is irreversible which is good because reversible chains tend to have undesirable characteristics like low chemical stability. Synthesis does require a few chemicals like melamine, calcium chloride, pyridine, and trimesic acid. Along with N-Methyl-2-pyrrolidone, the mixture eventually forms a gel. The team took pieces of gel and soaked it in ethanol. With some filtering, ultrasonics, centrifuging, and washing with water and acetone, the material was ready for vacuum drying and was made into a powder.
The powder is dissolved in acid and placed on a spinning silicon wafer to form a polymerized nanofilm. Other 2D films have been produced, of course, such as graphene, but polymer films may have a number of applications. In particular, in contrast to conventional polymers, sheets of this material are impermeable to gas and liquid, which could make it very useful as a coating.
According to the MIT press release, the film’s elastic modulus is about four and six times greater than that of bulletproof glass. The amount of force required to break the material is about twice that of steel. It doesn’t sound like this material will be oozing out of our 3D printers anytime soon. But maybe one day you’ll be able to get 2D super-strong resin.
For all their faults, conventional polymers changed the world as we know it. Some polymers occur naturally, and some use natural ingredients, too.















this stuff is going to be a big help in hydrogen storage and use,
and could be the key to lighter ,stronger ,gas cylinders of all kinds,thinking of compsite cylinders made in braiders,and then hey!,space craft need gas sealing,if this stuff can be sprayed and cured with ordinary gear in standard atmospheric conditions,then
that would be good.waiting now.
It has to be made by using centrifugal force which results in 2D sheets. No spray for you!
Some spacecraft rotate already :-) But normally they do not have a polished silicon wafer surface.
It has to be made with a centrifuge now, but tomorrow? That’s what research is for.
Sure. Remember when silicon-dioxide coating was going to make everything awesome? https://phys.org/news/2010-02-spray-on-liquid-glass-revolutionize.html
And then of course there’s the always-imminent miracle of graphene.
YAWWWWNNNNN
I didn’t see much discussion about permeability in the paper, so that may be a bit too much to hope for. But if it works, great. It’ll be a while one way or the other, though.
Twice that of steel – which steel? UHSS goes up to 1,400 MPa and twice that would be stronger than diamonds.
I have cold-rolled steel to almost 4GPa. (exotic steel, exotic heat-treatment and then massive cold-rolling reduction)
When you put it through the rollers, did it leave a dent the shape of the piece?
You raise a valid point. This was 30 years ago and I had forgotten some details. I got to over 2GPa on the 2-high rolling mill at the University of Leeds, until a sample failure in the mill sent me to the hospital to have part of that sample removed from my abdomen.
The final value was achieved with carbide rollers on the 20-high Z-mill at the commercial partner that I was doing the research for.
https://en.wikipedia.org/wiki/Z-mill
AtP of FJ1100 and UKRM fame? I remember you answering some technical questions for me about torquing fasteners on that forum.
Diminutive oblate spheroid, innit?
Drop back in for a visit sometime.
@Mark Olsen: As it happens, the old FJ1100 features in that story, as while I was waiting to be seen in A&E to have the metallurgical sample removed I became concerned for the security of the bike, walked back to the university, took the bike home, then got a taxi back to A&E. (The hole in my abdomen was not bleeding particularly much, as it went into a rather fatty section…)
From the first page of the paper:
Further fabrication offers highly oriented, free-standing films which exhibit exceptional 2D elastic modulus and yield strength at 50.9 ± 15.0 GPa and 0.976 ± 0.113 GPa, respectively.
But at the end of the day is a two-dimensional polymer that is less than 1 nm in thickness.
50 GPa is stretchier than aluminium (70 GPa) and 976 MPa is not “stronger than steel” let alone twice as strong. It’s about on par with certain varieties of tool steel, spring steel, or cold-rolled stainless steels.
They are probably trying to say it has higher specific strength than steel, in other words, higher strength per unit weight. Or looked at a different way, lower density than steel.
I work with cellulose nanocrystals, which have higher *specific* strength than steel. Unfortunately, everybody pitching for a research grant wants to use buzzwords like “sustainable” and “green” and “nano” without knowing a thing about cellulose, or even to make sure they understand the difference between “strength” and “specific strength”. And the university marketing departments write their press releases without bothering to understand the research. There’s also not a lot of fact checking with the “ignorant” country bumpkins who apply their training in physical chemistry, chemical and materials engineering to wood and other forest-based biopolymers.
Pet peeves. I have them…
+1
“According to an MIT press release the elastic modulus … bulletproof glass”
In contrast to the exactly-as-stiff non-bulletproof glass that you would compare it to outside of a press release?
Or the fact that there isn’t a substance called bulletproof glass because it’s a composite material.
Dude, all glass is composite. And what exactly is wrong with comparing something to laminated glass?
Is it also wrong to compare things to the strength of laminated wood?
If it is brittle and breaks sooner, it does a lot worse on the test.
You need more precise values of “exactly,” especially before attempting a pedanticism.
When your complaint is basically, “Those MIT people don’t use the technical words right,” you might want to carefully double-check each of your own words before embarrassing yourself.
“Atoms known as monomers”
Um. Not a chemical engineer, but I don’t think monomers are generally atoms.
Oops molecules, of course.
So… Dune’s plasteel it is! I hope they end caliing it plasteel.
They’ll need a 3d version, then probably.
I think the more useful discovery is not simply the material they made but rather how they made it. I wouldn’t be surprised if other 2D materials start to crop up.
Isn’t carbon fibre 2D in the sense they have layer it. Could this new material be used in the same way to make even stronger/lighter/thinner components?
Transparent aluminum!
That’s just synthetic sapphire (aluminum oxide).
I’m guessing the materials for this are still derived from the petrochemical industry, so a shame this had to come relatively late, whilst we’re all beginning to cut down on polymer usage – hopefully more sustainable methods can be developed – one question – can the same thing be done with PLA or other plant-based materials, or is the chemistry incompatible?
I don’t think petrochemical polymers are going away any time soon, they’re just too damn useful.
Remember we are talking about a film here. There is a lot dependent on how you use that film to make solid parts. It remains to be seen if a 1/8″ lamination of this still beats steel (and in what way) and there are a lot of characteristics other than the elastic modulus and comparing “breaking strength” to steel is a bit unscientific. The terms of the engineering world would be more like tensile strength, yield strength, etc. Would also like to see it compared to other polymer and composite materials already existing. Another thing to consider with polymer technologies is how they perform at high and low temperatures, these are normally domains where metals beat polymers by miles.
Also the failure modes seem to be important. You can have carbon fibre laminate with qualities comparable to aluminum sheet. But the carbon fibre fails by suddenly delaminating, while metals might fail by plasticaly deforming, which might give you some time by indicating problem early. Also the plastic deformation dissipates some energy in case of failure which might be safer when compared to rebound (spring-back) action of multiple shards.
Finally, a material that might be viable for a vacuum airahip.
I’d have a whole lotta love for a polymer zeppelin!
Delighted to see the low-key reaction conditions. “1 atm, RT, overnight”.
That _did_ read as a little bit of a chemist flex, no?
They stopped short of writing “sipping martinis poolside”, but they might as well have.
The production of continuous rolls of the product may be achieved using a silicon roller that has been surfaced etched with an electron beam to provide a template for the spacing and alignment of the molecular units. Further reduction of the dangling O groups may allow for the closing or reduction of the pore ones the sheet has left the templated polymerisation stage of the process. Other forms of functionalization of the O group would open up a “world” of designed properties for the film.
Speaking of 2d, remind you of anything?
Why can’t graphene be impregnated into this material