When it comes to the history of medicine and drugs, Aspirin, or more properly acetyl-salicylic acid, is one of the more interesting stories. Plants rich in salicalates were used as medicines more than four thousand years ago, and in the fourth century BC, [Hippocrates] noted a powder made from willow bark was an excellent analgesic. It was only in the 1800s that acetylated salicylic acid was first synthesized. In 1897, chemists at Bayer gave this ancient remedy a new name: Aspirin. It’s on the WHO List of Essential Medicines, but somehow millions of people don’t have access to this pill found in every pharmacy.
[M. Bindhammer] is working to make Aspirin for Everyone for his entry to the Hackaday Prize, using a small portable lab designed around chemicals that can be easily obtained.
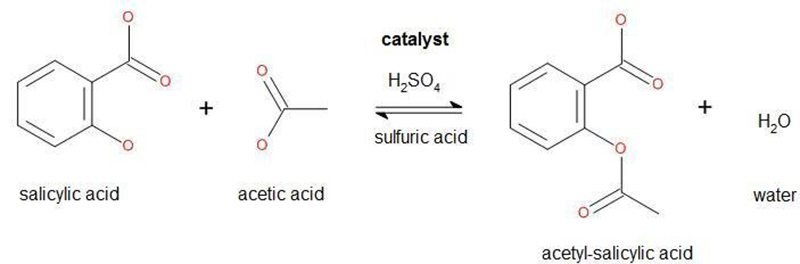
The most common synthesis of Aspirin is salicylic acid treated with acetic anhydrate. Acetic anhydrate is used for the synthesis of heroin, and of course the availability of this heavily restricted by the DEA. Instead, [M. Bindhammer] will use a different method using salicylic acid and acetic acid. If you’re keeping track, that’s replacing a chemical on a DEA list of precursors with very strong vinegar.
[M. Bindhammer] even has a design for the lab that will produce the Aspirin, and it’s small enough to fit in a very large pocket. Everything that is needed for the production of acetyl-salicylic acid is there, including a reaction vessel with a heating element, a water/oil bath, flask, an Allihn condenser, and a vacuum filtering flask. Even if shipping millions of pills to far-flung reaches of the planet were easy, it’s still an exceptional Hackaday Prize entry.




















This year’s hackaday prize theme is totally awesome!
Look beyond any negative connotations that seem obvious, and consider that someone is designing an automated self-contained reaction system, one that can be built by hackers and carried around.
Yes, the concept could lead to devices for making illegal substances. Yes, it’ll never pass FDA approval for use in the US. Yes, shipping these will be more expensive than shipping raw pills. Yes, the original formula is probably more cost-effective than the one he’s using.
But… damn!
I remember when people started tinkering with 3-d printing 20-ish years ago – they were extruding hot glue from a hot glue gun nozzle and making a solder-sucker bulb. 3d printers didn’t look very promising at that time, and I think this project has specifics that don’t look promising at *this* time – but it’s got huge potential.
..but it’s spelled aspirin.
Finally a post that doesn’t give me a headache.
+1!
Pretty cool, but wouldn’t it be easier and cheaper just to give aspirin to the people that need it?
The problem is not that givnig isn’t easier.
It’s what to do when the giving stops. Because they usually can’t afford what you’re giving them yourself.
Even giving has it’s downfalls – it subsidises the companies making an oligo-monopoly so they can both
a) make a slight loss (Good for the books)
b) fulfil arbitrary “social governance” requirements
It’s the teach a man to fish argument, but as always it goes far further than that.
You have to teach the village how to have – and thrive from – a fisherman. With then overfishing his catch.
Exactly this fisherman analogy I had in mind…
Except now you have to give them chemicals and lab gear, instead of pills.
If they can’t afford aspirin, then they can’t afford this.
of this his?
“that’s replacing a chemical on a DEA list of precursors with very strong vinegar.” Sort of. More the other way around though. Vinegar is acetic acid.
Also “and an 1897”? Err?
Care to explain why it’s ‘the other way around’? Like – yes, it’s acetic acid. So what?
Vinegar to acetic acid is like vodka is to ethanol.
Vodka is ethanol, but ethanol isn’t vodka.
So acetic acid can’t be called ‘very strong vinegar’, even though vinegar is acetic acid solution in water? Even wiki suggests we can call it so. BTW, in Russian chemistry it translates literally as something like ‘vinegar acid’ =)
Also, given that he’s on to a easily available solution, he can actually just use widely-available vinegar, get an azeothrope and apply this method:
“CRC’s Handbook of Chemistry and Physics lists an azeotrope of acetic acid and water as boiling at 76.6 degrees Centrigrade and being composed of 97% water and 3% acetic acid. The industrial method for the preparation of glacial acetic acid is distillation using sulfuric acid as a drying agent.” -> http://www.sciencemadness.org/talk/viewthread.php?tid=5528
Given that he’s already gonna have sulfuric acid, that won’t be much of a problem.
You grossly underestimate how nasty both acetic and sulfuric acid are, especially boiling hot. Getting pure aspirin from USP grade chemicals is hard enough. Complicating it needlessly and adding more expense to your feedstocks won’t make this already optimisitic route any more viable. You can cite foreign languages all day long but if you walk into a chem lab and tell someone to get you 500 mL of Vinegar, you’re gonna get 3-5% acetic acid, not Glacial.
The distinction exists for a reason.
Distilling anything takes tremendous amounts of energy. More than you’re likely to have in a place that can’t afford shipping on a bottle of aspirin. It also vastly increases the amount of sulfuric acid you’re gonna need. Yes, you technically can recycle the acid used in the distillation but again, it takes boat loads of energy to do so. If you ask some village whether they’d rather have a pain killers for their toothache or wood to cook their food, they’re gonna choose food nearly every time.
This poject is cool from the ‘lab-on-a-chip’ perspective, but there are too many logistical problems for it to do any of the claims it makes w/ respect to the developing world.
Sorry, no. Vodka contains ethanol. Vodka is not equal to ethanol, or did I miss the 100 volume percent version?
Vodka doesn’t refer to just water + ethanol. Vodka is a very specific concentration of ethanol, ideally 40.1% by volume in water. Vodka ceases to be Vodka when the concentration goes below 37.5% ABV and it goes up to 75% after which it changes to aqua vitae or spiritus fortis etc.
Vodka IS ethanol in a particular way, just as vinegar is acetic acid in a particular way.
To say that acetic acid is vinegar is like saying Everclear or Gem Clear is vodka.
Vinegar contains acetic acid (about 3%). I think your sentence “Vinegar is acetic acid” is incorrect. That’s like saying “Beer is alcohol.”
White vinegar (sometimes called “distilled vinegar”) is glacial acetic acid dissolved in distilled water to a concentration of 5%.
White vinegar is acetic acid. Red or other variations of vinegar have acetic acid in them.
The standard precursor is Acetic Anhydride – (CH3CO)2O, which is a controlled chemical and dangerous to work with. He’s proposing to replace that with Glacial Acetic Acid which is 100% CH3COOH, not the 3-5% in household vinegar. Glacial Acetic acid is not controlled and readily available. Its not nearly as dangerous as Acetic Anhydride to work with.
“Even if shipping millions of pills to far-flung reaches of the planet, it’s still an exceptional Hackaday Prize entry.”
Processed in several languages – still no clue what this sentence is trying to convey.
I think there are a few missing words. Try “even if it’s more expensive than…” or “even if he’s not…”
As for me, I dont give a flying rats fuck about grammatical or spelling errors. I have more than three brain cells to rub together and so can divine the true meaning in about 3 microseconds. Great writeup, great project which would be most useful in a zombie apocalypse. Thanks.
Same.
While this is a pretty cool project and certainly fits the THP bill, clearly the pragmatic solution to the problem is to mass produce aspirin using the standard reaction (which is almost certainly the most cost-effective way to do it if it’s the way it’s done in the industry) and then ship it to where it needs to go.
If the idea is to give up on shipping things to places with no aspirin and give people a way to make it themselves, I would imagine that it would be more useful to simply distribute the instructions (as listed on the project page) and let people hack together their own chemistry equipment (which I would guess is easier than letting people hack together their own lab on a chip). The project could focus on working out how to make the complicated parts using available parts (e.g. how to make a vacuum filtration set up from old soda bottles and baling wire).
“in the fourth century BC, [Hippocrates] noted a powder made from willow bark was an excellent analgesic.” And it still is today, why not just extract salicylic acid from the plants that produce it, hey if it’s good enough for some old greek guy it’s good enough for you.
“if it’s good enough for some old greek guy it’s good enough for you” – NO. Now we don’t extract chemicals without at least purifying them, which’s complicated, and the preferred method still is to be synthesis. Because medicaments made from plants suck unless purified, basically nobody knows what you’ll get along with the active ingredient. That’s why we’re using pills not herbs.
Only those of us with sense.
Of course, the safest way to use natural sources is by exploitng how homeopathy works.
Actually in willow bark there are many more compounds, actually supposed to making it more efficient, but at least with less side effects (e.g. less acidic on the stomach, because it only contains the precursor ‘salicin’ of salicylic acid which is then metabolized in the body) than ASS. It’s just more expensive, tastes bitter and is maybe less comfortable to ingest because you usually make a tea from it.
Aspirin actually causes internal bleeding which can be lethal when used extensively, because it’s both acidic and it prevents blood clotting. It’s essentially turning you into a hemophiliac while giving you ulcers, which quickly leads to anemia and other problems.
It’s not an exceedingly wise thing to give people in great quantities.
That’s why the painkiller of choice is paracetamol/acetaminophen.
Yes. Those are only harmful to your liver.
Only if you take it 4 grams at a time.
@DainBramage
Or take it while boozed. That’s how most people who get themselves in trouble with tylenol go about it, as I understand it.
1) Reye’s syndrome is now considered a rare, genitically-determined issue
2) The lethal overdose for an adult is significantly more than you can buy at once ni most jurisdictions
Aspirin has many more clinically significant side-effects and its role is limited in countries with access to COX-unselective NSAIDs and Proton pump inhibitors.
And yes, Aspirin probably does need a PPI when used for more than short-term analgesia. Aspirin, in therapeutic doses, does cause stomach ulcers (not because of acidity, but because of COX-1 inactivation) and prolong bleeding.
I believe all NSAIDs carry the risk of stomach bleeding when used for long times in high doses.
Acetaminophen isn’t very good to take often. As I understand it raises your blood toxicity rapidly once your body is saturated with the stuff and it doesn’t take much to cause that.
Choice of painkiller really depends on what kind of pain. If I had pain from swelling then I would take ibuprofen since it reduces swelling quickly and acts as a painkiller. A headache that wont go away I take acetaminophen. Joint pain I take aspirin.
Now being able to produce all three of those or similar standins would be great but the potential for abuse is really up there.
I’ve been taking paracetamol every day for years. Chronic pain. I’ve had blood tests for problems, just in case, absolutely no problem at all.
The benefits vs risk are much much much better than paracetamol, which is shitty stuff of which they don’t even know exactly what it does but each time they look at it they find another horrible thing. Including it being addictive btw..
As for paracetamol’s effectiveness as a painkiller, it seems about 20% better than a placebo from what I hear from actual users.
And incidentally, you can easily avoid ulcers from aspirin by either buying the encapsulated version or by taking it with vitamin C or simply by taking it with plenty of water. And also ulcers are a problem of the past – back when they had trouble fixing those. Now we know how to fix them and it’s a non-issue only mentioned by old people who don’t know we can cure ulcers now.
Paracetamol addictive!? You must know something medical science doesn’t. As a painkiller I find it very good. Americans call it Tylenol, acetominaphen. It’s either the most popular, or second-most to aspirin, painkiller there is, I’d bet.
No, but in the proper quantities, for 99% of people, it’s absolutely fine. The world would be a worse place for the lack of it. They sell it in every grocer’s. It’s just not that dangerous. It’s one of the miracle drugs, along with penicillin.
You’re wrong on all the important stuff here [Boing]. Salicylic acid is -more- harsh on the stomach. Willow bark contains mostly salicylic acid. That’s where it gets its name; the genus Salix contains every variety of willow. Willow tea also contains Indole-3-butyric acid, a plant hormone which is toxic to humans and may cause organ damage from long term use (not a unique quality in this tisane).
The reason it’s no longer used is because:
– it tastes horrible
– It’s got more side effects
– You have to drink a lot of it to get the same effect as one little pill
Looked impressive at first sight, but he’s not synthesizing salicylic acid, which I think is the most important part.
Acetyl- part of acetylsalicylic acid is only needed to reduce acidity of salicylic acid, so that patients can avoid chemical burns in their stomach. Salicylic acid itself can be extracted from plants, such as willows’ bark, but the point of the Aspirin is that it is now synthesized from plain phenol, improved with acetyl, packed into tablets and shipped to worldwide in containers.
So the project sound a bit redundant. If he can obtain salicylic acid in large volume, he could also obtain Aspirin of same amount.
Press the “Read more” button and you’ll find below a chapter called “Synthesis of salicylic acid at a laboratory scale”
Which requires oil of wintegreen. Even assuming we’re just producing on a village scale, if your local pharmacy has oil of wintergreen they probably have aspirin. Sure you can grow wintergreen or some other plant and do steam distillation but this is a very energy intensive process, not only for distillaion but wintergreen takes up land that could be used for food. If you’re gonna sacrafice farm land you might as well use it for a crop that is more directly usable.
Wood isn’t generally grown for fuel in poor countries, generally people gather it from woods and scrubland. There’s a whole lot of land doing nothing otherwise. Lack of land is not really a problem for poor countries.
Indeed neither is lack of food, really. Many starving countries export food to meet loan repayments to predatory banks. A few laws and a few less billionaire banking bandits would do more to help the world than anything else. But since that isn’t happening, any way of getting drugs to the sick is a worthy endeavour.
And I know if I had a toothache, I’d give up a LOT of food to be free of it. There’s almost nothing more miserable. Bone surgery is worse, but not much else.
The write-up and project description don’t indicate that this is aimed at the country scale but rather city or village. Perhaps as a way for aid workers to make on site what they need.
I was using wood fires as the most basic heat source required for distillation. The lab-on-a-chip (LoaC) requires more refined heat but one could use generic glassware over a fire. Many villages -do- still rely on wood gathering. My point is that you’re diverting energy from other more immediate requirements. If you’re starting from scratchl; ie steam distilling wintergreen, distilling wild fermented vinegar, and having to recycle sulfuric acid. You’re gonna need a ton of energy to boil all of this. More than a poor community may have.
You’re mistaking my claim about using farmland as needing it to grow fuel. That’s not what I wrote. This arible land would used for growing wintergreen for steam distillation. Again my point is it takes 4 steps: Grow > Distill > hydrolize, acetylate and lots of effort/fuel to get there. Wintergreen has a few % of Oil of wintergreen in it so you need a large field of it, assuming it even grows. You could grow poppies (which have their own problems, but also on WHO’s list) or another medicinal plant in that same space, that require less processing to be usefull. Lack of land may not be an issue, but Lack of ariable land is. As is the human and fuel costs associated w/ tending the fields.
This lab fails in the same way that 3D printing fails in revolutionizing the developing world: Supply chain. We’re expecting to be able to find concentrated oil of wintergreen, acetic and sulfuric acids, but not a $4 bottle of aspirin?
Then there’s the training on using the LoaC. Not impossible but IMO a waste of resources, when we could buy and ship a substantial pharmacy to even the most remote village for the same costs.
Sure, education is ideal but there aren’t too many pharmacy/chemistry jobs in the middle of the Congo. A medical mission isn’t gonna want to have to worry about making and purifying drugs when better QA and supply chains exist from Big Pharma. That pharmacy tech represents one less Dr on staff to treat patients, or one extra mouth to feed depending on funding.
Lack of arable land is mostly just a matter of fertiliser, which is used all round the world by now, I think even subsistence farmers sell some of what they grow to buy chemical fertiliser, it just makes sense to.
Still I agree on the complication of it all, assuming Wintergreen was going to be the source. The huge amount of messing about required to turn it into a reagent is probably a lot less practical than just making the aspirin in a factory to start with. Similarly with the lab. In some disaster scenario, aid workers would be much better off just bringing aspirin itself in place of this gadget. For the same weight and volume, vs the massive amount of trouble otherwise. I don’t think anyone’s ever going to make aspirin on such a small scale that would make this gadget useful.
Doesn’t make sense to plant a small crop of Wintergreens, then send off for the acids, etc, just to make a few hundred tablets a year or so later. The whole infrastructure you’d need would be better employed just buying and shipping aspirin from Chinese or Indian pharma factories.
All that is gold does not glitter,
Not all those who do not govern their every action with humorless, inflexible adherence to the precepts of minimalism and practicality are wasting their time
Anhydrous is hard to get? I bought a liter a few years ago without ID or forms. Got it for use in photography, it just says “glacial acetic acid” instead of anhydrous.
Acetic anhydride isn’t Acetic acid which should be latest clear when comparing the two molecular formulas
The “anhydrous” should give it away. Acetic anhydride is a powder. Adding water to it produces the acid (and a lot of heat). Unfortunately there’s no way of making heroin from the acid, you need the anhydride.
That said, places like Pakistan and Afghanistan must have a fair supply of AA for all the heroin they produce. Whether the manufacturers would be willing to spare some I don’t know. Heroin’s an even better painkiller.
You didn’t even take the time to read the wikipedia article on the topic. Acetic anhydride is NOT a powder.
I’ve seen a drum of it in a dye works and I’m pretty sure it was.
you’re probably thinking of maleic anhydride…
“Anhydride” and “Anhydrous” are not the same thing. Although you can buy anhydrous acetic anhydride (which is a liquid).
But you can buy aspirin off the shelf for almost nothing…why not synthesize something that is expensive?
Because some people can’t afford aspirin. This project is only worth anything if it puts aspirin into the hands of people who couldn’t get it otherwise. That assumes they can obtain the necessary precursors, of course.
I dunno that an on-site pocket-sized aspirin lab is going to be much use, it’s the sort of thing that’s best done in at least medium-sized factories. Improvised or cheap lab equipment would help poor countries a lot.
Little Suzy took a drink but not she drinks no more,
For what she thought was H20
Was H2S04…
That was my first thought too. The starting materials required can be dangerous.
“Acetic anhydrate” in the text should be “Acetic anhydride”.
That’s exactly what I was going to say.