Many years ago, I took a summer trip to the Maryland shore with some friends. One of my buddies and I got bored with playing football on the beach, so we decided to take a hike on one of the many trails back into the wooded area behind the dunes. At the trailhead we noticed a prominent sign, warning about the presence of “very aggressive mosquitos” and not to enter without first applying ample insect repellent. We scoffed at the warning as only young idiots could and soldiered on, bare-legged and confident that we’d be fine.
About three minutes into our hike, a small group came pelting down the trail in a panic. “It’s true! Turn back!” they shouted as they flew past us. Undeterred, or at least unwilling to appear that way to each other, we pressed on, only to discover a few minutes later that we were making a substantial blood sacrifice to the next generation of mosquitos on Assateague Island. We couldn’t bear more than a few seconds before turning tail and running back to the beach and jumping into the ocean to get rid of the last few dozen bloodsuckers.
I learned a valuable lesson from that experience, as well as developing a deep and abiding hatred of mosquitos. It turns out I’m in good company — pretty much everyone hates mosquitos, which are not just a nuisance but can be downright dangerous to be around. But if tests with genetically engineered mosquitos currently underway in Florida turn out well, we may be able to finally turn the tide against mosquito-borne diseases, simply by killing all the females before they ever reach adulthood.
The Circle of Death
To call mosquitos a scourge to humankind is perhaps underselling their true health impact over the millennia. The world’s 3,500-odd species of mosquitos are notorious for their ability to pick up a deadly payload of viruses, protozoans, and even worms when they feast on the blood of warm-blooded creatures, potentially transmitting them to the next hapless creature that comes along. It is estimated that there are over 700 million cases of mosquito-borne diseases every year, resulting in a million or more deaths. A single mosquito-borne disease, malaria, has been such a prolific killer throughout history that it could potentially have killed nearly half of the 110 billion or so humans that have ever lived.
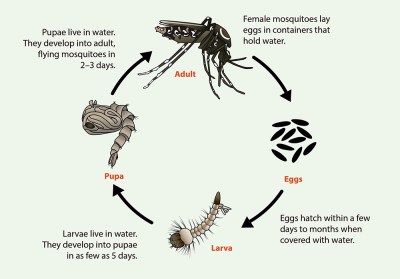
Mosquitos begin life as eggs laid in or near stagnant water. Dry eggs can survive for months; once exposed to water, they hatch within a few days. During their larval stage, mosquitos live and feed in the water. Within a week, the larvae enter the pupal stage, hanging just below the surface of the water as they develop into adults. The full-grown adults emerge two or three days later as they take wing and seek food and mates.
Both male and female mosquitos consume nectar from flowers and fruit, using the sugars for energy. The females, however, require a concentrated source of protein to produce a clutch of eggs, so after mating, they seek out a victim for a blood meal. Once the prey is located, the mosquito uses her mouthparts to saw into the flesh while secreting an anticoagulant in her saliva; it’s this exchange of fluids that carries the potential of transferring disease between parasite and host. A female may take several blood meals before accumulating enough nutrients for a clutch of eggs, which she lays in or near stagnant water before dying.
The semi-aquatic life cycle of most mosquitos seems like it would be the key to control of the insects and thereby, reduction of the diseases they spread. And indeed, a lot of the efforts of mosquito control projects over the last few hundred years have concentrated on eliminating or reducing mosquito-friendly habitats, by draining swamps and eliminating water catchments like tire dumps. Biocontrol using larvae-eating fish or introduction of bacterial spores that kill larvae have been tried, as have a host of pesticides, traps, and physical barriers, like thin films of non-toxic oils that kill mosquito pupae.
New Approach
Mosquito control is basically a numbers game, stacked in their favor. Since each female lays 100 to 200 eggs in a clutch, in wet climates, mosquitos are simply too prolific to get ahead of using standard means. Coupled with collateral damage to the environment — draining wetlands carries potentially huge impacts on a wide range of species, as does the indiscriminate use of pesticides — the search for new control methods with less harmful ecological side-effects has led to research into genetic methods of reducing mosquito populations.
The idea of genetically engineering insects is nothing new. The fruit fly Drosophila melanogaster has had its genome extensively modified for over 100 years, first using standard mating and crossing techniques and later using transgenic methods to insert, delete, and edit genes. The result has been a wealth of knowledge about how the genetics of higher organisms work, as well as models for human diseases ranging from diabetes to Parkinson’s.
But in general, transgenic fruit flies are simply model organisms destined to live and die in the lab. The concept of building a genetically modified insect for release into the wild is fairly new. Oxitec, the company behind the planned releases of transgenic mosquitos in Florida, has been working on the genetic control of a range of pest insect species since it was founded in 2002. They are currently on their second generation of genetically modified Aedes aegypti mosquitos, which is the insect that will soon be tested in Florida.
The mosquito, dubbed OX5034, has been genetically engineered to be self-limiting. Both male and female OX5034 mosquitos carry a synthetic gene that is lethal only to females. The plan is to release OX5034 male mosquitos into a wild population where they’ll breed with unmodified females. These females will take a blood meal and lay eggs that carry the synthetic gene. Only the male eggs in the clutch will develop into adulthood; the females will all die during the larval and pupal stage, which will eventually reduce the number of blood meals taken and the potential for disease spread.
Positive Feedback Turns Deadly
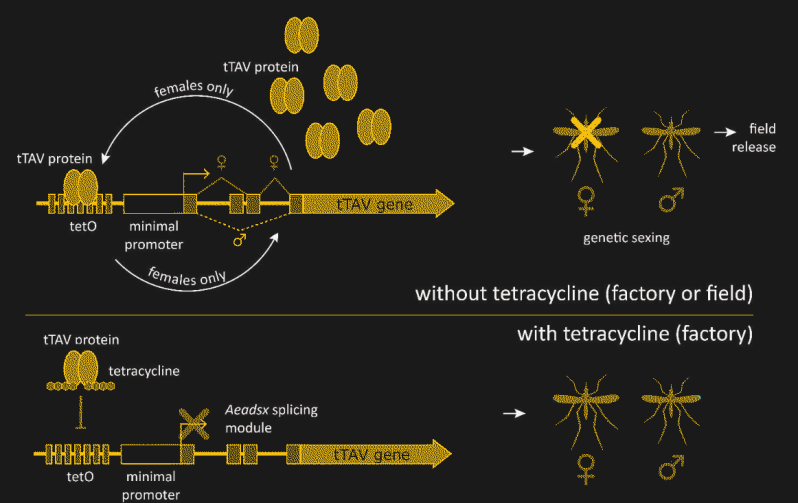
The details of how Oxitec has hacked the Ae. aegypti genome to achieve this level of control are pretty fascinating. The technique revolves around what’s known as a fusion protein, which takes pieces from two or more unrelated proteins and sticks them together into something new. In this case, the protein is called tTAV, or tetracycline-repressible transactivation. The tTAV fusion protein is essentially a nonsense protein, in that it doesn’t serve any useful biochemical function. But tTAV has a trick up its sleeve — it activates its own expression. Expression of the tTAV gene produces tTAV protein, which then binds to the gene to increase the production of tTAV. This positive feedback loop takes over the cellular protein production apparatus, filling the cell with useless tTAV protein and depleting the cell’s resources, killing the cell and eventually the organism.
The lethality of tTAV must be controlled, though, or it would be impossible to raise a population of insects that could survive to pass this trait along. That’s achieved by adding a small section of protein that binds tetracycline to tTAV. If the common antibiotic is present, it will bind to tTAV with a much higher affinity than tTAV binds to its own gene. That locks up the tTAV and keeps it from stimulating its own expression, breaking the positive feedback loop and allowing the insects to survive into adulthood. In the lab, tetracycline is added to the growth medium and food of OX5034 mosquitos, allowing the transgenic mosquitos to survive and breed.
There’s one more trait that has to be added to make this strategy work. To keep the modification active in the wild, tTAV lethal only to females. OX5034 relies on the fact that higher organisms often splice messenger RNA (mRNA) after it has been copied from genomic DNA. The spliced mRNA may have large sections of the original transcript edited out, or even have sequences from entirely different genes stitched together. The OX5034 gene has a sex-dependent splicing module added before the tTAV gene. In males, the OX5034 is spliced so that the gene for tTAV is never expressed, while in females, the tTAV protein is expressed in abundance, unless tetracycline is provided.
In the Field
Having created this controllable, female-specific lethal trait, Oxitec now plans to test it in the wild. The strategy is to produce a lot of OX5034 mosquitos in the lab in the presence of tetracycline so that the females survive. Once mated with the OX5034 males, the females get a meal of horse blood and lay their eggs. The eggs are collected, processed, and placed into special release boxes, which are deployed in the test locations. When activated with plain water, the release boxes act like incubators, allowing the eggs to hatch and the OX5034 mosquitos to start growing. In the absence of tetracycline, though, only the male eggs will progress past the larval stage.

The OX5034 males quickly mate with the native Ae. aegypti females in the test area, who take their blood meal and lay their clutch of eggs just like normal. However, her eggs will all carry a copy of the OX5034 gene, meaning that all the females in the brood will be dependent on tetracycline to survive, and die in its absence. The male eggs, also carriers of the gene, will survive to adulthood, carrying the lethal genetic payload forward to another generation of wild-type females. Tests have shown that OX5034 males are less hardy than wild-type mosquitos and disappear from the population within ten generations, which is about six months. Long-term control of Ae. aegypti would obviously then require a continuous introduction of OX5034 eggs into the environment.
As with any pest remediation project, the OX5034 approach has its risks. Oxitec’s plan for the release has been reviewed by multiple regulatory agencies, including the United State Environmental Protection Agency, which addressed many of the concerns related to the novel insects. One of the most obvious concerns is the presence of tetracycline in the environment. The EPA is therefore ordering Oxitec to not release any OX5034 mosquitos within 500 meters of potential tetracycline sources, including citrus groves, where the antibiotic is used to fight a bacterial disease known as citrus greening, or sewage treatment plants, which may contain tetracycline that has been excreted by humans. Five hundred meters might not seem like much, but field studies show that the vast majority of Ae. aegypti individuals are born, live, and die within a 30-meter radius, and in no case has a mosquito been trapped more than 200 meters from release.
Another potential problem is tampering. The release boxes look a little like plastic coolers and seem like they could be tampered with easily. The company addressed this concern in the Florida Keys test by enlisting only private property owners, mainly homeowners, to host release boxes. The tamper-evident boxes will mainly be secured in fenced yards and will be monitored regularly by Oxitec employees.
As can be expected, the Oxitec release has garnered considerable attention, and not all of it is favorable. This is a new technology, and releasing any transgenic organism into the wild is something that needs to be done with all due regard to safety. But if the current Oxitec tests pan out, genetically engineered insects may be just the leading edge of a wave in innovation in pest control and disease mitigation.
















I walked along side a wooded area on a Florida Gulf coast beach. No warnings and the trail was a vehicle path to allow people to get to a launch for their boats. I even had regular long jeans on. I didn’t go into the water to get them off but they were easy to kill, but they did stab me through my jeans.
Releasing them near a possible source of tetracycline would merely mean that they would be able to survive in the wild – but how is that worse than existing wild mosquitoes?
If anything, providing some tetracycline in the wild for a period of time would allow the gene to propagate further in the population – then remove the tetracycline and the population will collapse.
I think that’s right. It kinda reverts them to “normal” mosquitoes, although mosquitoes with a serious tetracycline addiction.
Would mess up the researchers’ data, though.
I think they should add tetracycline to the boxes so they also get plenty of females in the first generation. This would amplify the initial pulse of gene introduction. Perhaps by a large factor, but I’m still thinking about that one.
Because if they are able to self sustain, then they’ll last more than 10 generations and thus the possibility of a wild mutation being propagated increases dramatically. That increase the likelihood of this creating a ‘tTAV resistant’ strain of mosquitos or worse.
Something something unintended consequences something something slippery slope.
Jem’Hadar and Ketracel White as a “kill switch”.
Victory is life.
@ both of you…..+100 !
I can’t help but wonder about any unforeseen consequences from doing this. Not to mention the fact that a change like this has the risk of making the specific mosquito extinct, and that could be problematic.
Some say mosquitos aren’t having any actual purpose in nature, so why not remove this pest from the world?
But mosquitos aren’t pointless in nature, they do effect a lot of other species. Spreading disease is one thing (partly managing population control), but mosquitos are also a food source for other insects, amphibians, fish and a lot of other animals to be fair.
In short, I would be surprised if reducing the mosquito population didn’t have downsides.
In regards to the statment:
“But if the current Oxitec tests pan out, genetically engineered insects may be just the leading edge of a wave in innovation in pest control and disease mitigation.”
People thought the same about introducing Cane toads to Australia to fix a problem with beetles in sugar cane fields. Lets say that it didn’t pan out in the end.
Some might say that introducing a new spices is different than taking an existing one away, but from an ecology standpoint, it isn’t much different to be fair.
In the end.
At least the experiment is only on 1 species of mosquito, though one of the more common ones in the world.
I would have personally done things differently.
Instead of targeting specifically the female mosquitos, I would instead target both. Breeding them in a controlled environment where the gene doesn’t get expressed ensuring that they don’t die, then releasing these into the wild where the next generation is dead. This limits the risk of the modified gene from spreading like wild fire in the wild population, and thereby removing the concern about unforeseen consequences in the wild.
I feel like this ends with a mad scientist saying he can “eliminate all human suffering from cancer and disease!” and everyone is super pumped. Then they realize he means he is going to kill all humans. M. Knight Shamalamadingdong level twist!
What if we replaced the mosquitoes with other mosquitoes that do not bite humans?
Soil amending is also a renewable resource point to be considered. Ever heard of Chitosan?
Also, other nutrients present in their dead bodies. I’m not certain the contribution to Chitosan compared to all the other sources… however, I am guessing there is some significance over time.
What plagues me is why aren’t the biting insect being engineered to provide immunizations vs spreading disease?
Guessing some laboratories want to spread disease and have engineered bugs to do that role specifically vs provide immunizations from the diseases.
I sometimes wondered over the years about my theory that funguses have been deliberately spread to kill off bat populations so to further spread diseases induced by insects that the bats would consume.
Been following this for awhile, things I’ve read in the literature:
Anopheles mosquitos are not native to North America, there are many species of mosquitos and if we get rid of anopheles the other species will increase in proportion – to reach levels from before North American colonization.
Anopheles aegypti is only known to prey on humans (not other animals), it carries disease, and is not a significant source of food for bats and birds. (Bats eating mosquitos has been blown out of proportion, the energy return is small in proportion to the energy spent catching, and it seems that bats only eat mosquitos as a consequence of catching other insects.)
The first tests of this nature (releasing modified mosquitos) have already been done on a Caribbean island and Malaysia, the results have been studied, and reportedly reduced mosquitos by 80%. You can find descriptions and further links here:
https://www.sciencemag.org/news/2011/01/gm-mosquito-release-malaysia-surprises-opponents-and-scientists-again
There used to be a parasite named “screw worm” that infected cattle (and occasionally humans) that was completely eradicated in North America using this type of method: screwworm flies were raised and irradiated to make them sterile, then released (by airplane) over much of the southwest US. The released flies would mate normally, but the offspring would be sterile. There’s a wikipedia article about this, this used to be a thing when I was younger and IMHO is one of the crowning achievements of human civilization.
(All of this from memory, some statements may not be fully accurate, believe but verify.)
I’m not at all suggesting that there are no risks and that we should not be cautious, just know that some of the risks have been thought through and studied.
I’m hopeful that if this works out with, strong guidelines for usage, that we might be able to *solve* some of the invasive species problems such as cane toads in Australia, mongooses (mongeese?) and cats in Hawaii, and zebra mussels in North America.
There’s something like 3000 species of mosquitos, and only a tiny number of these are a problem for humans. The risk and cost of (human) disease makes them a good target for eradication, if we can do it with a high enough precision to avoid disturbing the rest of the ecosystem.
I see this coming to an uncontrolled transmission of this gene to a virus which would then cross the insect/human border thanks to the enormous amounts of aggressive mosquitoes at play… and a lethal epidemic of an(other) unknown illness
Eventually, humans disappear from the face of the Earth and, voila, problem solved.
Time to stockpile tetracycline!
There are some pretty big differences between mosquito and human physiology. (Understatement.)
Waiting for the “bloodbug” to result from this experiment :P
(yeah… I’ve been playing Fallout 4 too long)
It’s perhaps less of a hack, but there’s other, non genetically modifying methods used for this as well. This technique blocks the diseases (in A. Aegypti at least), doesn’t require releasing more mosquitoes each year forever, and uses bacteria (Wolbachia) already existing in nature. You’ll still get bitten though.
https://www.worldmosquitoprogram.org/
Although there are also other methods using Wolbachia to supress mosquito populations, by releasing only Wolbachia infected males like Google’s Debug project.
It blocks only one disease (dengue fever) and not all (Zika for example).
Bacillus thuringiensis israelensi is also known to block larva development, you can use it in wet places where you can’t use chemicals.
So they claim it blocks dengue, Zika, Chikungunya and yellow fever, and it stays in the wild population so it seems to be a one time cost, rather than suppression methods which need constant reapplication.
Doesn’t work for malaria (different species of mosquito), but there’s some new research on microsporidia species which could block malaria in a similar way.
What could possibly go wrong? I’m guessing there is a B-movie out there called “Attack of the Killer Mosquitoes.”
Ah yes. Victims sucked down like a straw in a drink. Who needs embalming. Just roll them up and stick in a drawer.
It’s already gone wrong. The modified mosquitoes have passed genes to wild populations.
https://www.nature.com/articles/s41598-019-49660-6
I’m just wondering how long it will take for one of these next generation of genetically engineered insect species to become the next generation of killer bees or worse.
Does anyone see an issue with assuming male mosquitoes wouldn’t change into female ones when there are only males left..? I don’t know how often this happens in this particular species but anyone who’s raised bunnies can tell you animals and insects have been often observed “adapting” to a lack of access to the other sex for reproduction.
[Citation needed]
Jurassic Park 😀 Nature finds a way
Here it is:
“animals and insects have been often observed “adapting” to a lack of access to the other sex for reproduction”
– One That Rised Rabbits ;)
Jokes aside – it is just hard to determine rabbits gender and people mistakenly take male and female instead two males or two females – yes I have googled that. I have heard similar rumor about hienas which appeared to be widely repeated misunderstanding. Wiki mentions clown fish as being able to change sex. Also some say that human can – to be checked as I can hardly believe that.
Once I’ve red interesting article on how to determine gender of a person. It appears it’s not that easy at all and current gender discussion seems to make it more difficult as it is focused on two polarized camps with exclusion of science.
Looks like I am mistaken in the case of bunnies, I do see where some fish, snakes and insects do this but in the case of mosquitoes looks like they aren’t known for this.
https://en.wikipedia.org/wiki/Sequential_hermaphroditism
The thing that bugs me about this (get it?) is that certainly some critter eats mosquito larvae. Killing all the mosquitoes puts pressure on that critter. Dragonflies? Recently un-tadpoled frogs? Damnit, I’m not an ecologist, Jim.
The reassuring bits about this technique is that it seems to revert out of the population over time, which means that people can put a dent in the mosquitoes without totally wiping them out, which is probably a lot safer.
But still, some part of the back of my mind keeps calling out “Cane Toads”. (Which, if you haven’t seen is an absolutely brilliant documentary as well as a man-made ecological disaster.)
I was pleased with our Mosquito Magnet Liberty, until it failed.
I looked into this a few years ago. I don’t have specific sources right in front of me but the conclusion I came to is that lots of things eat them but nothing eats *just* them. If mosquitoes were wiped out other insects would take their place in the food chain. They aren’t a keystone species.
Wasn’t this tried in Brazil 6 years ago?
Were the results so good (no more mosquitos in Brazil) that we must duplicate that success in Florida?
What could possibly go wrong?
https://www.inverse.com/article/59254-genetically-modified-mosquitoes
“Part of the problem, Powell told Yale News, was that expectations based on isolated laboratory results didn’t really translate to the field.” Something got lost in translation…
It’s unfortunately not hard to imagine this technology being used against humans.
Trust me, I’m a (genetics) engineer!
I’ve only ever made one mistake, granted it was wiping out all life on a planet. But rest assured I won’t make that mistake again!
Oh man those Assateague mosquitos are RAVENOUS. When I was out at the pads at the spaceport several other people in my group were getting eaten alive by em when they walked through a patch of grass by the ramps, they’re jokingly called the “Second state bird of the eastern shore”.
Mosquitos … to _deliver_ vaccines. Couple of tech issues to work out but who has never been bitten?
Fly, fly my pretties!