Seasoned Hackaday readers may have noticed over the years, that some of us who toil under the sign of the Jolly Wrencher have a penchant for older tech. After all, what’s not to like in a dirt cheap piece of consumer electronics from decades past that’s just begging for a bit of hardware hacking? For me at the moment this is manifesting itself in a selection of 8mm movie cameras, as I pursue a project that will eventually deliver a decent quality digital film cartridge.
When A Cell Is From West Germany, You Know It’s Old

The trouble with scouring junk shops for a technology superseded four decades ago is that the cameras I find have in most cases been sitting in a drawer since the early 1980s. They were a valuable item back in the day so of course they were hung on to, then they were forgotten about until one day the grown-ups who were once the kids featured in the home movies are clearing the house, and they start their journey to my bench.
The problem is that very few owners of 8mm cameras had the good sense to remove their batteries before putting them away, so I inevitably end up with a battery compartment full of crusty 1980s Duracells and rusted contacts. This has left me curious, just what has happened here and how can I fix it?
What’s The Leaky Stuff?
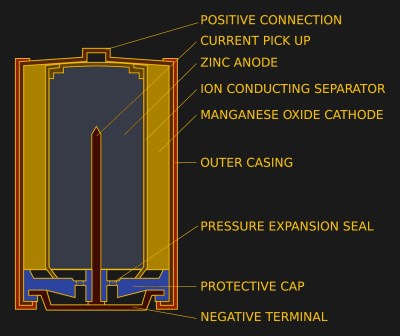
Non-rechargeable cells come in a variety of chemistries, but the commercial ones we’re most familiar with are zinc-carbon “dry cells”, and “Alkaline” zinc-manganese dioxide cells. The zinc-carbon variety are becoming less common here in 2022 and have an acidic zinc chloride or ammonium chloride electrolyte, while the alkaline cells have a higher capacity and a basic potassium hydroxide electrolyte. They both have different failure modes that result in the leaky cells, so it’s worth taking a look at each one.
The failure mode of a zinc-carbon cell is a chemical one, the acidic electrolyte reacts with the zinc can anode, and eventually eats through it. The leaking electrolyte then attacks the surrounding circuitry and battery clips. It’s hardly a concentrated acid, but it’s enough to do plenty of damage over the years.
Meanwhile an alkaline cell has a build-up of hydrogen as it degrades. It incorporates a vent which allows the hydrogen to escape, however the hydrogen pressure can instead force the electrolyte out through this vent. The electrolyte will then corrode the battery terminals and any other electronics it touches. A feature of alkaline cell leakage is a white crust, this is potassium carbonate formed from the reaction between the potassium hydroxide electrolyte and carbon dioxide in the air.
The Global Parts Bin To The Rescue

How much damage has been done is usually a function of how long the leaking batteries have been in the device. Sometimes one is lucky and the battery contacts are salvageable, otherwise they are too far gone and a replacement has to be found. A past me tried all sorts of home-made solutions using stiff copper wire and other materials, but today thanks to the miracle of international commerce it’s usually possible to find a contact the same as or very similar to the old one. A quick AliExpress search on terms such as “AA battery spring” will return numerous options, and it’s simply a case then of paging through to find the one you need on the terms you like.
So those of you who like retro tech will find something familiar in the last few paragraphs, but there’s a lesson to be found in dealing with ancient batteries. Here in 2022 we’re more likely to have lithium polymer cells in our consumer devices and so the need to keep a pile of Duracells at hand is reduced. But the thought of today’s equivalent of a Super 8 camera lying forgotten in a drawer for decades with a cheap li-po pouch cell inside it is far more frightening than something with some crusty manganese cells. Have we just found the root cause of house fires in the 2040s?
















I’m often collecting old electronics like CRTs, cameras, VCRs, computers and stuff because they contain valuable metals. I’m most interested in gold and silver but lead (solder), aluminium and steel make money too, not much but better than nothing. Plastics, carton and paper I usually shred. I’ve recently welded together simple pellet press and I sell this at cheaper price to people who can’t buy real pellet because of price or shortages.
Do you check to see if a working picture tube will fetch a better price first? You’d be surprised what people pay for them now
I second that. Not mindlessly destroying parts and instead selling those parts intact (if they are) can be a win-win situation. CRT monitors or just the working tubes isn’t a bad idea. Same goes for old motherboards. 486 PCs are nolonger considered old scrap, for example. Same goes for Pentium 1 to 4.. Or vintage ISA cards, soundcards in general.. People active in the metal scrapping scene should know that and always be up to date, IMHO. 😉
How true, I got top dollar for the last IBM XT mono screen I sold.
I’ve a bunch of ISA cards that need to hit fleabay soon, network cards mainly, another IBM screen, a couple of PC-XT’s, some old PC cases (great for a retro-look with up-to-date PC innards)
Even 486’s eh? At last! my hoarding will pay off….
Can you show us what you pellet press design looks like? Maybe show and explain how to build one?
Huh? You’re selling pellets of mixed plastic + cellulose for use in pellet stoves?
“root cause of house fires in the 2040s? Nah. A Li-Poly cell that slowly discharges and puffs up will, if punctured let out some evaporated electrolyte which is flammable, but without any ignition source, absolutely nothing happens.
The only thing is, puncture it with something metal gives you a short and with a little luck a spark. There you have your ignition. The metal that punctures it, is the lithium itself, conductive enough to create a short. So empty cell will eventually cause severe fires…
The lithium in a LiPo battery is in a paste solution. It’s not puncturing anything.
Guess what happens when the lithium metal inside the battery gets exposed to air and humidity?
Lithium ion batteries don’t have metallic lithium; fire is caused by overheating causing internal shorts and thermal runaway.
Many mild cases Dremel wire brush does a good job. My big gripe is with manufacturing soldered on the board rechargeable memory backup battery that becomes a time bomb and leaks into the traces causing much damage. NiCd or NiMH coin sized cells stacked in 2 or 3. They can leak in 10 years or less, but they will leak!
Indeed, normally the ignition source is the stored energy in the cell, for with deep-discharged cells, this isn’t an issue.
Don’t attempt to charge them though!
The best way to go for this is probably a (very small) modification to the battery holder itself (which would need “manufactory involvement” and a bit of absorbent paper.
The Idea is not to prevent the batteries from leaking, but acknowledge that they leak every now and then, and then absorb the moisture, and keep it in an area where it does no harm.
Then, when you have discovered the batteries have leaked, you just replace the paper and put in some new batteries.
When alkaline batteries leak, the squeeze out a bit of liquid, but you rarely see that. Within a few hours or days that liquid dries and forms the crunchy crystals that slowly start eating away at your electronics. If that bit of paper keeps the liquid away from the area’s it can do harm, then it does not matter much if the battery leaks or not.
I have a quite strong preference for 9V batteries for my DMM’s. Usually these are connected with a clip and leads, and that is easy to isolate from the rest of the electronics. Just wrap a bit of toilet (or kitchen) paper around the battery when you install it. Then at most the battery clip itself gets destroyed, but that’s also easily replaced.
Over the years I got into the habit of dismantling used Zn-C cells to extract the small graphite rod. Soak them in a dilute lye solution, rinse and heat them with a torch. Its amazing how useful the graphite sticks can be for all sorts of experiments and impromptu glassblowing tools.
I have tried every battery name under the sun and they have all leaked at one time or another. About a year ago I decided to stop storing anything with batteries. Bought a package of Ray-o-leaks at the store and all of them leaked within 8 months of purchase. The batteries destroyed two of my daughters toys. I threw out all the remaining batteries in the package. I now use Duracell Procell batteries but find they go dead after being put in a device with an actual on/off switch (meaning no parasitic draw) after about 3 months. What have all yous guys found to last the longest without leaking?
Every time I open the battery compartment of a remote and find the white crust it’s been Duracells. I don’t buy them anymore.
Duracells used to be great until about 10 years ago. Now I won’t buy them either.I don’t know if it is associated with the sale of the brand or not.
I recently bought a Weston 689 resistance meter. Surprisingly, when I tried it, the needle moved, but it was not accurate. I opened it up and there was a big old D cell Eveready Energizer battery with no sign of leakage. Based on the Wikipedia entry, it was manufactured sometime between 1968 and 1974.
I’ve noted Duracells start to break down way before their expiration date.
We’ve had Duracell Procells destroy a dozen microphones over the years. The ones (AA) marked to expire March 2023 leaked out in the package, thrown away while still registering over 1.5 volts.
I’ve had the same experience. Virtuall all of the items that have had battery leakage over the past 10 years have been Duracells. I’ve had a few Energizer and Ray-o-vac, but not enough to be concerned about. I’ve found that if you spend a little more and get the “industrial” batteries, they seem to leak less, but that may just be my impression.
Duracell used to have a no-leak guarantee and they would repair or replace anything damaged by them. That, plus heavy discounts, and they got all the police and first responders to use them. The first responders have [mostly] stayed loyal to Duracell even though the guarantee is long gone. Good marketing on Duracell’s part. Energizer and Ray-O-Vac now has the guarantee.
the FAA needs to get with the times – Duracells are mandated in flight-critical equipment for the above historical reason, but as demonstrated, that is no longer a guarantee.
for my own purposes, I did the monster math and realized that around five recharges, a set of 4 USB-AA/AAA Lithium-Buck cells has amortized itself. Bought a set for my VR controllers (surprisingly high draw, with all the tracking LEDs and the haptic motors) and have no regrets thusfar
Brand / Model # of those USB AAA/AA cells please!
Dave at the EEV Blog Youtube Channel did a long-term test of various brands of battery. Surprising him as much as me, the Duracells haven’t leaked after several years, while other brands have. He’s in Australia, perhaps the Duracells sold there are bertter than the ones in the US? Because I’ve found Duracells to be very leaky.
I suspect poor quality control leads to entire lots being bad while others aren’t. I’ve had that experience with normally reliable Amazon AAs, but then unfortunately bought a 24 pack which only had a fraction of their rated capacity. Other reviewers had the same problem.
On leakage, Duracells never used to do so. Then, suddenly, everyone started having problems. So, once again, I suspect it’s quality control failure for various lots which may be related to where they were manufactured -OR- QC failures on the part of cell component suppliers which I suspect would probably be in China as too many things are these days.
Wouldn’t surprise me at all. And we give China preferential treatment as a trade partner whether we admit it or not. So they can get away with cheapening products. I guess slave-labour wages isn’t a big enough benefit to Corporate. But let me interrupt my rant.
I’ve had good luck with those Energizer lithium primary cells…you know, the silver ones you can get at the drugstore. They’re pricy, but I haven’t had a leakage failure yet. I figure a matter of time before the bean counters notice and do their own version of “quality control”.
As I said, they’re relatively expensive. But still cheaper and less aggravating than corrosion. Unfortunately I cannot find these in the C or D sizes. Oh well. No, according to the package, not made in USA (what is?). But it seems to be a smaller country (Singapore) which has yet to acquire the clout to try these tricks.
I second that! A 100% of my equipment damaged by battery leak along the last two decades involved Duracells.
Just stop using one time use batteries. Get some NIMH batteries. I had some alkaline batteries almost destroy a $500 radio. I completely swore off them after that. Never had an issue with all the eneloop batteries I bought.
I think they still can leak, but not nearly as often. I think the main old-computer-killer is Ni-Cd, with special mention to the Li-SOCl2 that Apple used to use, and which are super corrosive and allegedly also pretty toxic once they leak.
Every time I’m by IKEA I pick up a few packs of their “ladda” AA and AAA NiMh cells which appear to be rebranded Eneloop and Eneloop Pro cells. Great price for high quality made in Japan LSD NiMh cells.
I was going to post the same advice. The rechargable ENELOOP series by Panasonic are the cells I’ve ever used. Zero leaks so far after 8+ years, long term voltage stability with minimal self discharge, high current capacity. I’ve replaced everything I can with ENELOOP cells and never regretted it. Leaky cells destroy so many electronic devices.
For devices where the battery is a backup, the device isn’t used often or has a very low discharge when in use I started using Energizer Ultimate Lithium. They have more energy and very long shelf life with low self dischagre. They are very unlikely to leak. Expensive but worth it for use where the battery may be in place for years. Eg the flashlight in your car for emergency use, smoke alarm backup, hand held instruments, etc.
I’ve never had NiMH batteries leak, and I started using Sanyo Eneloop LSD batteries for almost everything almost 15 years ago.
THAT! Never seen NiMH leak, and Eneloops (the real deal, which is quite hard to find) have really low self-discharge. Only drawback is the slightly lower peak voltage (around 1.2V) of rechargeables confuses some electronics which were made to work with alkalines (much closer if not slightly above 1.5V) and so plenty of “less then smart” electronics complain of dead batteries when presented a fresh set of rechargeables. My label maker does that, for example.
Sometimes you can put 3 NiMH in a device instead of 2 Alkaline, then at 3.2 you are right around a 1.6v fresh Alkaline. Of course a fresh set is 1.4v so you would be at 3.8v. If your circuit will take it the runtime is good, it might even make the power conversion circuitry more efficient if it is a buck style.
Seconding this. Just use Eneloops or some other good NiMH battery. No leaking, last just as long, and (as important!) less toxic stuff in landfills. They easily pay for themselves after the 2nd or 3rd recharge.
True, even cheap NiMH’s seem leak free, although some develop an inability to be charged, while other go on for years.
The old NiCd’s always seemed to leak eventually, and I would hazard to guess, quite toxic stuff!
Another note cheering NiMH batteries, Eneloops in particular. I tell everybody to stop using alkaline cells – they’re bad for the planet, your wallet, and the device they leak all over.
One of those 1980s West German cells still had 1.5 volts, amazingly.
Sony, Panasonic, or Hi-Watt. Pretty much anything you find shipped with remote controls. Sony and Panasonic can be bought online in the US, Hi-Watt seems limited to Asia and Europe.
Energizer non-rechargeable lithium batteries. They are expensive and I only bought them thinking that they were rechargeable and was really mad when I found out otherwise. HOWEVER, they lasted 5 or 6 years in a various stored items without leaking.
I have digital camera [Pentax K-?] that won’t work with any other AA batteries. It works okay with its rechargeable packs.
I found a really small camera, and decent pixel count. But it uses some odd battery. And if the camera is bad, I waste money on the battery.
It is my perception that all modern cells leak with frequency much greater than they did in decades past. I think manufacturing “optimization” has cheapened the materials or simplified the sealing process so as to reduce costs…but with dire consequences.
In my opinion, this is false econony. I have ruined three expensive Maglites, for example, when electrolyte leaked and not only corroded the interior of the body, but cemented the cells inside…so firmly that they could not be driven out with a hammer!
“Remove cells when the device is not in use,” some say. Well, it materially reduces the value of a flashlight in an emergency if the first thing I have to do to use it is dismantle it to install cells.
I’d add that one of those flashlights were ruined by leaking batteries that still produced bright light… so there was no reason to believe that the batteries were in decline.
In some cases, now, I have created plastic or cardboard battery sleeves to help contain the corrosive goo. In other cases, I have wrapped battery columns in clear packing tape.
“…so firmly that they could not be driven out with a hammer!”
Yeah, I ended up using a drill and some Vise-Grips to get some out.
Had a few “Mini-Maglights” suffer a similar fate; ended up pitching them out. Use those “Energizer Ultimate Lithium” cells instead. Couldn’t agree more about not storing the light sans batteries. In an emergency, the batteries would probably be stored separately too
I’m wondering if there is a common alloy that is resistant to leaky batteries
It’s called “plastic”
It’s fantastic!
But it doesn’t conduct electricity.
Duracell quality is long gone, they’re just a Procter & Gamble brand with a severe quality problem.
Lawsuit was unfortunately kaiboshed:
4/2016 “A federal judge {Koh} threw out a lawsuit accusing Procter & Gamble Co PG.N of misleading consumers by guaranteeing that Duracell batteries would not fail for 10 years, when in fact the batteries might leak when used or stored normally.”
“Koh said reasonable consumers would understand that P&G’s representation that the batteries were “guaranteed for 10 years in storage” was a warranty to repair, replace or refund batteries that failed within that timeframe, and not a promise that the batteries “have no potential to leak.”
“She also said in her 33-page decision that the complaint did not identify any cause, including any design or manufacturing defect, as to why the batteries might leak.”
It’s terrible they can get away with selling such junk that has ruined many devices I own. As if consumers can diagnose the cause and reverse-engineer the junk design.
https://www.reuters.com/article/us-procter-gamble-lawsuit-duracell-idUSKCN0WI23A
Bought a new Xbox One controller back in the day, and it came with Duracells.
They started leaking in no time (we’re talking less than a week). Called them, and they required me to send my brand spankin’ new (but now corroded) controller, and then told me “You might not get it back if we think it’s not broken.”
F them.
I have had both Energizer and Duracell alkaline batteries leak even though the companies “guarantee” they won’t. I have switched to Energizer Ultimate Lithium cells for expensive devices, ones that are in extreme heat/cold, and ones that are rarely used. I use Eneloop rechargeable Ni-MH for the rest of my devices that get used rarely, frequently, or deal well with the lower-voltage. My understanding is that the lithiums and eneloops will not leak, although my Ni-Cd rechargeables from the 1980s eventually leaked after years of being forgotten about.
White vinegar is what you need to clear up Duracell gunk. It fizzes and bubbles in quite an entertaining way but when it has finished it usually leaves the battery contacts in a servicable condition,
But the thin plating is gone and the contacts will rust from moisture .
Use Eveready Ultimate Lithium for use cases where the battery will be in the device for a long time. Eg. Smoke alarm backup, multimeters, etc or where the usage is low or the discharge rate is low. They have atveast a 10 year life, good low and high temp performance, higher energy content, and they will not likely leak. Expensive but worth for the appropriate usage. Like the backup for my solar powered weather station on top of a pole thats a lot of effort to swap out. I expect them to last for years. Only available in AAA, AA and 9 volt because the government is worried theyll blowup . Lithium iron disulfide has a low likelyhood of catching fire
That sounds quite plausible.
Vinegar can also be used to remove mill scale and other oxides.
I once let a brand new piece of hot rolled steel sit in vinegar for a week (I forgot it) and apart from the rust removal, it also etched the steel underneath a bit, and you could see some remnants from the rolling which was not visible on the rest of the steel (even when mill scale removed).
It was regular household rinegar, probably 4%.
Jup. Especially if the crust is preventing battery removal, the encrusted alkaline electrolyte readily dissolves away in vinegar and the cells will more freely come out. As far as contacts they come back crust-free and in original form but usually seem to have some sort of imperceptible insulating (oxide?) layer that then needs to be scraped through
I do this with hydrochloric instead of vinegar. Same idea, though: remove base with acid. HCl might rinse cleaner, but it’s probably moot. Use what you got!
HCl is wayyyy too aggressive for most small electronics that have fallen victim to The Crustys, especially if you can’t get the afflicted components in isolation. Also requires much more stringent handling. Vinegar is literally Food-caliber acidity, but it’s plenty for cleaning battery crustys
I usually go with lemon juice for cleaning those contacts, surprinsingly efficient, plus it leaves a frash scent :D
I have also used lemon to clean the sulfation of batteries and it has been very good 🙂
Greetings from Spain
Thanks for featuring the Mignon cell from good old West Germany. 😍
I’ve still got a few of those old 1980s “batteries” here.
Interestingly, not all have leaked yet or are dead. Some hold still a charge.
PS: A acquaintance of my family, he’s from East Germany, always corrects us about the term “Batterie” (battery):
A battery is a set of primary cells. Thus, a single cell alone makes no battery yet.
So an AA/Mignon/R6 is no battery. It’s a “Primärzelle” (primary cell).
I guess he uses the term battery like in the military speech. A “battery” of rockets, a “battery” of grenades.. ;)
He uses the term “battery” as it was orignally applied to electrochemical cells and batteries – which indeed came from the military usage, but from Benjamin Franklin all the way back in 1749. https://en.wikipedia.org/wiki/Electric_battery#Invention
I once had an older alkaline battery which was used together with other batteries in a device. When I measured its voltage, I was very surprised: The voltage was reversed at around 0.5 V. I cannot explain this, maybe someone else can?
The battery had probably less capacity left than the others (higher leakage, reused ‘good’ one, different age … typical reasons) an got reverse charged by the others when it was empty. This also is a common reason for rechargables to die in use.
If you replace a set of series connected batteries, measure the voltage of the old ones. Wildly varying voltages are a sign of bad manufacturing process handling, or originate from mixing different production batches (which is minimized by only combining batteries from the same package).
Energizer Max has a leak warranty:
https://www.energizer.com/about-batteries/battery-leakage
Alkaline cells have so many issues. It’s really time for a new battery standard. AAs have been wonderful but we can do better.
For one thing, you have to put them in the right way. Devices have to be designed with protection, and users have to stop and think about it. It’s also just an unnecessary hassle for some products to have extra wiring because connectors are on both sides.
The contacts are exposed and can start a fire if you dump a bunch of them in a bucket.
There’s no compatibility between sizes without adapters. A more advanced standard could allow the AA equivalent to also accept the AAA equivalent, so you could use a single small size for all your stuff if you don’t need the extra power.
There’s no onboard smarts, they’re choking hazards, they’re round which is a slight waste of space and makes stuff like onboard USB ports have ackward angles… There’s a lot that could be improved.
XKCD, 15 standards
“Alkaline cells have so many issues. It’s really time for a new battery standard. AAs have been wonderful but we can do better.”
They eventually leak, but they don’t explode!
Even if shorted, Alkaline and Carbon-Zink types “just” get hot – and catch fire in an extreme case.
But they don’t detonate (normally).
Lithium type batteries that are designed to replace alkaline types do have protective-electronics built-in (for example, the lithium 9v battery).
To prevent explosion and other hazards.
Unfortunately, electronics may fail. Unlike natural, physical/chemical processes.
LTO batteries have similar safety profiles to alkaline as far as I know, and even conventional lithium fires are still surprisingly rare considering they are everywhere.
9v batteries are not entirely free of fire hazards either, so it’s possible that something like LTO or even LiIon plus a protection circuit could be safer.
But we all know that those will only last for a few months in storage before dropping in voltage below the minimum recovery cut out, and that’s the end of that device for 99% of users.
I have a pile of stuff that needs a new internal lithium battery – everything from a hoverboard with a 36V stack of 18650s that fitted after a month in storage, to little usb charged LED lights. Currently, I have only one thing with alkaline corrosion to sort!
There are lithium based devices that have been fine for years in a drawer. It’s just a matter of how good the electronics are at not draining them when idle.
For a new standard, why not create rechargeable 4,5v lantern batteries?
https://en.wikipedia.org/wiki/Lantern_battery#4.5V
The 3R12 used to be popular in Europe and the Soviet Union, comparably to the 9v type in the states.
It was such a kind and forgiving battery! And lasting, powerful.. Even if only made of carbon-zink.
It was THE battery used in school, in electronic construct kits etc.
Such a shame it was never produced as a rechargeable!
Let’s think about it! The R12 was the original thinkerer’s battery in Europe.
At 4,5v, it sits between 3,3v and 5v (TTL), can power both CMOS and TTL devices directly.
And it roughly had 1,7 Ah to 6,1 Ah, depending on the material used.
That’s great. And at roughly 5v, it’s not lethal yet.
The coal-zink types were sonetimes dismantled to get the carbon pens.
These carbon pens were used to build carbon microphones and sich things.
It’s surprisingly close to a lot of modern proprietary batteries. The connector on top is just all kinds of awful, just asking to be shorted out(Plus it lacks a thermistor/data pin) but otherwise it would make a pretty nice form factor.
The only issue is what kind if devices need something so large? It’s too big for handhelds, too thick for laptops, and it’s not really needed in power banks because power banks pretty much *are* already a new standard battery type.
The only thing I can think of is speakers and UPSish applications, It would be a good standard for non-portable stuff.
My favorite was the EN6 – a spraypaint can-sized carbon-zinc cell with screw terminals. They were great for high-current stuff like electromagnets, and when they finally wore out you could extract a massive carbon rod from them. Somewhere in China they’re still being made, you can find them online.
Thanks, I looked up EN6 and learned about Fahnestock Clips
Most of the experiments in kids books on electricity when I was in grade school in the ’60s used these batteries. Some had Fahnstock clips, others had knurled metal nuts for terminals. they were for sale pretty much everywhere then. They were often called “Ignition Batteries.”
I am Spanish, I still remember the pocket flashlights of my grandparents (in Spanish they are called de petaca) that used the 3R12 batteries that you mention, unfortunately those batteries were too big to continue being sold nowadays, if you like that type of batteries I recommend you that you use the 6LR61 batteries they work at 9V, they are smaller and are still being sold.
Actually there is a polarity protection: the + has a bump on it, and the battery holder has plastic bars next to the metal on the + side, so if the battery is backwards there is an air gap between the contact and the – end of the battery.
I agree that this protection always misses when you eventually put in the batteries backwards, but it surely is there, when you try to use batteries designed with solder strips (which are therefore missing the + bump).
You lost me at “Choking hazards.” Who is going to choke on a AA battery?
If your concern is toddlers, then the issue is not a design flaw in the battery. It’s an execution flaw in parenting.
Redesign of AAs might address some of your more well-placed criticisms, but it won’t render the toddler-scape safe from stones, erasers, coins, keys, paperclips, fridge magnets, nails/screws, or ANY of a hundred kinds of food or candy that an unsupervised infant could choke on.
The EU has long mandated screw or other types of toddler-proof battery compartments on toys!
Now I wonder if dielectric grease would be a defense against it.
Petroleum jelly. Cheap and available.