Platinum-group metals (PGMs) are great catalysts, but they’re also great investments — in the sense that they are very, very expensive. Just ask the guy nicking car exhausts in the Walmart parking lot. If one could replace PGMs with a more common element, like, say the aluminum that makes up over 8% the mass of this planet, it would be a boon to the chemical industry, and a bane to meth addicts. Researchers at King’s College, London have found a way to do just that, with a novel form of aluminum called cyclotrialumane.
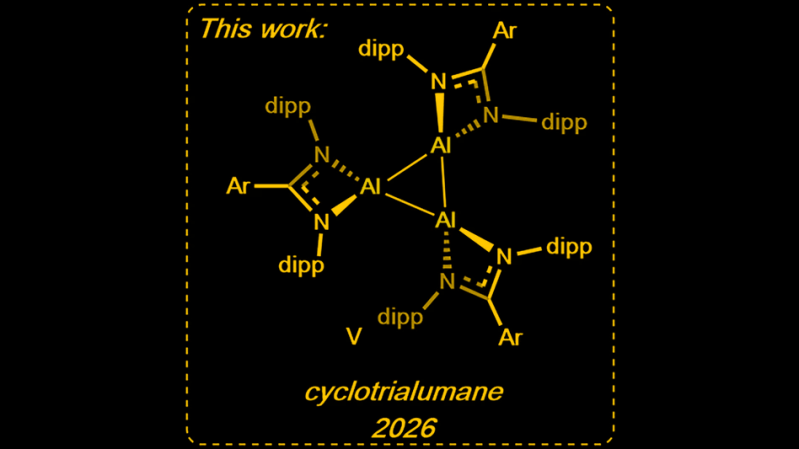
The aluminum trimer is exactly what the ‘tri’ in the name makes it sound like: three aluminum atoms, bonded in a triangular structure that is just pointy and stick-outy enough to poke into other molecules and make chemistry happen. OK, not really — you can see from the diagram above it’s not nearly that simple — but the point is that the shape makes it a good catalyst. The trimer structure is useful in large part because it is very stable, allowing reactions to be catalyzed in a large variety of solutions.
The researchers specifically call out their trialuminum compound as effective at splitting H2 in to H+ ions, as well as ethene polymerization. Both of those are important industrial reactions, but that’s only a start for this trialuminum wonder catalyst, because the researchers claim it can catalyze totally new reactions and create previously-unknown chemicals.
If you never took chemistry, or it’s been too many years since you last slept through that class, we have a primer on catalysts here. By accelerating chemical reactions, catalysts have enabled some neat hacks, like anything involving platinum-cure silicone.
Thanks to [Lightislight] for the tip! Hacks do appear here on their own, but you can always use our tips line to catalyze the synthesis of a particular article.
Header image adapted from: Squire, I., de Vere-Tucker, M., Tritto, M. et al. A neutral cyclic aluminium (I) trimer. Nat Commun 17, 1732 (2026). https://doi.org/10.1038/s41467-026-68432-1















Won’t someone please thing of the meth addicts? :)
Can’t someone invent a catalyst that splits methaddicts back into humans and nasty chemicals?
Cathepepsins and collagenases? Unfortunately the resulting human retains only limited functionality.
That’s just proper universal health care you’re talking about.
Maybe it’s not cool for me to comment on this because I submitted it as a tip but, this is exciting.
Once upon a time I worked with some synthetic chemists. One of them told me after getting their degree they went into industry and worked at a lab with considerable precious metal catalysts. When you spill any of these reagents on say your shoes they take them and burn them in a furnace to recover everything they can. Think of the chemists shoes here people.
But more seriously, the first portion of this is very approachable to a hobbyist trained in the arts. Some of the other portions less so, but nevertheless I hope we see more approachable catalysts either financially or in the ability to make them. The implications for whacked out energy schemes or mad scientist shenanigans pleases me.
by “exciting” you mean, that it is like supraconductivity of chemistry and a ticket for a Nobel price. ;)
must say, you had me at “The implications for whacked out energy schemes or mad scientist shenanigans pleases me.”
Rocket belts
I wonder, what catalyst is optimized for the process of synthesizing a chemist?
In before Nile Red synthesizes it with a strange Chinese machine, or Explosions and Fire spends a day complaining his side product is yellow.
this is definitely an interesting one to look into further. although there are multiple alternatives to pgm catalysis in various reaction schemes, industrial (mainly petrochemical) use still heavily relies on pgm’s.
i wrote an article on kitco years ago detailing the 5-10 year market cycle between platinum and palladium – the gist of it is that, historically, when one is high the other is low. industry and academia take note of this, and the cheaper one becomes the highlight of new research until it dethrones the other. prices swap, and the cycle repeats anew. rhodium has always been signifigantly costlier than either, so it is generally immune to the cycle.
lately, though, with all the fear-mongering around the world, all precious metals have risen/fallen in lockstep with no industrial use considerations behind them. in this new scenario, we may finally see actual investment into non-pgm catalysis. nickel, iron, complexes like this one may actually be used outside of the lab in a meaningful way.
I invested in platinum, the stocks are really down. Hope Pt goes up
As someone whose chemistry knowledge stopped at a single class in undergrad, this is fascinating.
How do you even product these special molecular shapes without an impossibly large amount of trial and error?
There is a lot of theory behind chemistry. Even still simulating molecules accurately is a huge nightmare. There are modeling methods that give good enough approximations though. Big hint they don’t involve AI it’s physics and high performance computing.
A lot of early days chemistry was trial and error. Think alchemy. With the foundations of quantum mechanics but lacking compute there were a lot of rules and patterns. After the advent of computers and a lot more theory, we can get pretty good guesstimates of things. A lot of chemistry is building off of others discoveries though by established theory. It’s really really hard to foresee all the ways chemicals will interact, and empirical results are king.
A lot of the fun stuff is discovered by happenstance still. That doesn’t mean chemists are gamblers, but sometimes a student puts a voltmeter to a pile goop and discovers they made a conductive polymer. They don’t publish it like that, they follow up with rigor and measurements.
This however, if I were guessing, it looks to me like someone, likely the PI, had a strong intuition about this being possible, perhaps did some modelling and tried a half a dozen ways to see if it could be done. You’d have to ask the authors though.
I learned more about chemistry while professionally brewing beer than I ever did in a undergrad class.
The head brewer had been through several classes taught at hop farms and breweries in Skagit Valley. The way he described it, the esters phenols and flavonoid interactions are almost impossible to predict without a lot of heavy lifting on the computer side of the work, and even then the outcome was only a model informed guess. The Maillard reaction in the mash tun and kettle only added complexity to things. Yeast fermentation puts yet another spin on it.
You can bend the “brew rules” pretty hard by keeping the kettle at around 200 to 205 for an hour and never letting it boil and chuck all the interactions you think you know out the window.
There are Nobel prizes to be won in studying beer and hop chemistry.
Interesting stuff. You know what is funny? We still don’t fully understand how smell works. And smell is some large percentage of taste. Nobel prize right there.
Brewing has a lot of complexity. I’ve done some work on technologies that could aid that sort of thing but you are right, at the end of the day, it’s incredibly difficult to do predictive work there.
As I was researching quantum computing for its impact on cryptography, I found the projected uses for quantum computing include “green chemistry” (among many other broad topics.) If quantum computers can deliver on even half their promises, it’s possible new formulas will be found to produce other inexpensive catalysts like this, to reduce the use of hazardous chemicals, or to find formulas that produce less hazardous byproducts.
Seems like a pretty exciting time for chemists.
I would say maybe. Quantum computing could be powerful and it has basis in N body type problems which plague a lot of chemistry simulations. The solution spaces can still be insanely vast. I am a little skeptical at anything beyond greater throughout unless there is something I don’t know about.
There are three things about chemistry a lot of people including myself forget about. One is that, predicting what happens is hard and sometimes it’s faster to go into a lab and just do it. Especially with combinatorial equipment. Another is, even though when know quantum mechanics really well, we are still surprised by things that happen in chemical reactions. Even reactions from the 1800s, text book stuff, we don’t know how they actually happen at the quantum level. A third is that, the human element/physical experiment is often more telling than anything in silicon. The tiniest bit of humidity can make or break something, a slight change in solvent can change a reaction. Someone noticing something happening in a flask or a material property can be the difference between chemical waste or a huge discovery.
I think more computational methods are always welcome. But I think we need more asymmetrical thinking, freedom to explore or re explore old tracks and of course funding to really progress these things. So far I’ve seen the more automation heavy groups not produce big things with big funding and the more creative teams continually churn out important stuff doing cowboy chemistry. Not universally, but I think the human element needs more reinforcement than the computational one at this point in time.
Tons of labs had to shut down recently and are getting by on almost nothing at all. I am not convinced we can compute ourselves out of the deficit many institutions are in. Not in the next 10 years anyway
I kept awake in my Chem classes but early morning economics was tough to not snore. China reports that they have a hexagonal diamond which is harder than natural diamond, Chem or physics? And now there is lab grown gold, and gemstones. Chem class is extremely useful, I even use it every day – 3 scoops of coffee grinds for a pot of coffee. I usually get it right unless hung over. Then there is the tricky Chem formula of dash of NaCl for scrambled eggs.
“…with a novel form of aluminum called cyclotrialumane.”
Sounds like the Enterprise crew needed another whale.
Sounds to me like Kira needs a new phaser rifle:
Cyclotrialumane, count to four,
Cyclotrialumane, then three more,
Cyclotrialumane, if you can see,
Cyclotrialumane, you’ll come with me
Can this replace platinum in cars?