Few days are worse than a day when you hear the words, “I’m sorry, you have cancer.” Fear of the unknown, fear of pain, and fear of death all attend the moment when you learn the news, and nothing can prepare you for the shock of learning that your body has betrayed you. It can be difficult to know there’s something growing inside you that shouldn’t be there, and the urge to get it out can be overwhelming.
Sometimes there are surgical options, other times not. But eradicating the tumor is not always the job of a surgeon. Up to 60% of cancer patients will be candidates for some sort of radiation therapy, often in concert with surgery and chemotherapy. Radiation therapy can be confusing to some people — after all, doesn’t radiation cause cancer? But modern radiation therapy is a remarkably precise process that can selectively kill tumor cells while leaving normal tissue unharmed, and the machines we’ve built to accomplish the job are fascinating tools that combine biology and engineering to help people deal with a dreaded diagnosis.
Controlled Killing
Simply defined, radiation therapy is the application of specific kinds of ionizing radiation with the intent to treat a disease, which is usually but not always cancer. This differs substantially from using radiation to gather diagnostic information, as in the case of medical X-rays, CT scans, and nuclear medicine. All ionizing radiation has the potential to cause cellular damage, but in diagnostic radiology, doses are kept as low as possible to protect against cellular damage while still getting the diagnostic information needed. Radiation therapy, however, uses doses of ionizing radiation with the express intent of killing cells in as controlled a manner as possible.
To understand how radiation therapy works, it’s important to know a few simple facts about cancer. Cancer is not one disease, of course, but all cancers share a trait: uncontrolled cell growth. Cancer cells divide more or less continually and proliferate, often forming solid masses called tumors. Cancer cells also tend to be relatively undifferentiated cells; that is, they lack the specialized structure and function of normal cells.
These characteristics provide weaknesses that can be exploited for therapies. For cells to divide they must replicate their DNA, and while DNA is replicating, it’s particularly vulnerable to damage. Damage can come from powerful drugs like those used in chemotherapy, or by exposure of the cells to ionizing radiation. Damage the cell enough and it dies. Damage enough cells and the tumor starts to shrink.
Cancer cells are more vulnerable to damage from drugs and radiation because they are replicating more rapidly than the surrounding normal cells. But the normal cells are replicating too, and can incur collateral damage while the tumor cells are being targeted. Being able to spare the surrounding tissues from damage while killing the tumor is the goal of any cancer treatment, and this is where radiation therapy shines.
High Energies
Radiation treatment works by forming a very precisely shaped beam of ionizing radiation that can illuminate a tumor without exposing the surrounding tissue. The problem is that bodies are three-dimensional structures, and no matter which way you aim a beam, normal tissue will be either above or below the tumor, and will get dosed. Luckily, cells are sensitive to accumulated doses of radiation, so it’s possible to deliver a partial dose from multiple angles, limiting the damage to structures above and below the tumor. This is accomplished by precisely rotating the therapy beam axially around the patient with the tumor at the focus. Over time, the tumor builds up enough of a dose to start dying, while the surrounding normal tissues are spared lethal doses.
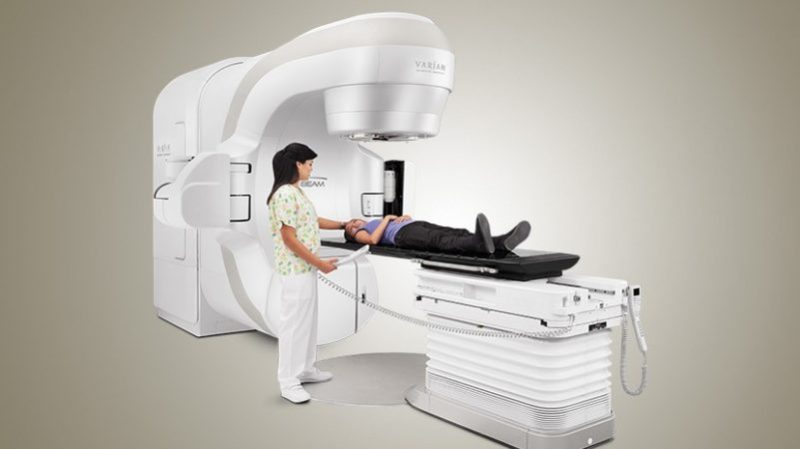
The machines used to deliver external beam radiation therapy can be immensely complicated. Not only do they have to generate extremely powerful beams of ionizing radiation, they have to control, filter, and shape the beam. They’ve also got to position the beam with extreme precision so that the treatment plan developed by the radiation oncologist and the medical physicist is correctly executed. On top of that, the machine has to have multiple redundant layers of safety interlocks to protect the patient and technicians from potentially lethal exposures.
Options for sources of ionizing radiation vary, with different sources offering different therapeutic options based on the energy of the photons they produce. Orthovoltage X-ray machines are essentially high-power X-ray tubes that generate beam energies in the 200 to 500 kiloelectron volt (keV) range. Like diagnostic X-ray tubes, orthovoltage X-ray tubes work by accelerating electrons into a tungsten target to create powerful beams of photons.
Further up the energy scale are the linear accelerator machines, or linacs. These can provide either X-ray beams or electron beams in the 4 to 25 Megaelectron volt (MeV) range. Where the electron source in diagnostic and orthovoltage X-ray tubes is generally a simple hot cathode design, linac electron beams are produced by injecting electrons from a tungsten filament into a long waveguide. A magnetron produces a standing RF wave inside the waveguide which accelerates the electrons to huge kinetic energies. The electron beam can be used directly, or the beam can be used to strike a tungsten target to produce a high energy beam of X-rays.
Staying in Shape
One of the most interesting parts of a radiation therapy machine is the collimator. Collimation controls the shape of the beam and limits unwanted exposure. A diagnostic X-ray machine’s collimator is simple — two sets of lead or tungsten leaves set 90° to each other can be moved in or out of the beam and produce a rectangle of various sizes. A radiotherapy beam needs to be able to match the irregular profile of a tumor, so a multileaf collimator (MLC) is used instead. An MLC has a large number of tungsten plates that can be moved in and out of the beam to control its size and shape. The MLC is set to project a beam based on the two-dimensional profile of the target tumor as seen from a certain angle, which changes as the beam is rotated around the patient.
MLCs are highly engineered mechanisms. Not only must every leaf be precisely and accurately positioned to match the therapeutic plan, it must do so while being bathed in high-energy photons. The leaves have to interlock and overlap so there’s no leakage between leaves, but thermal expansion must not be allowed to jam the leaves. The leaves also have to be thin enough that the “pixelation” of the edge of the beam is minimized.
https://www.youtube.com/watch?v=Llxe9t8wwB0
These aren’t the only therapeutic modalities available for radiation therapy, of course. Some external beam therapies use a fixed radioisotope source like cobalt-60 rather than an accelerator, and some therapies use beams of particles like protons to kill cancer cells. But no matter the physics behind the treatment, the engineering that goes into controlling beams of lethal radiation to kill only what needs killing is something to admire.
[Featured images source: Varian Medical Systems]















No article on radiotherapy is complete without mentioning Therac-25: https://hackaday.com/2015/10/26/killed-by-a-machine-the-therac-25/ and the Leveson report that’s required reading for any firmware developer: http://www.cs.umd.edu/class/spring2003/cmsc838p/Misc/therac.pdf
Also, this non-rendered video of the leaf collimators in action is mesmerizing: https://www.youtube.com/watch?v=KJst8hpw1z0
Therac 25 is undeniably a disaster….however that particular disaster was software that drove the hardware poorly. IEEE did a great review and root cause analysis. I’m afraid we have learned very little and medical software is still a dicey business. Not enough copies to justify high coding standards.
“Few days are worse than a day when you hear the words, “I’m sorry, you have cancer.” Fear of the unknown, fear of pain, and fear of death all attend the moment when you learn the news, and nothing can prepare you for the shock of learning that your body has betrayed you.”
Considering how complicated our insides are, it’s amazing more doesn’t go wrong. We’re like a chemical factory with a low accident rate.
We’re a chemical factory with no central management, and everything is made of gelatin and gravy.
Technically, even if distributed or localized management throughout the body… the genomics DNA is the leadership, transcriptome RNA is the management making sure replication is correct, the proteome is next in line like maintenance making sure the system doesn’t fall apart with the metabolome (nucleotidome, peptidome, aminome, lipidome, carbome, vitaminome and mineralome) providing raw materials for intermediates and finished products for operations along with operations support. What seems chaotic is actually very well known and documented.
The knightmare is the bio-equivalent medicine isn’t so well diagnosed and used for treatment.
Petrochemical synthetics and worse seem to dominate the first line of defense… almost like a mild form of armed robbery with masked assalants.
That is why I was grilled with a confident awareness that M.D. Researchers are the MOST FRAUDULENT unethical researchers on the planet. I’d say J.D.’s are the next though I wasn’t trained in that way. However from experience and my reading comprehension skills along with Lean Sigma Training and Leadership is a Hardware and Software 21 CFR 210/211/820/11 compliant Quality and AR&D environment… I can confidently make that observation at least in the majority of the lower level jurisdictions I’ve experienced.
I got 48 treatments in a machine nearly identical to the one in the top image, in 2011. (Stage 3 lung cancer) I kept telling them I was going to bring tools one day so I could take it apart to see how it worked. ;) The IMRT is an absolutely amazing bit of hardware and it did an excellent job in killing the cancer. (It did leave me with a mild “sunburn” on the lining of my throat though…Made eating a challenge towards the end of the treatment cycle…)
This statement is incorrect – “radiation therapy is a remarkably precise process that can selectively kill tumor cells while leaving normal tissue unharmed”. Radiation therapy treatments are designed to minimize the damage (radiation dose) to healthy tissues and maximize the radiation dose delivered to cancer cells (e.g. a cancerous tumor). Collateral damage to some healthy body tissue (cells) is usually inevitable during radiation treatments.
Compared to Chemo it’s precise. Chemo is a race, “Consume this poison, with any luck the cancer will die from its effects before you do.”
As someone currently on chemo for cancer, I’d say that statement isn’t entirely correct. Having lost my right kidney, part of my liver and about half an adrenal gland during surgery for RCC (Renal Cell Carcinoma, stage four) I’m happy to say that not all chemo is as difficult to deal with as one might imagine. What I’m taking now is working and working very well. Three months ago, I was declared tumor free and I’ve managed quite a normal quality of life after the surgery (Oct 20th., 2016) and if I was faced with the same situation again, knowing what I know now, I’d do it again in a heartbeat.
Currently, I’m taking a Sutent (37.5 mg) capsule daily and very recently had it explained to me that although it’s not necessarily a “cancer killing drug” is is, instead, a blocker that prevents any further cancer from developing. It also helps deprive the existing cancer cells from acquiring what they need to continue to live and as a result, they start to die off while being replaced by healthier, non=cancerous cells. What I do know, is that I can’t recall ever having as much energy to burn or feeling as well as I do know. To me, it’s like having regressed to a ‘much younger, healthier self’.
Medicine has made some major strides with new medications over the last decade or so, and I feel as though I can several more years to my life’s expectancy even though this type of cancer doesn’t ever go into remission (or so I’ve been informed).
The whole point is, besides X-ray and other forms of radiation treatment are still available if it’s needed, but at this point, I’d much rather take a daily pill at home than having to make countless trips back and forth for some type of radiation treatment.
By the way, after the surgery I was up and walking two days later and back home a few days after that, and I haven’t slowed down since. Again, having gone through all that and dealing with my cancer as I am, neither the surgery (after the fact) or what I’m doing now wouldn’t cause me the slightest bit of hesitation if I was to be confronted by the same scenario all over again. A lot of worry over nothing. The only difficult thing I’ve had to seal with while in the hospital was the bland food (had toe foresight to take a salt shaker with me and hide it) or deciding which daytime program wasn’t so boring I couldn’t watch it.
Even if you are able to precisely deliver radiation to a target volume (cancerous tissue), it doesn’t imply that healthy tissue isn’t affected. I wasn’t comparing radiation to chemo treatments. I was just stating that there was an incorrect statement made in the article. Basic radiation physics/biology are at play. To say that normal tissue in unharmed by radiation treatments is wrong. There will always be healthy cells that are damaged. Even with proton therapy, there is some energy loss (dE/dx) when the proton first hits surface tissue and the underlying healthy tissue before the cancerous target volume is reached. That energy loss causes cellular damage – to any healthy cells. This is true of any form of ionizing radiation used for cancer therapy.
protontherapy has a 0.1mm precision
I work on a radiation therapy machine, a proton therapy one. And it’s amazing how different it is compared to the linacs with photon radiation. Here’s a link to a brief video showing it. https://vimeo.com/80077597
You too!?! I first engineering job was also working on an improvement to proton radiotherapy.
My first job was to enhance a positioning chair in a protontherapy center
Ha ha, cool! My first job was to create a system for real time proton beam tracking (and dosimeter) for a scanned and modulated small proton beam. (At the time they were still using huge apertures to shape a large uniform beam.)
I helped commission a 4 gantry proton therapy system that boosted the linac output with a synchrotron.
If you want that extra dimension of precision you need Proton Therapy. Since it can control the irradiation even by depth using effect called bragg peak. https://www.nature.com/news/three-ways-to-make-proton-therapy-affordable-1.22660
Also check youtube for proton therapy, but simplified explanation is depositing radiation through human tissue in a 3Dprinter like manner without significantly irradiating the tissue between output nozzle and the tumour.
My father, who was one of the inventors of the side-coupled RF cavities that are the standard design for radiation therapy linacs, also did research in the 70s on pion therapy, which is like proton therapy on steroids. If you think the Bragg peak for protons is sharp, you should the dose deposition curve for pions! Unfortunately, making pions is prohibitively expensive and the results of clinical trials were not very impressive, so it is no longer considered a viable option
Interesting is the elementary di-particles that most have never heard of that are created by elementary particles.
Years ago I, complete with a “target” drawn with sharpies, made the daily trip past a massive cement door, down a cement corridor, around a wall and onto the gurney portion of a machine not unlike that pictured. Once the sharpie lines were aligned with the laser beams firing in from 3 or 4 places in the room, the medical staff would leave – assuring me they would be back soon if anything happened.
Being a geek – I was fascinated, intrigued and amused.
Being human – I was clearly aware of how deeply, freakishly unsettling this whole experience could be.
It seemed to have worked. Haven’t been back in over 20 years.
And my supportive friend in university during the time was kind enough to stick a copy of this in my locker:
https://img0.etsystatic.com/002/0/5599387/il_570xN.382517436_sy36.jpg
It’s good to have friends ;-)
ObXKCD: https://xkcd.com/933/
That reminds me of the saying;
“Scars are like tattoos, but with better stories.”
I have read about some more exotic treatments, perhaps experimental. These, involve giving the patient a “safe” substance intravenously. The body absorbs the substance, and the cancer, being greedy, absorbs it in larger quantities. When the area is irradiated, the previously “safe” substance transforms into a nasty poison, killing cells around it. Basically focused chemo. Unfortunately I can’t remember the substance itself.
UW Madison Medical Physics program had some research going at one time using something like you note where I want to say they were using IR Lasers with success.
Basically, what I envision you are describing is another one of those methods that I’ve dreamed about since back up at Tech and younger using Quantitative Structural Activity Relationship site specific agents to bind to specific targets so to either as a chemotherapeutic agent destroy the undesired growth or use as a contrasting like agent to resolve out the detail of the target so to use an active imaging and resonating non-ionizing radiation to destroy the undesired growth.
What you are thinking I think is more for ionizing or particle radiation.
I’ve envisioned these methods for use for sure now I think about more before Tech after reading “Lasers: The Light Fantastic” back in 4 or 5th grade. Man, I don’t even think there are AWARE classes anymore around here.
Probably because how dangerous growing up and now experienced with my parents and grandparents being targeted and most likely their valid generation being forensically clean murdered by the NAMBLA mafias, terrorists and war criminals that are blatant careless dumb murders trying to act all cute, sweet and innocent.
there are some methods that “freeze” (cryosurgery) or “cook” (RFA) the cells inside the tumour in a more traditional way. and once they are dead, the body does the garbage collection procedure on them. it is maybe an alternative to the surgeon who needs ~1inch extra perimeter for the clean cut or in harder to reach places.
Another type of therapy that is interesting is the Gamma Knife ( http://gammaknife.com/ ) and Cyber Knife ( http://www.cyberknife.com/ ).
Does anyone know what is the smallest resolution delivery system that can target growths specifically, accurately and repeatably (I suppose other quantitative method validation parameters can be tested too)?
Strange to me that there are not more real time imaging and emission systems. Seems almost like there is something fishy as is with the drug industry where like most systems if not all can and legally are supposed to be used as medicine though are used to poison and kill in some if not many cases in certain studies.
Proton therapy has sub mm precision in all dimensions(even tissue above tumour is significantly less irradiated), is non invasive(no operation or anaesthesia) and in can be done in 2 months with 15min therapies for work day.
Technology has matured less than decade ago, that is why it is only now you are starting to see mass adoption(also because it requires custom building for the machine).
Interesting. I recall reading years ago that the proton therapy was the first radiation treatment that wasn’t so dangerous where one of the pioneers invented or used for his mother or grandmothers treatment. That’s great there is 0.1mm precision and penetration effects. I assume there are contrasting agents also to aid… though I need to read on how the systems actual function in regards to their effect causation.
Cancers of the esophagus or other organs that
move with breathing, heartbeat, or peristaltic action are difficult to treat with proton beams. Precise targeting requires a stationery target, although the last I read, Japan now allows for a system that tracks and predicts the location of the tumor for proton firing.
Is the issue the relaxation of the sensors? The resonant frequencies of the different human body frequencies you noted can be easily detected and predicted I’d think other than having to train and model the prediction algorithms to accommodate for changes from the proton emission effect on those organs resonant processes.
Oscillations are not that complicated. Other than timing issues with transmission and detection systems sensors like relaxing or maybe a change in the sensors relaxation version complete (I am forgetting the term that is opposite of saturation) discharge I will use.
Then I’d guess the speed in the controls systems positioning motors in the X-Y-Z or whatever coordinated system being used in relation to the human body resonant frequencies. Doesn’t seem that complicated.
Edit: “… sensors relaxation “version”…” with “versus”
I’m two months past a CyberKnife treatment for an egg-size meningioma just above my left eye. I’ve gone from a #8 headache with 10-12 hours of deep sleep and 24 hours of garbled speech about every five days to a #2 headache with 3-4 hours of sleep and half an hour of dysphasia every two weeks and getting milder all the time. The technicians were delighted to get a geek to work on and showed me everything about the facility. The CK has a set of about a dozen tungsten collimators to choose from and can sense the difference between brain and tumor tissue to compensate for movement from respiration, etc. The treatment was for five 45-minute sessions. I had no discomfort whatsoever, though I was a bit fatigued for a couple of hours following a session. Verdict: more uncomfortable than a haircut but nowhere nearly as bad as a dental checkup.
That’s what the systems are for and neat to read some are opening up to uses other than brain as I recall they used to be only used for brain when my Dad was going through treatment that was no way localized and I even wonder with the Lakeland Healthcare network… required. Great to read you are in the correct trajectory (i.e. maintenance, treatment & improvement).
Imagine like radiation therapy being around since the early 1900’s and the pathetic advancements in health an safety in some locations and situations…. laser therapy not being implemented and replacing broader beamed not as site specific radiation therapy since safer and healthier is even more disturbing. This documentary demonstrates advances circa 1967: https://www.youtube.com/watch?v=ssg67GLfTtw
There are even FDA approved light therapies that are OTC that NASA even validated.
What happened to advancing safer therapies?
Like noted around the 13:20 minute point as well as other points in the above referenced video.
Personally, I think intersecting multiple lasers is the way to go so the threshhold is specific more at the intersecting target point also noting potential for timing to ablate and de-gas so not to air/gas embolize or cause ischemia elsewhere, where the later ischemia is easier to prevent with medications… like even with naturally occurring in the body compounds found in weed or vitamin E. Pathetic. I was just reading another study noting less hospital admissions and post surgery complications when using cannabinoids. I do recall liberals purging Stanford’s valid research data in the late 60’s or 70’s and wonder if some of the basic healthcare methods used in advanced ways were also purged.
Seen a lot of remarks in comments about protontherapy and happy to discover former colleagues.
Protontherapy doesn’t only use a collimator which is going to conform to thee 2D profile of the tumor seen from the beam incidence (let’s say it’s X and Y coordinates).
It will also use a modulator, a high-speed rotating disc with variable thickness that will allow to put a spot along the depth of the tumor (the coordinate Z).
As the tumor is not a perfect cube, depth is going different along the X and Y coordinates so protontherapy will add a bolus to fit to the 3D shape of the tumor
https://image.slidesharecdn.com/the-use-of-highenergy-protons-1233610364226521-2/95/the-use-of-high-energy-protons-21-728.jpg?cb=1233588996
This balistic precision is the main reason why protontherapy is the preffered treatment where precision is even more vital than usual, near cortex or spinal cord.
Man, seems like the proton therapy can be improved even more with real time imaging and also beam splitting to have the different beams convergence point be the target growth to destroy.
That’s great the penetration isn’t so deep and can be tuned based on energy intensity to a desired depth.
Now, in regards to the source initial energy and path in between… there seems to be room for improvement. Modeling of the tissues in the path before the target along with phase alignment to assure amplification on not cancellation would be the more challenging aspect of my new design idea.
Interesting… I haven’t read about the proton therapy in a while and I guess wasn’t as dangerous or something to that matter where the most dangerous ionizing and chemical treatments need improvement or different methods used entirely that are known to be more effective, healthier and safer. I had to read the wiki ( https://en.wikipedia.org/wiki/Proton_therapy ) and am still not sure the exact reason the DNA is damaged… I assume the proton and ions break the DNA/RNA bonds, damage tissue in general and cause immune system responses to be more aggressive to detect issues and repair issues on a more local level if the target to be destroyed has adapted and integrated with the vascular system.
From the blog;
“Cancer cells are more vulnerable to damage from drugs and radiation because they are replicating more rapidly than the surrounding normal cells.”
Oxidants damage DNA by oxidizing DNA material, basically it rips molecules apart faster than the DNA can repair itself. That is why anti-oxidants are important for maintaining healthy cells. But when proton therapy damages tumor DNA, it helps to have oxidants “at the ready” to hinder tumor DNA repair.
Ionizing radiation is causing oxidation and reduction reactions to occur. Duh… that makes sense. I used to think of electron beams that are at such high frequency that they act like bullets and literally blow nanometer or smaller size holes in their trajectory. They may do that also… though I am thinking they cause electron transitions in orbitals to increase to the outer orbital or be free, like that range of Quantum Energy does in regards to electronic transitions.
Flo rockin the proton therapy yo!
I’ve never been able to view it as a life-saving therapy. It seems to primarily be used when chemotherapy has failed and the patient needs the blessing of pain relief from metastatic tumors, if the patient has sufficient insurance or funds.
The last I heard, Mayo Clinic charges insurance companies the same for radiation or proton beam therapy (PBT). It keeps the insurance companies from denying PBT because of the cost.
check this one out implanted beta radiation in trials now.
https://www.youtube.com/watch?v=A2yMwoK3xp4&feature=youtu.be
I can’t wait for nanotechnology to get to the point where we can inject teeny tiny machines carrying small radiation sources directly into the tumor.
Wouldn’t it be more effective for them to deliver poison to the tumor?
Technically, from what the vision of Opnwatr.io is, there must be some sort of way just with resonance to not cook or poison the tissue at the cellular level… there must be a way to instruct the cell what to do. That is either morph into the correct tissue or die.
That is my interpretation of the electromagnetic effects of non-ionizing radiation at specific quantitative frequency or frequencies on cells. Sounds like a research project and paper in the waiting.
That frequency and charge with potential 4 dimensional field effect(s) is like the current of injury that Dr. Robert O’Becker and Gary Selden wrote about. There is the potential for laser tweezers and scalpels though the later two devices are more morbid in my opinion even though not compared to modern methods.
Exactly!
Nuclear Medicine does exactly this, and has for a long time. The nucleatide is mixed with chemicals that will carry it directly to a specific organ once injected. But it’s a tiny dose just enough for the gamma camera to develop an image of the tumor(s). It’s not life preserving as too little to affect a tumor, but does let a physician follow the spread and growth of tumors to better predict the patient’s probable expiration date. Not of much use to the patient but does generate a cash flow. It was all the rage back in the AIDS epidemic. Then you go back home and hug the kids… and over the next few days the nucleatide goes down the toilet.
It’s value is in places like accident victims with a damaged liver, the portion that is still alive will take up the nucleatide and be imaged but dead portion took up none and will be dark. Another winning application is hairline bone fractures. These do not show in an xray but a nucleatide is absorbed there and makes these areas easily visible.
As a radiation therapy it is not workable as the radionucleatide levels are quite low.
After a certain development into my chemistry background (reading the Journal of Medicinal Chemistry, et.al.)… I was amazed at the ability to clearly use radio-isotopes as tags to some molecule, like you note Biomed for nucleotides, for tracing metabolic routes, organ absorption and radio-contrasting.
My focus at one time was to perform computational chemistry of binding for Quantitative Structural Activity Relationships (QSAR) to screen bio-equivalent drug candidates in particular using human nutrients as the parent molecule with some other mineral or molecular entity combined that created a stable marketable patentable medicinal entity. Even with radio-isotopes used for treatment; say like not radioactive potassium iodide taken to prevent too much absorption of the damaging radio-isotope iodine protocols… similar pre-chemo treatment can be performed to prevent other organ damage for the terminal chemo entity methods. Non-terminal entities require more thorough chelation, absorption or binding operations if ever conceived… though is interesting the different preparatory entity methods that seem to be ignored for the most part in regards to optimal health and bio-equivalent medicinal treatments.
I think that got me on the RADAR/SODAR big time since like is noted… “cash flow competition” and when dealing with some industries later… in particular a State of Michigan Congressman at the Capital during a model legislature event… I realized there is also a Funeral Home, Cremation, Cemetery Industry that is influencing modern society in regards to influencing administration of Boards, Commissions, etc. due to their, like the Defense Contractor Industries not targeting outside the U.S. for U.S. Defense interests and concerns for their cash flow and performing OFFENSIVE strategies to create business.
I don’t think this is the case in all jurisdictions… though has been changing in the U.S. post 1870’s immigration waves and Roman Catholic Ecclesiastical Provinces descent (Axis POW’s, mafias, pedos, etc.) taking over the traditional W.A.S.P. communities with malicious Tribunal operations in our Courts with their no way Justices of the Peace or Peace Officers. Opiate or depressant epidemic leadership big time pulling the wool over our eyes!.
Interesting demonstration in general and with some info at about 11 minutes in on the number of accelerators (beside CRT’s and smaller) in the World. https://www.youtube.com/watch?v=jLmciZdh5j4