Whenever anyone mentions the word “antibodies” these days, it’s sure to grab your attention. Thoughts generally flow to the human immune system and the role it plays in the ongoing COVID-19 pandemic, and to how our bodies fight off disease in general. The immune system is complex in the extreme, but pretty much everyone knows that antibodies are part of it and that they’re vital to the ability of the body to recognize and neutralize invaders like bacteria and viruses.
But as important as antibodies are to long-term immunity and the avoidance of disease, that’s far from all they’re good for. The incredible specificity of antibodies to their target antigens makes them powerful tools for biological research and clinical diagnostics, like rapid COVID-19 testing. The specificity of antibodies has also opened up therapeutic modalities that were once the stuff of science-fiction, where custom-built antibodies act like a guided missile to directly attack not only a specific protein in the body, but sometimes even a specific part of a protein.
Making these therapies work, though, requires special antibodies: monoclonal antibodies. These are very much in the news recently, not only as a possible treatment for COVID-19 but also to treat everything from rheumatoid arthritis to the very worst forms of cancer. But what exactly are monoclonal antibodies, how are they made, and how do they work?
Poly vs. Mono
It helps to have a general idea of how the immune system works before diving into the specifics of monoclonal antibodies. Luckily, the basics are pretty straightforward. In a nutshell, there are two systems of immunity in the human body: the innate and the adaptive. Both are comprised of specialized white blood cells called lymphocytes. The innate immune system is a “fast attack” system, capable of quickly discerning friend from foe and digesting invaders. The remnants of these invaders, primarily the fragments of their proteins, are called antigens, and they are presented to lymphocytes of the adaptive immune system, which are studded with antibodies known as immunoglobulins. These antibodies are a highly diverse population of proteins that represent a chemical memory of antigens the body has encountered before. Any antigen that matches an antibody will bind to it, setting off a series of events that causes a huge build-up of lymphocytes bearing that antibody, which then seek out and destroy the invader with great specificity.
While antibodies produced by the adaptive immune system are highly specific for a particular antigen, there’s still some “wiggle room” in them. The antigens that are presented to the adaptive immune system are actually pretty large, and it’s entirely possible that antibodies exist to different features of the same protein, called epitopes. This is an advantage, since it increases the likelihood that the adaptive immune system will be able to recognize the antigen, no matter how chopped up it gets by the innate immune system. It’s also helpful in case the virus mutates, which might change an epitope enough to no longer be recognized. Antibodies to multiple epitopes, which are called polyclonal antibodies, provide redundancy and help increase the chances of a robust immune response.
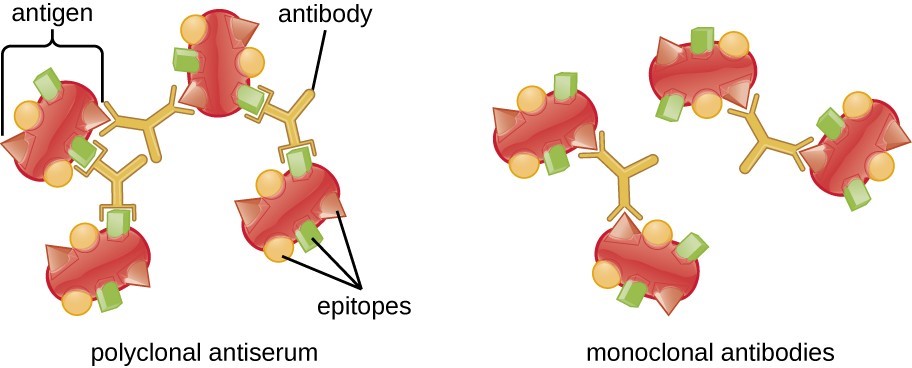
While so far I’ve described how the immune system works in your body, it’s important to keep in mind that antibodies are also powerful tools for research and for clinical diagnosis. The ability to create a population of antibodies that can bind to a specific protein has been a huge boon to biological research. Creating antibodies usually involves injecting a rat or a mouse with the target antigen, harvesting the animal’s white blood cells, and purifying the antibodies to get just those that bind to the antigen of interest. It’s a process that requires a lot of time — three months or more is not uncommon — and a lot of specialist skill. The end result is a set of polyclonal antibodies.
While traditional antibody production techniques have done a lot of the heavy lifting in biomedical research over the years, there are times when the polyclonal antibodies they produce are just not the tool for the job. In some cases, more specificity in binding is needed, which is what monoclonal antibodies are for. Monoclonal antibodies, often abbreviated “mAbs”, are antibodies that recognize a single epitope of the target antigen. This can be an incredibly powerful tool for research, as antibodies can be created that bind to just a single region of a large protein. This can be used to explore the function of that region; for example, a monoclonal antibody that binds only to a specific epitope can be used to probe whether that region is a binding site for another protein by physically blocking access to it.
Cancer Cells Made Good
While the natural immune system is basically a polyclonal antibody factory, that’s not to say that monoclonal antibodies don’t show up naturally. Unfortunately they are most often seen in patients with multiple myeloma, a blood cancer that affects the blood cells involved in immunity. All cancers are characterized by overproduction of a particular tissue type, and natural myeloma cells gave researchers a tool for creating massive populations of identical cells — clones — that each produced a single antibody.
The trick, though, was to figure out how to program the myeloma cells to produce a specific antibody, and in the 1970s when all this was first being explored, molecular biology was still in its infancy. Researchers therefore had to hack around a bit to make this happen. The basic idea they came up with was to fuse together immortal myeloma cells with cells bearing antibodies raised up against antigens of interest. These hybridoma cells each carry the genes for a single antibody, and can be grown in abundance once the work of creating them is done.
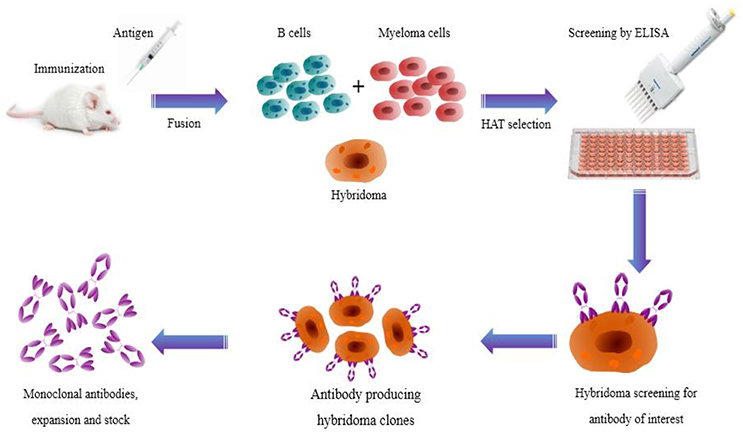
Hybridoma cells are tricky to produce. First there’s the problem of growing up a lot of antibody-producing cells, called B cells, in mice or other mammals. Then the B cells have to be fused to the myeloma cells, either chemically by treating the cells with polyethylene glycol, or by using an electric field. There’s also the problem of separating the few cells that successfully fuse together from the unfused myeloma and B cells. This is accomplished by using myeloma cells that are sensitive to the chemotherapy agent aminopterin. Myeloma cells that successfully fuse will be “rescued” by the intact version of the gene in the B cells, and will be able to live in a growth medium containing the drug.
The next trick is to turn the polyclonal population of hybridoma cells into a number of monoclonal populations. This is done by diluting the surviving fusion cells to the point that, on average, there’s only a single cell in a given volume of growth medium. Placing that volume in each well of a microtiter plate allows the single cell to grow, producing a clonal cell line that carries the genes to create antibodies against a single epitope. Finding the cell line that reacts with a specific epitope is then a matter of screening all the hybridoma lines using a variety of immunochemical methods, like fixing the antigen to a solid substrate and using that to bind only the hybridomas that are specific for it.
In the decades since hybridoma techniques were first explored, many other methods for mAb production have been developed. Phage display, where viruses that infect bacteria are modified to express antibodies on their protein coats, can be used to screen large numbers of antibodies against a specific antigen, and then have the DNA that codes for the antibodies readily available for cloning. Transgenic mice which bear genes for human antibodies can also be used to produce libraries of mAbs; almost all of the currently approved monoclonal antibody therapeutics on the market today were developed via transgenic mice.
The Guided Missiles of Medicine
From the above example of using mAbs to probe binding domains on proteins, it’s a pretty short leap to seeing how they can be used not only diagnostically, but therapeutically. The specificity of monoclonal antibodies makes for extremely targeted therapies, as opposed to, say, taking a drug orally and hoping for it to be transported through the entire body to the therapeutic target. This is where monoclonal antibodies get their well-earned reputation as the “guided missiles” of medicine.
The missile metaphor starts to break down a bit when you consider how mAb therapies work, though. Where a missile generally needs to deliver a warhead to be effective, in mAb therapy, sometimes just the fact that they can bind to specific proteins is weapon enough. This is particularly the case with therapies for autoimmune diseases like Crohn’s disease and rheumatoid arthritis, where the patient’s own immune system mistakes their own cells for invaders and starts attacking them.
These diseases are treated with mAbs that target and bind to tumor necrosis factor-alpha (TNF-α), a protein that regulates the immune system, and turns down the self-destructive cascade. Some cancer therapies use a similar approach, with mAbs binding to certain proteins that regulate cell division, thereby targeting them for cleanup by the patient’s immune system.
But still, some mAb therapies also carry a payload to their target. In some cases, a radioisotope can be connected to the mAb to deliver a dose of radiation directly to cancer cells; other mAbs can deliver with molecular precision either drug molecules or pro-drug molecules which can later be converted to their active form.
Monoclonals for COVID
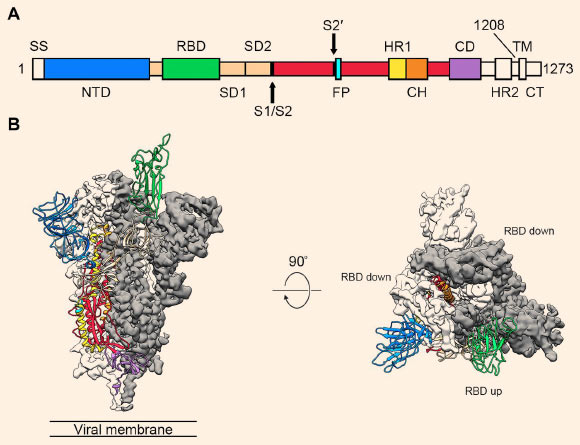
All this brings us to the current pandemic, and how monoclonal immunotherapy is being leveraged to help COVID-19 patients. The US Food and Drug Administration has so far authorized three mAb immunotherapies for COVID-19 under Emergency Use Authorizations (EUA). Two of them are currently recommended, and are being used for patients hospitalized with mild to moderate COVID-19. One is a cocktail of two mAbs, called casirivimab and imdevimab — you can tell a drug is a monoclonal antibody by the –mab suffix — which is interesting because while each mAb binds to the fabled spike protein of the SARS-CoV-2 virus, they each bind to different but overlapping epitopes of the protein.
Both epitopes are in the receptor-binding domain (RBD) of the spike protein, meaning that once the mAbs bind to the spike proteins on a circulating virus particle, it’s no longer able to bind to its receptor, the ACE-2 receptor’s lining the epithelium of the respiratory tract. The upshot is that the virus is no longer able to enter cells and replicate, which hopefully reduces the viral load and leads to quicker recoveries.
For casirivimab/imdevimab, which is marketed by Regeneron under the name REGEN-COV, the numbers look pretty good — giving the mAb therapy, which is either an IV infusion or a set of four subcutaneous injections, within the first ten days of symptoms reduces the risk of a mild to moderate case of COVID progressing by about 70%. It also has indications for prophylactic use post-exposure for immunocompromised individuals.
Monocolonal antibodies have been a game-changer in biological research, and in the treatment of a wide range of dire diseases. The development of new techniques to make them easier to manufacture should broaden their use as the guided missiles of medicine.















i love reading journal articles on the biochemistry of the immune system. it’s always astonishing how much is known, how many complicated tests are possible. but then it’s a little sobering to realize, while sometimes they really understand the process, most of the time it’s more like figuring out why the computer crashes more often by counting the number of conditional branch instructions in core. it’s like a side-channel attack on a machine that you don’t quite know enough about yet.
Agreed. I also marvel at the thought that the “machine” being studied is different from the one before and the one after. You have to study a large enough number to start finding ones that branch in the same way. What a debugging challenge!
“guided missiles of medicine” Indeed very specific.
I was wondering how bats (25% of all mammal species are bats) evolved to be such a virus carrier and how their own immune system is coping with the quantity of different viruses they carry, and i found this interesting article:
https://www.nytimes.com/2020/01/28/science/bats-coronavirus-Wuhan.html
In short, the energy needed for flying is so intense some body cells break and their DNA is released, Normally, mammals immune system interprets this as disease caused and responds with inflammation.
Bats have actually weakened their immune response to react less to virus infection, and became carrier of many viruses without being sick.
That seems il-logic explanation. Where is the cells breakage limit? By the third generation, natural selection would be lacked of bodies to work with OR they should reduce energy consumption by, pardon colloquial, sitting more on their asses?
Not necessarily;
Bats have lost some genes involved in that response, which makes sense because the inflammation itself can be very damaging to the body. They have a weakened response but it is still there. Thus, the researchers write, this weakened response may allow them to maintain a “balanced state of ‘effective response’ but not ‘over response’ against viruses.”
Remember that the most famous RESPIRATORY viruses generally don’t do that much – It’s the overreaction of your immune system that turns certain flu strains (1918) and the various coronaviruses of the last couple decades into killers. You wouldn’t be drowning in your own fluids if the battle wasn’t fought to the death in your lungs.
Same with autoimmune diseases – It’s not that your immune system is weak, it’s that it has severely misplaced its priorities.
“Remember that the most famous RESPIRATORY viruses generally don’t do that much – It’s the overreaction of your immune system that turns certain flu strains (1918) and the various coronaviruses of the last couple decades into killers. You wouldn’t be drowning in your own fluids if the battle wasn’t fought to the death in your lungs.”
HydrogenSulfide gas is similar. Highly deadly in small amounts because of the body’s response.
Emergency treatment involves an atropine injector to counter the response.
Thanks for simplifying the topic so well!
I’ve noticed a lot of drugs in the media lately have the -mab suffix, and was wondering what that signified. And now I know!
And if you wonder “we have some genes that make something, but how can we just make that thing?” then this guy explains it pretty well. Yes, he plays kaizo Mario, but he also helped make a potential new HPV vaccine by making a plant virus that causes a plant to make empty HPV shells. Complete, but harmless, virus shells give the immune system some practice with what the actual virus “looks” like.
I don’t know where the video went on his Mario channel where he discussed the immune responce titers of the mice. But the published results were really strong.
https://youtube.com/channel/UC5KJaXK5Yn7P-2RX-xk4RpA
They don’t go into monoclonal antibodies, but there’s some excellent layman-explanation of CRISPR cas-9 and cas-13 in “The Code Breaker”, and it goes into some of the ways the critical research that led to Covid-19 vaccines developed more-or-less directly from curiosity-driven research into RNA. The middle bogs down into the business side of the research industry, but the first and last thirds I loved.
I don’t know anything about this stuff, so it’s interesting.
Is the COVID-19 test specificity as discriminating as our antibodies? With over 1 million people being tested daily and a few hundred deaths being attributed to the virus daily, does that mean the test specificity is >99.999%?
Seems like it is more like a Roman pilum than missle…