Solid state batteries, we are told, are the new hot battery technology that will replace lithium-ion batteries. Soon. Not that we haven’t heard that before. One reason it isn’t dominating the market today is that it’s prone to short circuits during charging. [Dr. Yuwei Zhang and others have published a paper detailing why the shorts happen, which could lead to strategies to improve the technology.
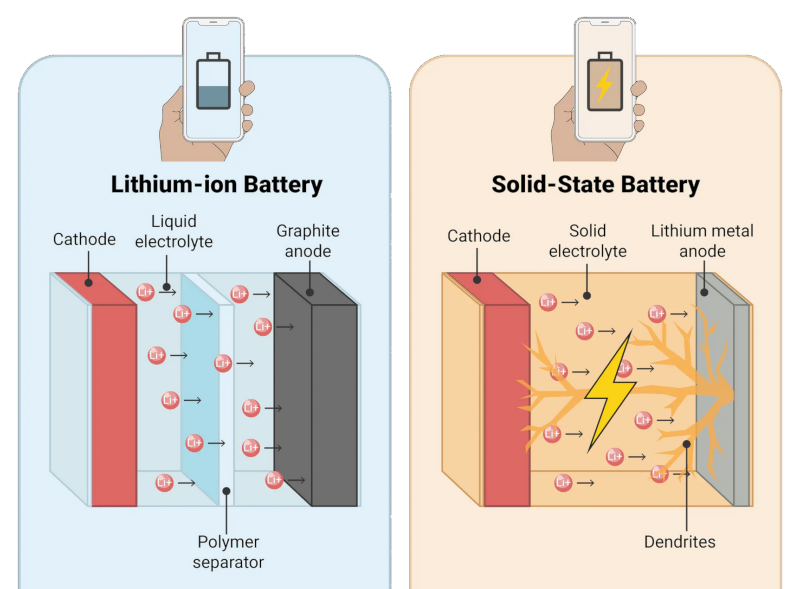
Solid state batteries employ a solid electrolyte and a lithium anode. It is known that, sometimes, lithium metal from the anode forms dendrites that penetrate the ceramic electrolyte and cause it to crack. This is somewhat of a mystery as the lithium is a soft metal: to quote [Zhang], “like a gummy bear”.
There were two leading hypotheses for the observations. [Zhang’s] team showed that hydrostatic stress made the lithium dendrites act like a water jet, enabling them to penetrate the hard ceramic.
There is still work to figure out what to do about it, but understanding the root cause is certainly a step in the right direction. We’ve looked at these batteries before. We’ve also seen how changing the anode construction might help with the problem.















There are numerous technical challenges with solid state batteries, including the fact that without a liquid electrolyte the layers can become mechanically separated, so they need to be packed and compressed together tightly. That in turn creates the issue where the electrodes grow and shrink physically when the ions move in and out, so there’s tremendous cyclical mechanical stresses inside the battery that tend to break the structure after a while. That’s what’s behind the “hydrostatic stress” mentioned in the article.
This seems backwards. Lithium batteries with liquid electrolytes and plastic separator membranes have been extremely well documented as having problems with dendrite growth piercing the plastic separator and causing shorts that lead to runaway heating. Solid state batteries on the other hand, with their solid electrolytes are supposedly so resistant to dendrite growth that they’ve switched to solid lithium metal electrodes instead of lattices which merely hold ions, which is where solid state batteries generally get their doubling of capacity vs traditional cells.
There are solid state Lithium battery on the market. It shouldn’t be hard to cut one open to figure out how the manufacturer solved the issue.
There are zero all solid state batteries in the market, there are semi solid state batteries as manufacturers backport new tech, sometimes marketed as solid state but they are in fact not without liquid electrolyte. And if you mean donut lab, that hasn’t even been proven to not be a scam yet and is most likely just a good NMC pouch.
True solid state batteries are also a bit overhyped as well. For instance, they’re not supposed to burn; in reality they just don’t burn quite as easily or quite as violently as liquid electrolyte cells. You still have reactive materials inside the cell, which will generate heat spontaneously when combined – because the battery essentially discharges into itself.
The trick: though the heat released is only 30% of regular lithium cells, when you increase the energy density by 2-3x compared to regular lithium, it burns just as violently. It may take 200 C instead of 90 C to reach the point of self-combustion, but once it does it’s still the same problem: it burns like gunpowder and cannot be put out by spraying water on it, because that just makes it angry.
D’abord passer l’objet au scanner ( rayon x basique ou modèle densitométrique ) et ensuite vous la cassez .
The dendrite growth in liquid electrolytes has a different mechanism – it’s essentially lithium crystals growing out of the SEI like dandelions growing through asphalt.
In solid state batteries, the dendrite growth is a result of the solid electrolyte forming tiny cracks due to mechanical stresses, and the lithium metal squeezing into those cracks until it bridges the cell.
I wonder if they could use a solid electrolyte that is easy to melt and increase cycle life by periodically heating the battery to melt it.
Would the melting cause the materials to flow around more easily, causing the lithium to mix into the electrolyte and ruin the cell?
It would probably release the mechanical compression that keeps the cell together and ruin it anyways.
That explanation makes way more sense, brittleness is inherent to ceramics.
Anyone who has seen a dandelion grow through concrete knows why those dendrites are getting through lol
Current use of ‘solid state battery’ is misleading: (Hydrated) ions are still the charge carriers.
https://www.freepatentsonline.com/y2023/0029346.html suggests a real ‘solid state device’
IMHO the only surefire way is to find an electrolyte that’s at least slightly more conductive (or “transportive”) than lithium metal. This way lithium will start depositing at the deepest parts and naturally smooth out.