The flow battery is one of the more interesting ideas for grid energy storage – after all, how many batteries combine electron current with fluid current? If you’re interested in trying your hand at building one of these, the scientists behind the Flow Battery Research Collective just released the design and build instructions for a small zinc-iodide flow battery.
The battery consists of a central electrochemical cell, divided into two separated halves, with a reservoir and peristaltic pump on each side to push electrolyte through the cell. The cell uses brass-backed grafoil (compressed graphite sheets) as the current collectors, graphite felt as porous electrodes, and matte photo paper as the separator membrane between the electrolyte chambers. The cell frame itself and the reservoir tanks are 3D printed out of polypropylene for increased chemical resistance, while the supporting frame for the rest of the cell can be printed from any rigid filament.
The cell uses an open source potentiostat to control charge and discharge cycles, and an Arduino to control the peristaltic pumps. The electrolyte itself uses zinc chloride and potassium iodide as the main ingredients. During charge, zinc deposits on the cathode, while iodine and polyhalogen ions form in the anode compartment. During discharge, zinc redissolves in what is now the anode compartment, while the iodine and polyhalogen ions are reduced back to iodides and chlorides. Considering the stains that iodide ions can leave, the researchers do advise testing the cell for leaks with distilled water before filling it with electrolyte.
If you decide to try one of these builds, there’s a forum available to document your progress or ask for advice. This may have the clearest instructions, but it isn’t the only homemade flow cell out there. It’s also possible to make these with very high energy densities.






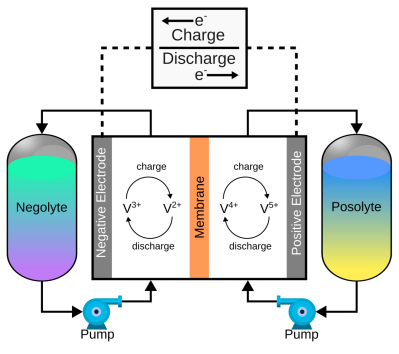 of a pair of electrolytes. These are held externally to the cell and connected with a pair of pumps. The capacity of a flow battery depends not upon the electrodes but instead the volume and concentration of the electrolyte, which means, for stationary installations, to increase storage, you need a bigger pair of tanks. There are even 4 MWh containerised flow batteries installed in various locations where the storage of renewable-derived energy needs a buffer to smooth out the power flow. The neat thing about vanadium flow batteries is centred around the versatility of vanadium itself. It can exist in four stable oxidation states so that a flow battery can utilise it for both sides of the reaction cell.
of a pair of electrolytes. These are held externally to the cell and connected with a pair of pumps. The capacity of a flow battery depends not upon the electrodes but instead the volume and concentration of the electrolyte, which means, for stationary installations, to increase storage, you need a bigger pair of tanks. There are even 4 MWh containerised flow batteries installed in various locations where the storage of renewable-derived energy needs a buffer to smooth out the power flow. The neat thing about vanadium flow batteries is centred around the versatility of vanadium itself. It can exist in four stable oxidation states so that a flow battery can utilise it for both sides of the reaction cell.










